-
Call Now
1800-102-2727
Coordinate Bonding: Covalent Bond, Coordinate covalent Bond, Characteristics, Examples of Coordinate Covalent Bonds and Properties of Coordinate Compounds, Practice Problems and Frequently Asked Questions
A covalent bond has a subtype called a coordinate bond. It's a type of covalent bond in which just one atom shares the electron pair. In other words, the electrons in the shared pair are both from the same atom. Coordinate bonds may also be referred to as Dative bonds or dipolar bonds. A coordinate bond is also known as a Coordinate Covalent bond since it is a special type of covalent bonding. It can also be made using metal ions and ligands. There is a type of covalent bond called coordinate bond, in which one atom shares the electron pair. It may also be called a Dative bond or dipolar bond or even a coordinate covalent bond. This is also the type of bonding we see in metal ions and ligands.
Coordination compounds are widely used compounds in a wide range of industrial applications as well as biological functions. There is a central metal atom present in these compounds, which is then connected to various ligands that surround it. Vitamin B12, haemoglobin, chlorophyll, and pigments are some of the most common examples of coordination compounds. They are also commonly used as catalysts in a variety of processes.
Table of content:
- Covalent Bond
- Coordinate Covalent Bond
- Characteristics of coordinate covalent Bond
- Examples of coordinate covalent bonds
- Properties of coordinate compounds
- Practice problems
- Frequently asked questions(FAQs)
Covalent Bond:
Before understanding the concept of the Coordinate bond, let’s recall everything about the covalent bond.
