-
Call Now
1800-102-2727
Law of Definite Proportion: Statement, Example and Limitations of Law of Definite Proportion, Practice Problems & FAQs
Have you ever watched any cooking recipe video?
Well, we all love watching it and enjoying the dish in imagination.
In the last segment of the video they do tell us about the ingredients and their ratio.
What will happen if we don’t follow the ratio mentioned in the ingredient table?
Will it make the same dish or something else will be formed?
The answer is very subjective, if you don’t deviate too much, then you won’t even notice the change.
But this doesn’t apply while forming compounds. They always form in a constant ratio. Hence, it is important for us to understand one of the important laws of chemistry and that is law of definite proportion.

TABLE OF CONTENT
- Law of Definite Proportions
- Examples of law of definite proportions
- Limitation of the law of definite proportion
- Practice Problems
- Frequently Asked Questions
Law of Definite Proportions:
The individual elements that make up a chemical compound are always present in a fixed ratio, according to the law of definite proportions, also known as the law of constant proportions (in terms of their mass). This ratio is independent of the chemical compound's origin or the process used to make it.
- Chemical compounds are made up of components that are present in a fixed ratio by mass, according to the law of constant proportions. This indicates that the same components will always be present in the same proportion by mass in any pure sample of a compound, regardless of the source. For instance, the mass ratio of hydrogen to oxygen in pure water will never change. The ratio of hydrogen to oxygen in water is roughly 1:8, or 0.11 grams of hydrogen to 0.88 grams of oxygen.
- The law of constant proportions is also known as Proust's law or the law of definite proportions.
- Joseph Proust, a French scientist, developed the law of constant proportions in 1794 as a result of his research on sulfates, metallic oxides, and sulfides. The scientific community in the 18th century opposed this law vigorously. This law was supported by the development of Dalton's atomic theory, and in 1811, the Swedish chemist Jacob Berzelius discovered a connection between these two ideas

Examples of law of definite proportions:
- Hydrogen and oxygen atoms make up the chemical compound water, which is denoted by the symbol H20. Water is produced when one oxygen atom joins two hydrogen atoms.
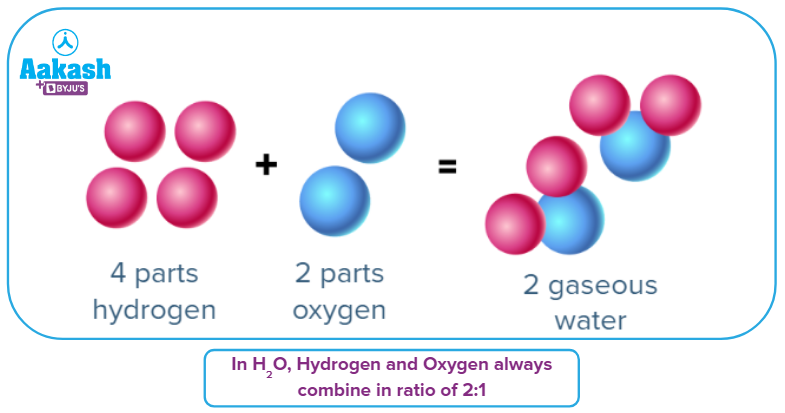
The atomic mass of a hydrogen atom is 1, while that of an oxygen atom is 16. As a result, water contains 89 percent oxygen and 11 percent hydrogen. Water can only be produced when hydrogen and oxygen are mixed in exactly the same ratio. 20 percent hydrogen and 80 percent oxygen cannot be combined to form water because the ratios are incorrect; doing so would result in the formation of a different chemical compound.
- Sodium (Na) and chlorine (Cl) are the two chemical elements that make up salt, also known as the compound NaCl . Salt can only be produced when sodium and chlorine are mixed in the exact same ratios.
- Hydrogen, sulfur, and oxygen are the constituents of sulfuric acid. The chemical is identified as H2SO4. To make sulfuric acid, the same ratios of hydrogen, sulfur, and oxygen must be combined.
- The elements hydrogen and nitrogen combine to form the common household product known as ammonia. The symbol NH3 denotes the presence of one nitrogen atom along with three hydrogen atoms. 82 percent nitrogen and 18 percent hydrogen make up anhydrous ammonia. A completely different chemical complex would result from any other hydrogen and nitrogen ratio.
- Chemically, glucose is denoted by the formula C6H12O6. Accordingly, a substance needs to contain 6 carbon atoms, 12 hydrogen atoms, and 6 oxygen atoms in order to be classified as glucose. The carbon makes up 40.001% of glucose; the hydrogen makes up 6.714% of glucose; and the oxygen makes up 53.285% of glucose. It wouldn't work if you tried to create glucose with the carbon and oxygen equally represented by combining 45% carbon, 45% oxygen, and 10% hydrogen; the chemical compound that would result would not be glucose.
