-
Call Now
1800-102-2727
Cupric Chloride-Introduction, Preparation, Properties, Uses, Practice Problems, FAQs
Have you ever visited any laundry for washing your clothes?
I guess most of you must have seen laundry men having a marker on his ear! Isn’t it? What do you think about that marker? This marker is used to give identification on clothes or to make designs.
Well, the marker used by the laundry man contains a most useful chemical element. Can you guess what is that chemical present in the laundry ink?
QThe chemical present in the laundry ink is cupric chloride. Yes, cupric chloride is a major compound of copper.

Copper has a connection to our historical era as well. Between 7,000 and 10,000 years ago, our ancestors made the life-changing discovery that copper is malleable, retains a sharp edge, and can be fashioned into tools, decorations, and weapons more readily than a stone.
Let’s study the important compounds derived from copper i.e, cupric chloride and see how beneficial they are. So come and let’s begin!
TABLE OF CONTENT
- Introduction
- Occurrence of Cupric Chloride
- Preparation of Cupric Chloride
- Properties and Reactions of Cupric Chloride
- Uses of Cupric Chloride
- Practice Problems
- Frequently Asked Questions-FAQs
Introduction:
Cupric chloride is a chemical compound having the formula, CuCl2 . The anhydrous form is yellowish brown, but when it absorbs moisture, it turns blue-green and forms a dihydrate compound (CuCl2.2H2O)
Occurrence of Cupric Chloride:
Both the extremely rare anhydrous mineral tolbachite and the dihydrate eriochalcite, are found close to fumaroles. Some Cu mines have naturally occurring forms of copper (II) chloride. Mixed oxyhydroxide-chlorides, such as atacamite Cu2(OH)3Cl , found in the oxidation zones of Cu ore beds in arid climates, are the more prevalent (also known from altered slags) mineral of copper.
Preparation of Cupric chloride:
- Copper metal and most of the copper compounds like cupric oxide, cupric hydroxide, and copper carbonate react with conc. HCl. On crystallisation, the resultant solution yields green crystals of hydrated cupric chloride.
- Copper metal heated in excess of chlorine gas produces anhydrous cupric chloride as a dark brown mass.
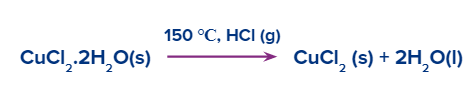
- Cupric chloride when heated in HCl gas flow, at 150°C, forms anhydrous cupric chloride.
- It is commercially viable to mix copper (II) oxide with an excess of ammonium chloride to create copper chloride, ammonia, and water.
Although hydrochloric acid cannot oxidise copper metal alone, it can react with copper-containing bases like copper (II) carbonate, hydroxide, or oxide to produce CuCl2.
Properties and Reactions of Cupric chloride:
- It is a deliquescent chemical that dissolves easily in water. The concentrated solution is green, but the dilute solution is blue in colour. In the addition of Conc. HCl , the solution turns yellow. When both are present, the blue colour is due to complex cation and the yellow colour is due to complex anion .
- Because of hydrolysis of cupric chloride, the aqueous solution is acidic in nature.
While, on strong heating, the hydrated salt produces CuO, Cu2Cl2,HCl and Cl2.
- Copper turnings, SO2 gas, hydrogen (nascent-obtained via the action of HCl on Zn), or SnCl2 are all good reducing agents, reducing cupric chloride to cuprous chloride (Cu2Cl2).
- When NaOH is added, a pale blue precipitate of basic cupric chloride i.e; CuCl2.3Cu(OH)2, is formed.
Uses of Cupric Chloride:
- Several procedures that create chlorine by oxychlorination use copper (II) chloride as a catalyst. At a temperature of 400 to 450 °C, copper chloride is used in the Deacon process. In Deacon's method, it acts as a catalyst.
- It's also utilised in medicine and in producing organic dyestuffs as an oxygen carrier.
- Co-catalyst in Wacker process: As a co-catalyst with palladium (II) chloride in the Wacker process, copper (II) chloride has significant industrial applications. In this procedure, water and air are used to transform ethylene (ethene) into ethanal (acetaldehyde). CuCl2 is used to reoxidize PdCl2 after it has been reduced to Pd during the process. The cycle can then be completed by air oxidising the resulting CuCl back to CuCl2.