In simple terms, covalent bonding is the mutual sharing of valence electrons between atoms in order to achieve the noble gas configuration of the individual atoms involved.
The electrostatic force of attraction holds individual atoms together in a covalent bond. The positively charged nuclei of the linked atoms and the negatively charged electrons they share have an electrostatic attraction.
A covalent bond's shared pair of electrons is known as the bonding pair of electrons, and it results in the formation of a discrete group of atoms known as a molecule.
A covalent bond can form between two atoms of the same element or between elements in the periodic table that are near to each other. This bonding is most common between nonmetals, but it can also occur between nonmetals and metals.
Let’s gloss over the idea of a covalent bond first:
- It is the mutual sharing of valence electrons between atoms so as to achieve an octet/noble gas configuration in all participating atoms.
- The covalent bond is essentially an electrostatic force of attraction. The oppositely charged linked atoms are held together by an electrostatic attraction.
- The shared pair of electrons is called a “Bonding pair” of electrons. The formed entity is called a molecule.
- It can form between two unique atoms (heteroatomic) or even two similar atoms (homoatomic). While it is commonly observed between two non-metallic atoms, it can also occur between nonmetals and metals.

Coordinate Covalent Bond:
A coordinate bond (also known as a dative covalent bond) is a type of covalent bond (a shared pair of electrons) in which both electrons originate from the same atom.
Each atom contributes at least one electron to the formation of a simple or typical covalent bond, however this is not always the case. In a coordinate covalent bond, one atom supplies both electrons while the other atom supplies none.