- Vinegar is another common household item. Its chemical formula is C2H4O2, which indicates that it contains 2 carbon atoms, 4 hydrogen atoms, and 2 oxygen atoms. Vinegar couldn't be made with any other ratio of carbon, hydrogen, and oxygen.
No matter how much of a chemical compound is being produced, the Law of Definite Proportions ensures that the same proportions are always used.
Limitation of the law of definite proportion:
The law of constant proportions, though a foundation in the evolution of chemistry, does not apply to all chemical substances. The following is a list of limitations of the above law.
- Different samples of some non-stoichiometric substances have different elemental compositions. Instead, these compounds adhere to the law of multiple proportions.
- Wustite, an iron oxide with the chemical formula FeO, is one illustration of this. The proportion of oxygen to iron atoms can vary from 0.83 to 0.95.
- This is due to the crystallographic vacancies in the samples caused by a disorderly arrangement of atoms.
- The isotopic composition of a compound's constituent elements may differ between different samples. The mass ratios may change as a result of this.
Practice Problems:
Q1. 3.60 g of a certain metal burnt in the presence of air 5.4 g of its oxide is formed. How many g of the same metal should be heated in steam gave 3.2 g of its oxide. (these results illustrate the law of constant proportion).
A. 6.4 g
B. 4.5 g
C. 2.2 g
D. 3.5 g
Answer: (A)
Solution:
case 1:
mass of metal = 3.6 g
Mass of its oxide = 5.4 g
Weight of oxygen = 5.4 g - 3.6 g = 1.8 g
case 2:
Let, mass of metal = x g
Mass of its oxide = 3.2 g
Weight of oxygen = 3.2 g - x g
These results are according to the definite law of proportions. So,
According to question,
Q2. One mole of a Noble gas at 1 bar & 273 K occupies 22.7 liters. This experimental result was deduced from:
A. Law of gaseous volumes
B. Dalton’s atomic theory
C. Law of constant proportions.
D. None of these
Answer: (D)
Solution: according to Avogadro's hypothesis, an equal volume of any gas with the same number of moles of gas or the same number of the molecule (not same number of an atom) at same temperature and pressure.
Q3. Which of the following pairs illustrates the law of multiple proportions?
A. H2O & HCl
B. NO & NO2
C. H3PO4 & H2O
D. All of these
Answer: (B)
Solution: Statement of the law of Multiple proportions: When one type of atom combines with the other type of atom to form at least two or more different compounds, the mass of one atom, which combines with a constant mass of the other atom, bears a simple ratio to one another.
Example: Carbon and oxygen atoms combine to form two oxides NO and NO2
Case 1: In NO, 14 gm of carbon combined with 16 gm of oxygen.
The mass ratio of
In NO2, 14 gm carbon combined with 32 gm of oxygen.
Mass ratios of
Thus, we can see the mass of oxygen which combine with a constant mass of nitrogen (14 gm) bear simple ratio of or .
Q4. Did Dalton fail to explain which law?
A. Gay Lussac’s law of gaseous volumes
B. Law of mass conservation
C. Partial pressure of gaseous mixture
D. None of these
Answer: (A)
Solution:
According to experimental observation Gay Lussac’s law of gaseous volumes was
Hydrogen gas + Chlorine gas Hydrochloric acid
1 V 1 V 0
0 0 2 V
One unit volume of hydrogen gas combines with 1 unit volume of chlorine gas to form 2 unit volume of hydrochloric acid.
According to Dalton, atoms take part in chemical reactions:
Hydrogen gas + Chlorine gas Hydrochloric acid
1 atom 1 atom 0
0 0 2 hydrochloric vapor
For 1 hydrochloric vapor numerically we need ½ atom of hydrogen and chlorine gas both, according to Dalton's theory atoms are indivisible. At that time atoms and their free form of existence was not known.
Frequently Asked Questions-FAQs:
Q1. What is the purpose of a law of definite proportions?
Answer: The Law of Definite Proportions ensures that chemical compounds are always created using the same proportions, regardless of the amount of the compound which is being made.
Q2. What type of matter does not have definite proportions?
Answer: No, the Law of Definite Proportions applies only to compounds. When two or more elements are chemically bonded together, a compound is created.
Q3. Do chemical reactions proceed under the law of definite proportions?
Answer: The law of definite proportions, which states that a given chemical compound always contains the same elements in exactly the same proportions by mass, was quickly discovered after it was discovered that mass was always conserved in chemical reactions.
Q4. Why do mixtures not obey the law of constant composition?
Answer: A mixture is not chemically united, only physically. You can put as much of one thing in with the other because there is no limit, they are only mixing physically. Compounds, or elements that are chemically combined, are subject to the law of definite proportions.