- Other organic synthetic applications:
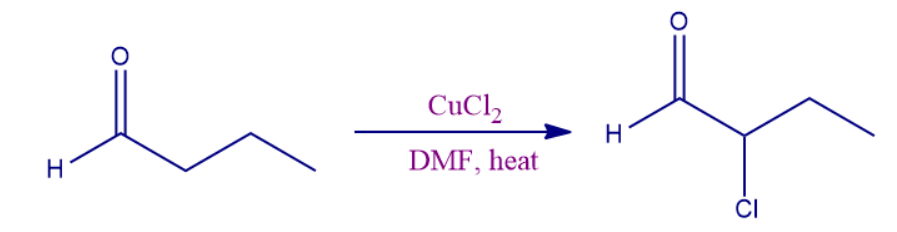
This is a kind of substitution reaction. This reaction takes place in a polar solvent like dimethylformamide (DMF), frequently with lithium chloride present to speed up the process.
- In pyrotechnics, copper (II) chloride is also employed as a blue/green colouring ingredient. Like all copper compounds, copper chlorides exhibit a green-blue flame when tested.
Practice Problems:
Q1. CuCl2 on dissolving in ammonium hydroxide gives a ___________ solution.
A. red
B. deep blue
C. light green
D. pale yellow
Answer: B
Solution: CuCl2 forms a deep blue solution when it dissolves in ammonium hydroxide. Deep blue tetramine cupric chloride crystals are formed by evaporating this solution.
Q2. _____________ is the oxidation state of Copper in cupric chloride.
A. 0
B. +1
C. +2
D. +3
Answer: C
Solution: Let the oxidation state of copper in CuCl2be x.
For calculating this we know that the oxidation state of the oxygen we consider is -2.
Now, calculating this we get,
Hence, +2 is the oxidation state of Copper in CuCl2 .
Q3. Anhydrous form of CuCl2 is ______________, but it slowly takes on moisture to become a dihydrate that is _________.
A. Yellowish brown and blue-green respectively.
B. Blue-green and yellowish brown respectively
C. Brown-red and blue-green respectively.
D. Brown-red and yellowish brown respectively.
Answer: B
Solution: The chemical compound with the formula CuCl2 is copper (II) chloride. The anhydrous form is yellowish brown, but it slowly takes on moisture to become a dihydrate that is blue-green.
Q4. _______________ is used along with CuCl2 as a co-catalyst.
A.SnCl2
B.PdCl2
C. PtCl2
D. ZnCl2
Answer: B
Solution: As a co-catalyst with palladium (II) chloride in the Wacker process, copper (II) chloride has significant industrial applications. In this procedure, water and air are used to transform ethylene (ethene) into ethanal (acetaldehyde). CuCl2 is used to reoxidize PdCl2 after it has been reduced to Pd during the processes.
Frequently Asked Questions-FAQ
Q1. Exactly how harmful is copper chloride?
Answer: When breathed in, copper chloride will have an effect on you. Touch has the potential to seriously irritate and burn, resulting in possible eye and skin damage. The skin might thicken as a result of repeated exposure. Breathing in copper chloride may irritate the nose, throat, and lungs, resulting in coughing and wheezing.
Q2. What takes place when water and copper chloride interact?
Answer: Blue-green in colour, copper (II) chloride is solid. The crystals turn vivid green when submerged in water, and the water above the crystals takes on a faint blue tint. The crystals break down and create a pale blue solution when swirled.
Q3. Is burning copper chloride safe?
Answer: The undamaged skin is only faintly irritated by copper chloride. If substantial quantities of copper chloride come into contact with open wounds and burns, there is a chance of tissue damage and systemic copper uptake.
Q4. Is copper chloride acts as a salt?
Answer: Cupric Chloride is another name for copper (II) chloride. It is an inorganic salt and can be considered as the product of a neutralisation reaction between a base say cupric hydroxide and an acid hydrochloric acid. This salt is employed as a chemical synthesis catalyst. Because of the salt's blue/green colour, we also employ Copper (II) chloride in the production of pyrotechnics.