The bonding of metal ions to ligands is an example of this type of bond. Nonmetals, on the other hand, can participate in this bonding.
A coordinate covalent bond is formed by the reaction of Lewis acid and base. Lewis bases or ligands are neutral compounds or ions with a lone pair of electrons. These lone pairs of electrons are given to metal ions. Metal ions function as Lewis acids (acceptors) or electron-deficient species.
Ammonia (NH3), water (H2O), and & halide ions (Cl-,Br-) are common ligands. Because they share electron pairs with the metal ion, ligands are considered Lewis bases. Metal ions are always positive because they have less electrons and are more attractive likely to attract lone pairs of electrons in Lewis bases (donors).
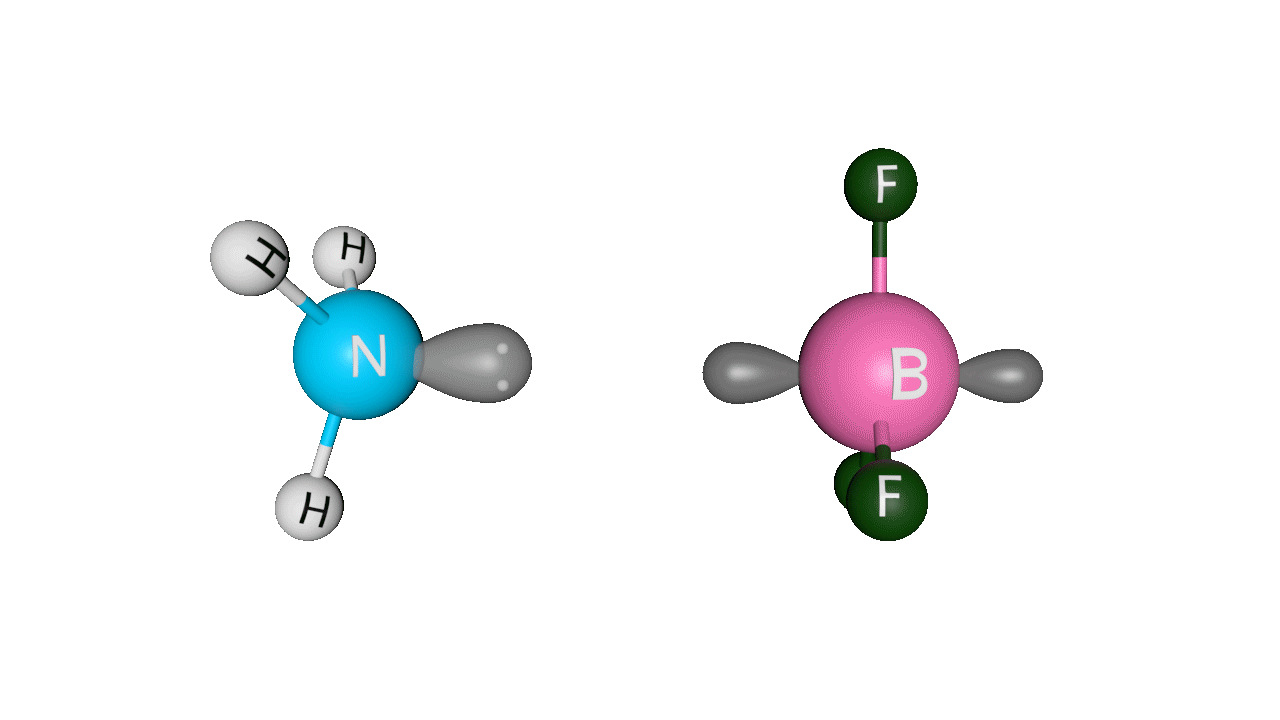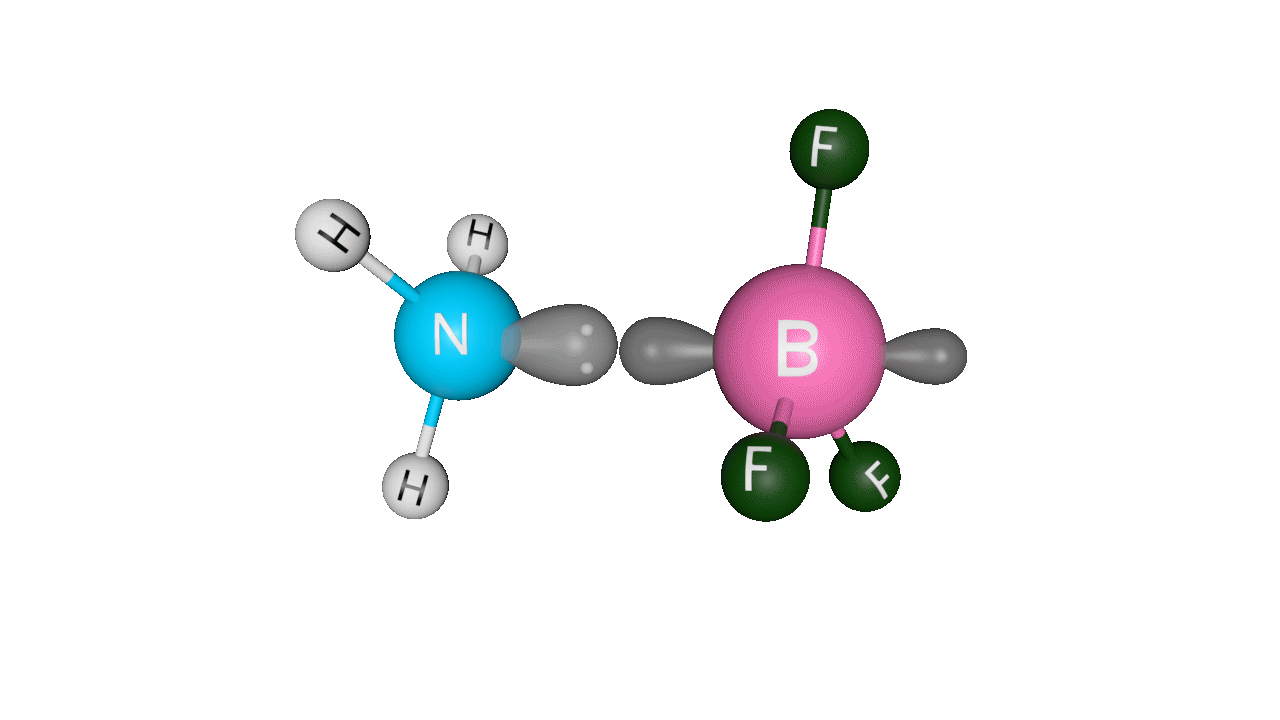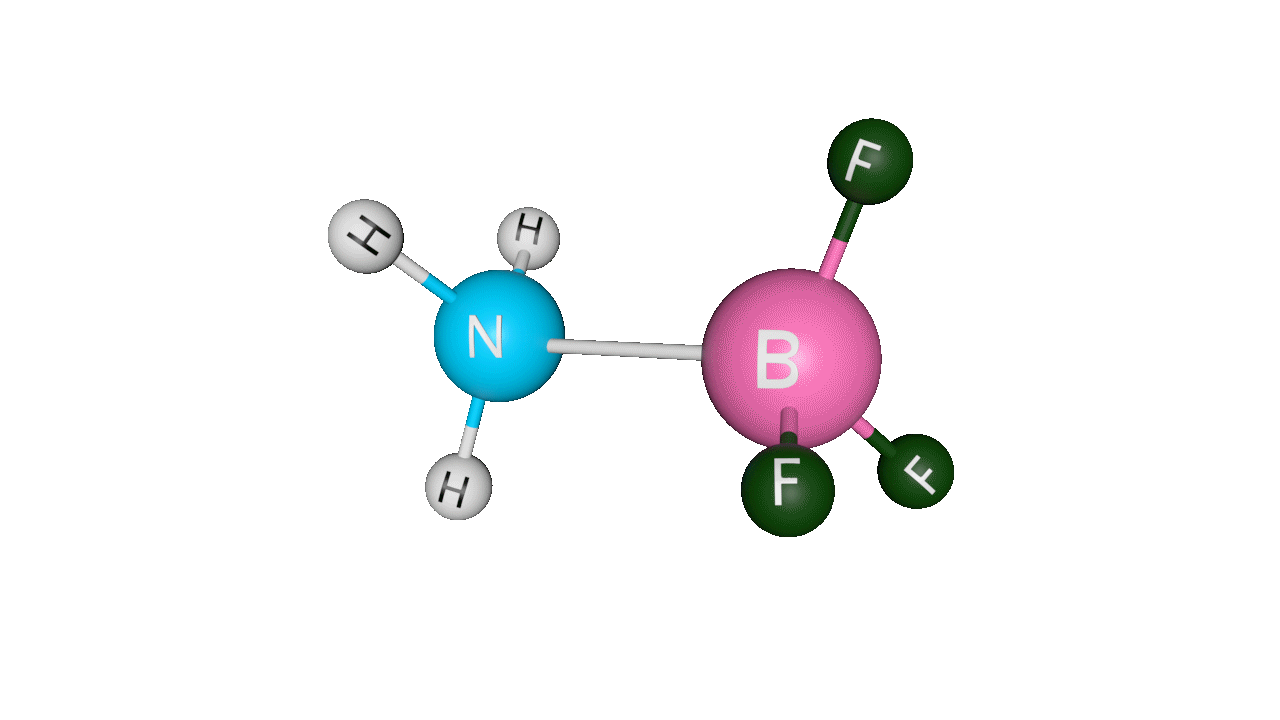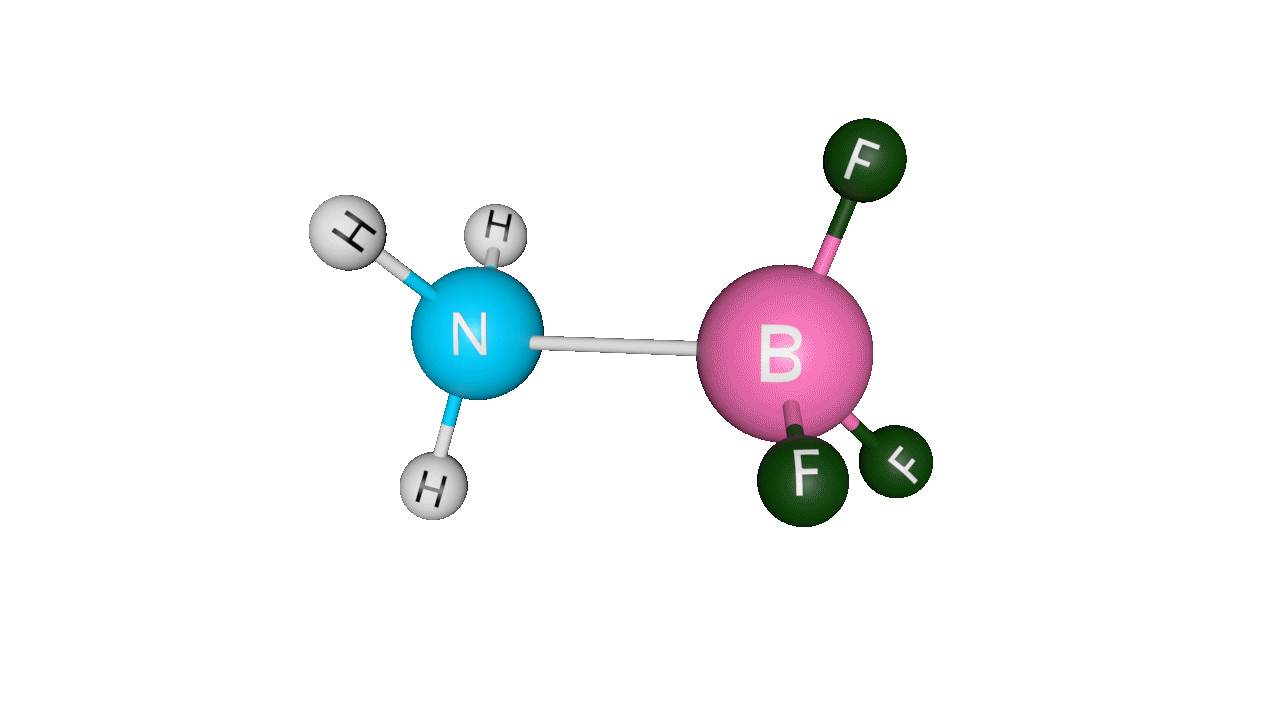
Let’s see the coordinate bonding between BF3 and NH3:

Characteristics of coordinate covalent bond:
- Coordinate covalent bonds are most commonly formed between different atoms.
- The atom or lewis base that donates an electron pair from itself is known as the donor in this bonding, while the atom or lewis acid that accepts the donated pair of electrons is known as the receptor or acceptor.
- In this kind of bonding, there are two species: the receptor/acceptor and the donor. As the names suggest, the donor donates the pair of electrons while the acceptor accepts the pair of electrons.
- This bond is represented by an arrow symbol pointing from the donor atom to the acceptor.
- The bond length and strength of the coordinate covalent bond are nearly identical to those of the covalent bond.
- Each atom gains stability after sharing an electron pair.
Examples of coordinate covalent bond:
Let's look at some examples of coordinate covalent bonds now:
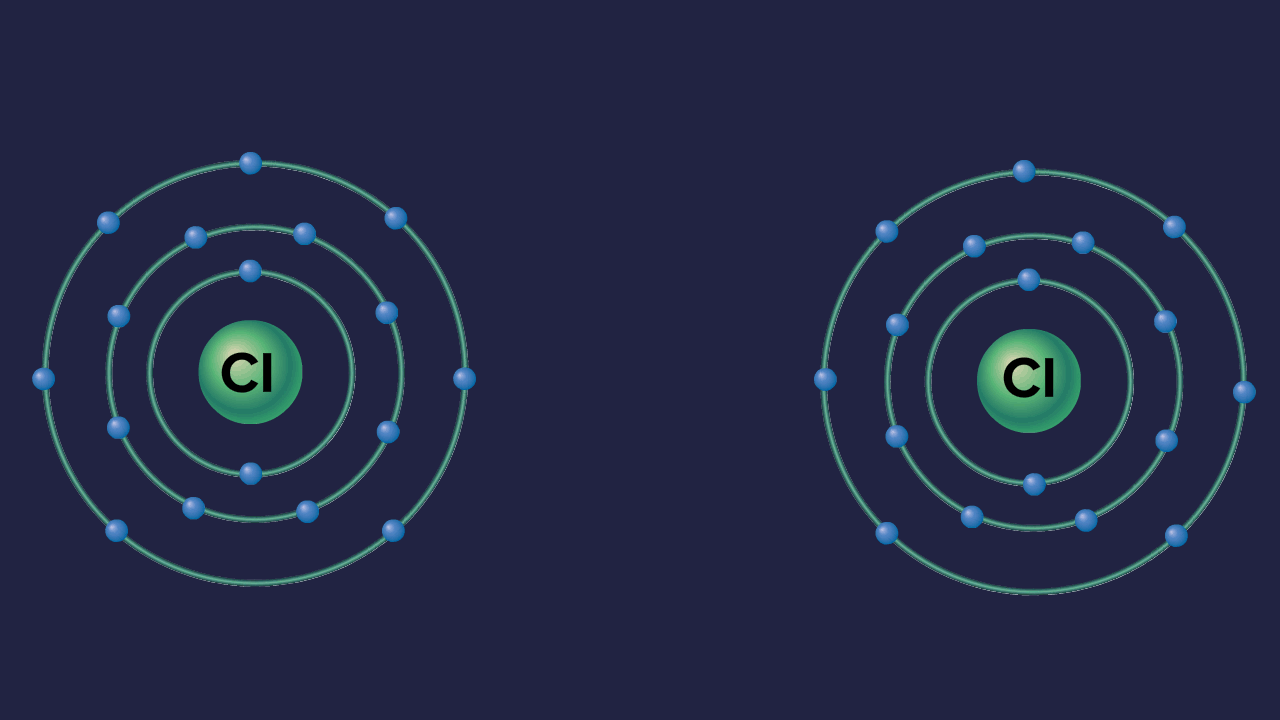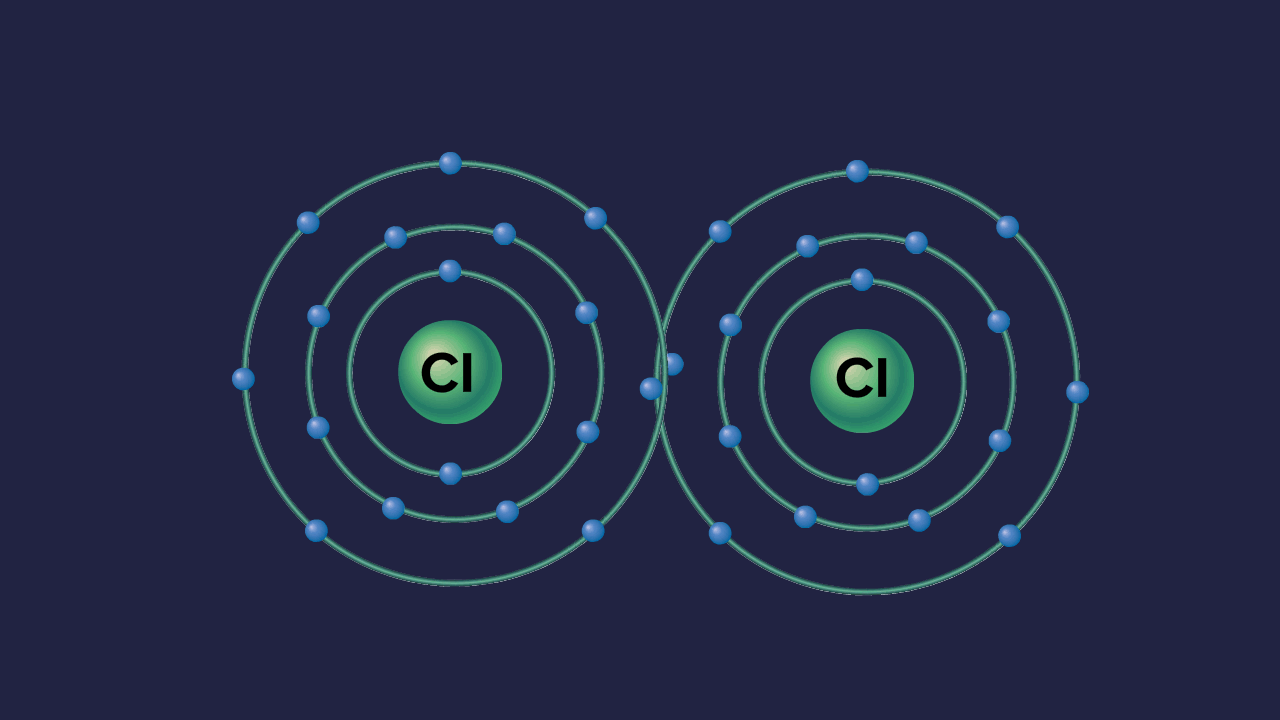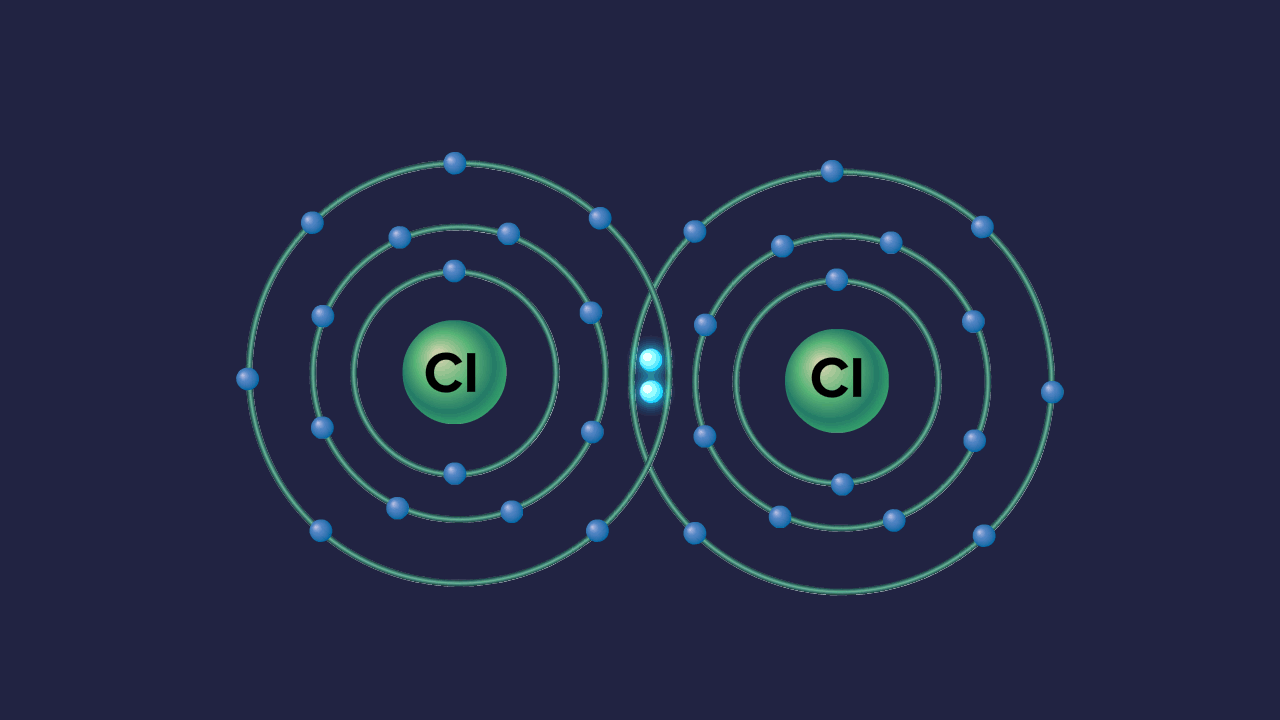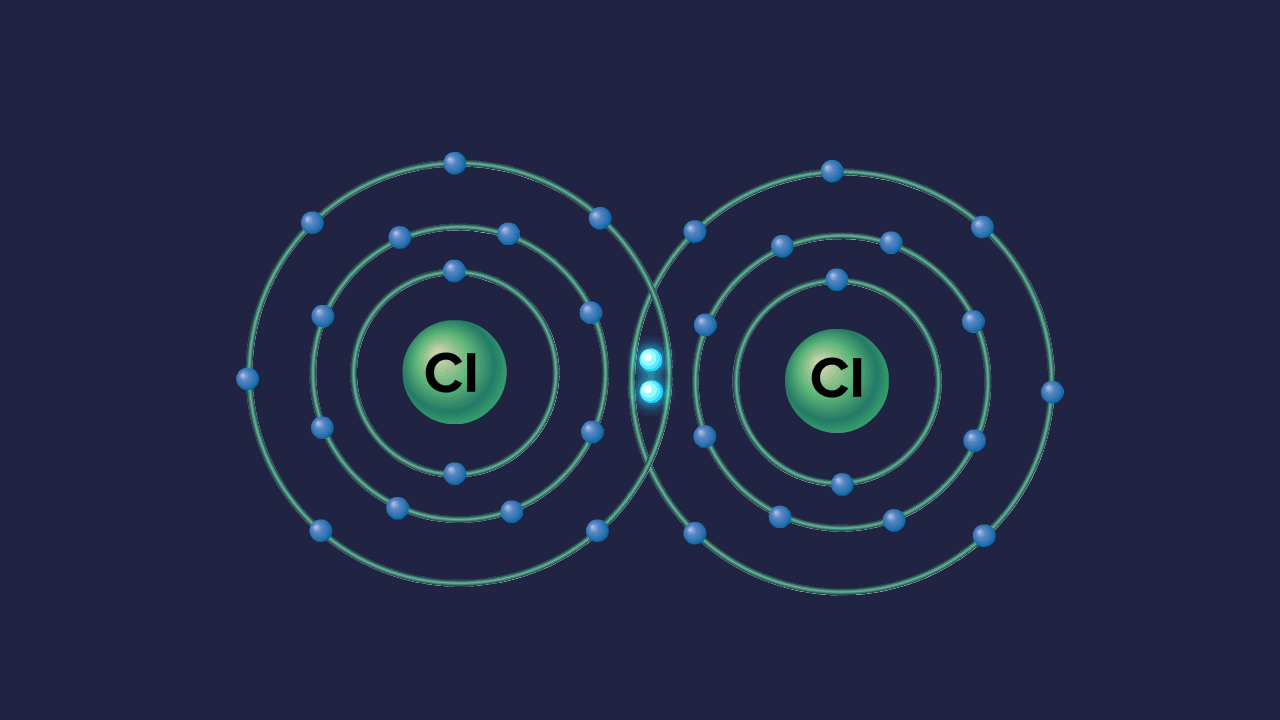
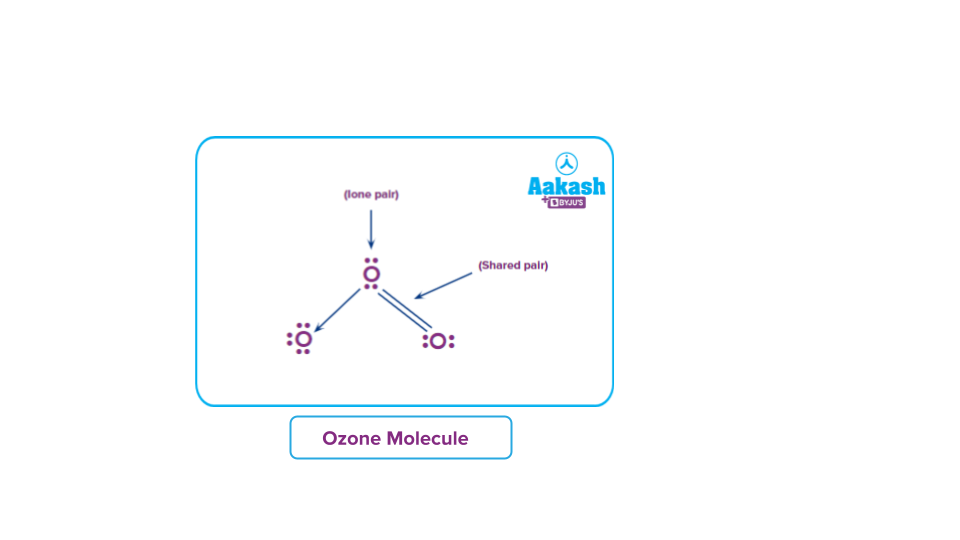
Formation of ozone molecule:
Each oxygen atom in O2 contributes one electron to each bonding pair of electrons. As a result, two 'regular' covalent bonds or a double covalent bond O=O are formed. In addition to the two bonding pairs of electrons, each oxygen atom has two nonbonding pairs of electrons known as lone pairs of electrons.
An oxygen molecule,O2, combines with an oxygen atom in the atmosphere to form an ozone molecule, O3.
Each oxygen atom in an O2 molecule has the noble gas Neon's stable electronic configuration. As a result, the only way for a third oxygen atom to join the oxygen atoms in O2 is one of the oxygen atoms in O2 to contribute one non-bonded electron pair to the new oxygen atom. As a result, a coordinate covalent connection is formed between the existing O2 molecule and the new oxygen atom:
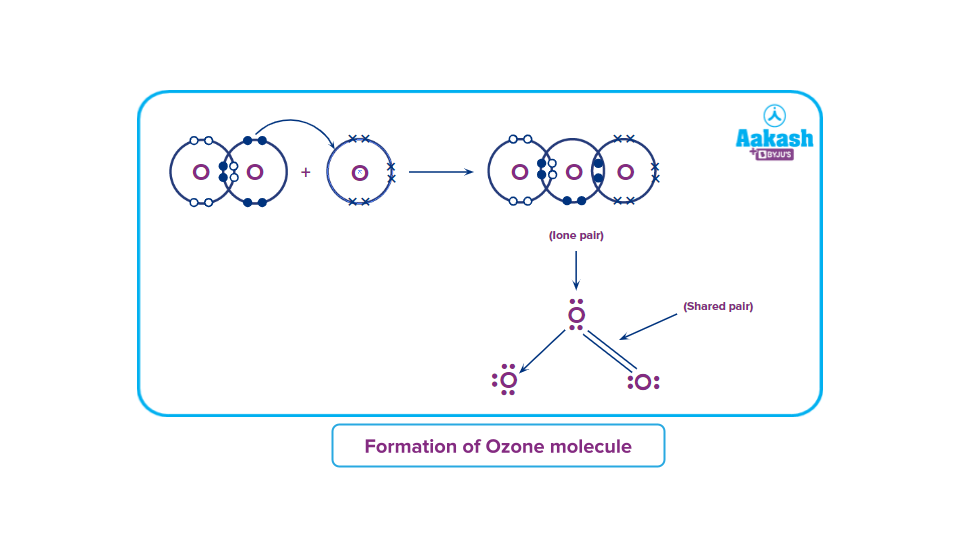
One of the oxygen atoms from the original O2 molecule, depicted in black dots in the diagram above, has provided one pair of its non-bonded pair of electrons to the third oxygen atom (denoted by X).
Because the X oxygen atom contributes none of its existing electrons to this bonding, it is a coordinate covalent bond (dative bond). However, once formed, the coordinate covalent bond is no different from a 'regular' covalent bond.
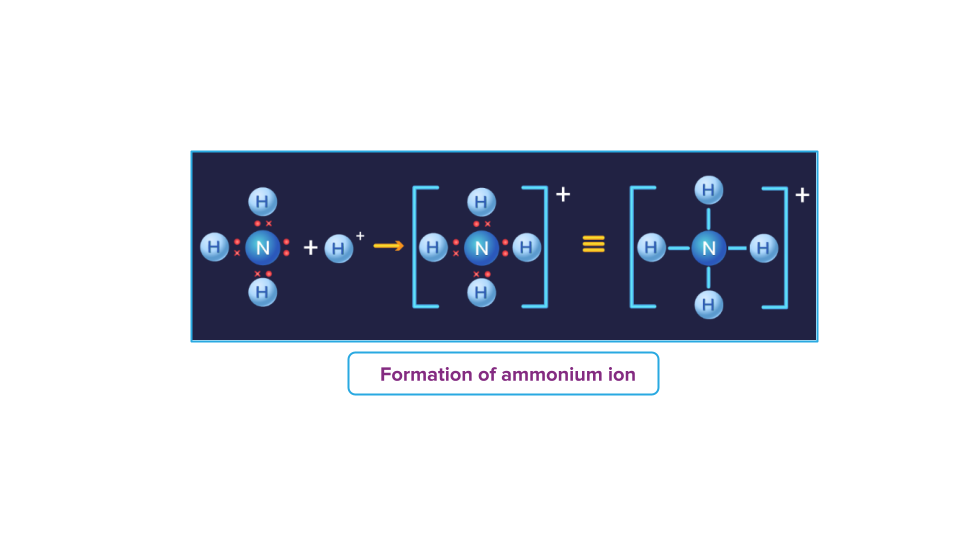
Formation of Ammonium ion:
The nitrogen atom acts as the Lewis base in ammonia, donating its lone pair of electrons to the vacant orbital of the H+ ion, which acts as the Lewis acid. As a result of the formation of a co-ordinate bond, nitrogen is the donor and H+ is the acceptor.
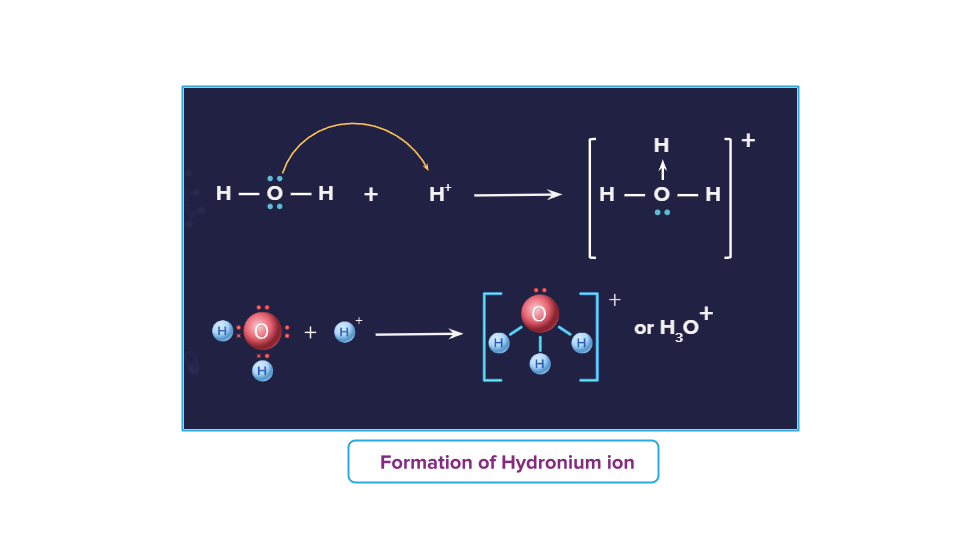
Formation of Hydronium ion:
Water is the Lewis base for the formation of hydronium ions. The lone pair of electrons from an oxygen atom in water are donated to the empty orbital of the H+ ion. H+ ion acts as Lewis acid. Between water molecules and H+ ions, a dative bond is formed. The donor atom in water is oxygen, while the acceptor atom is H+ion.
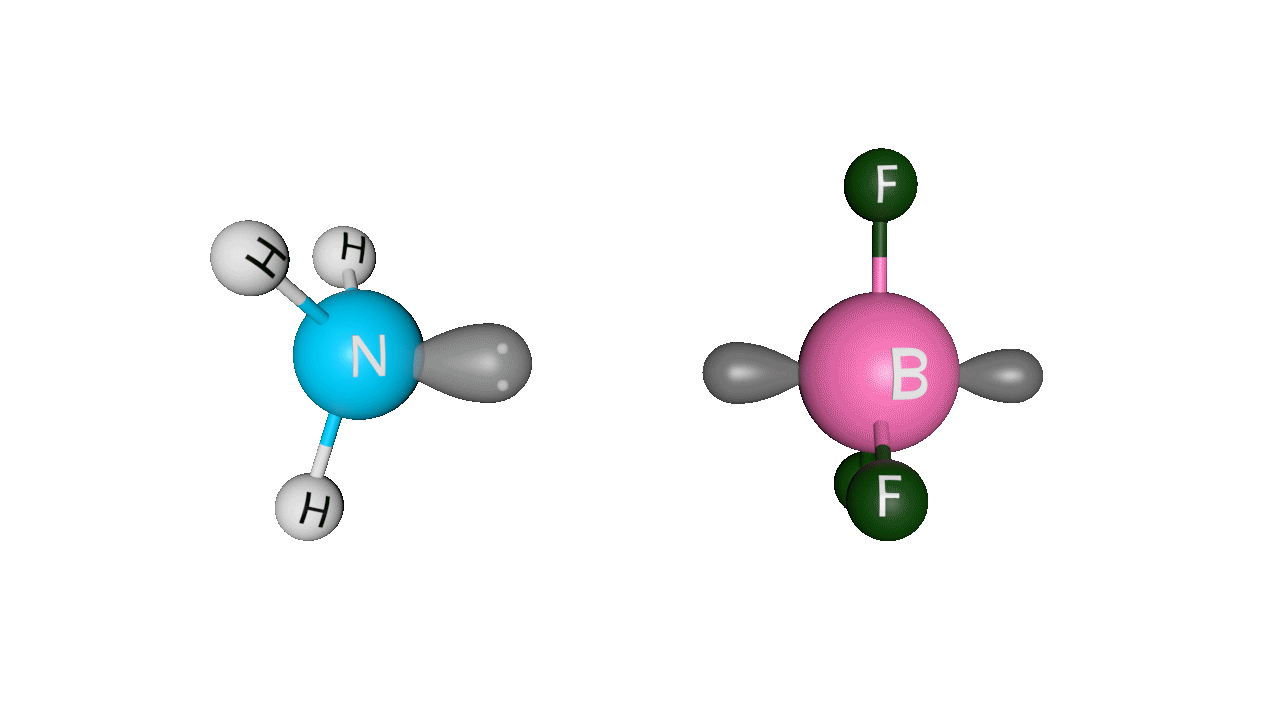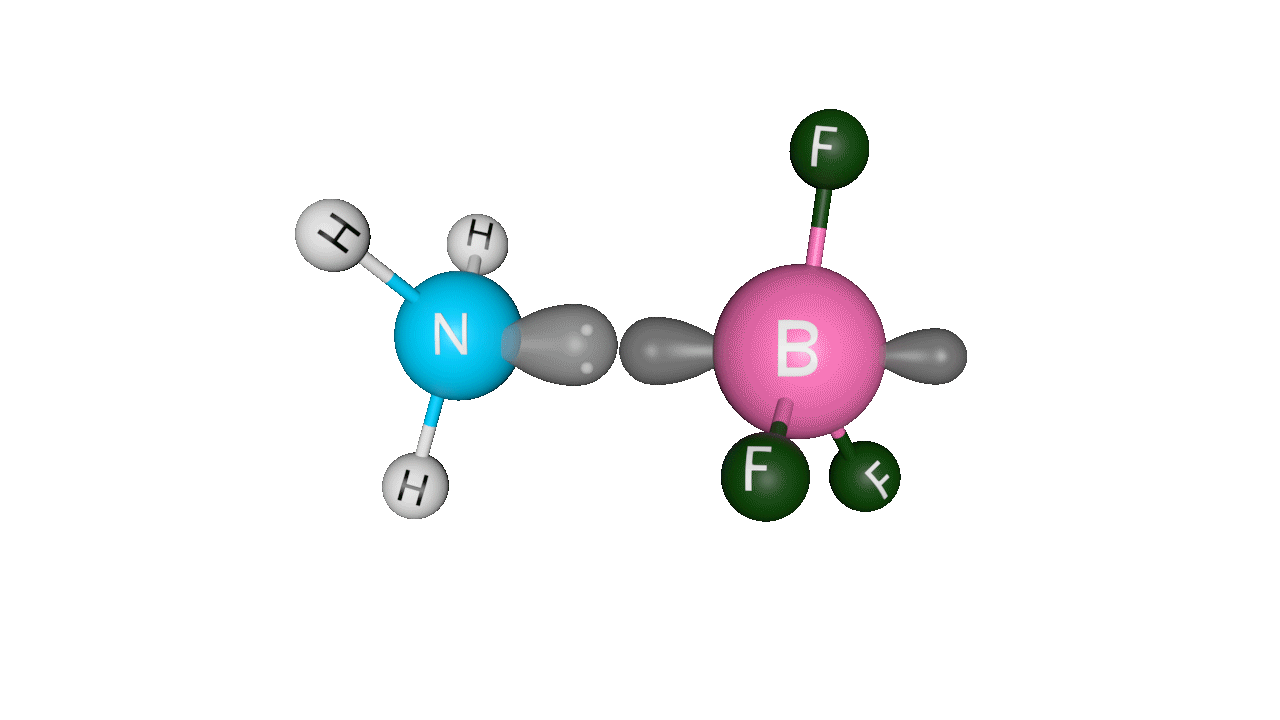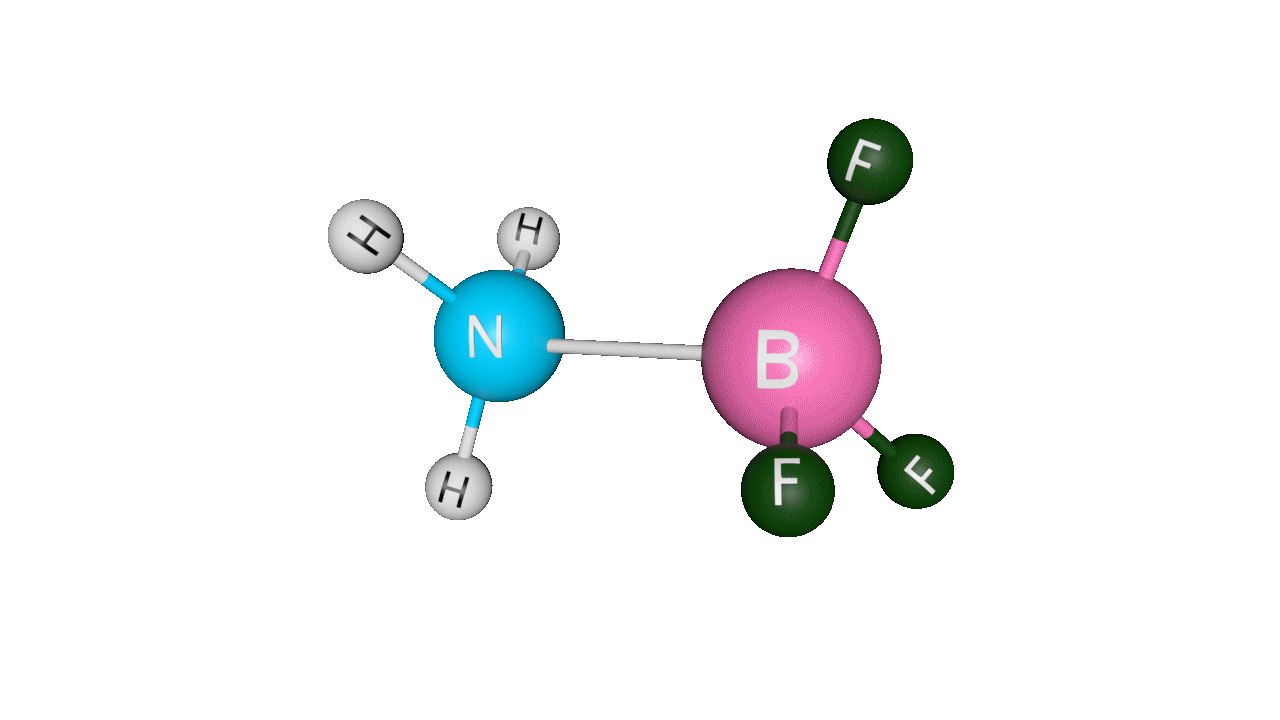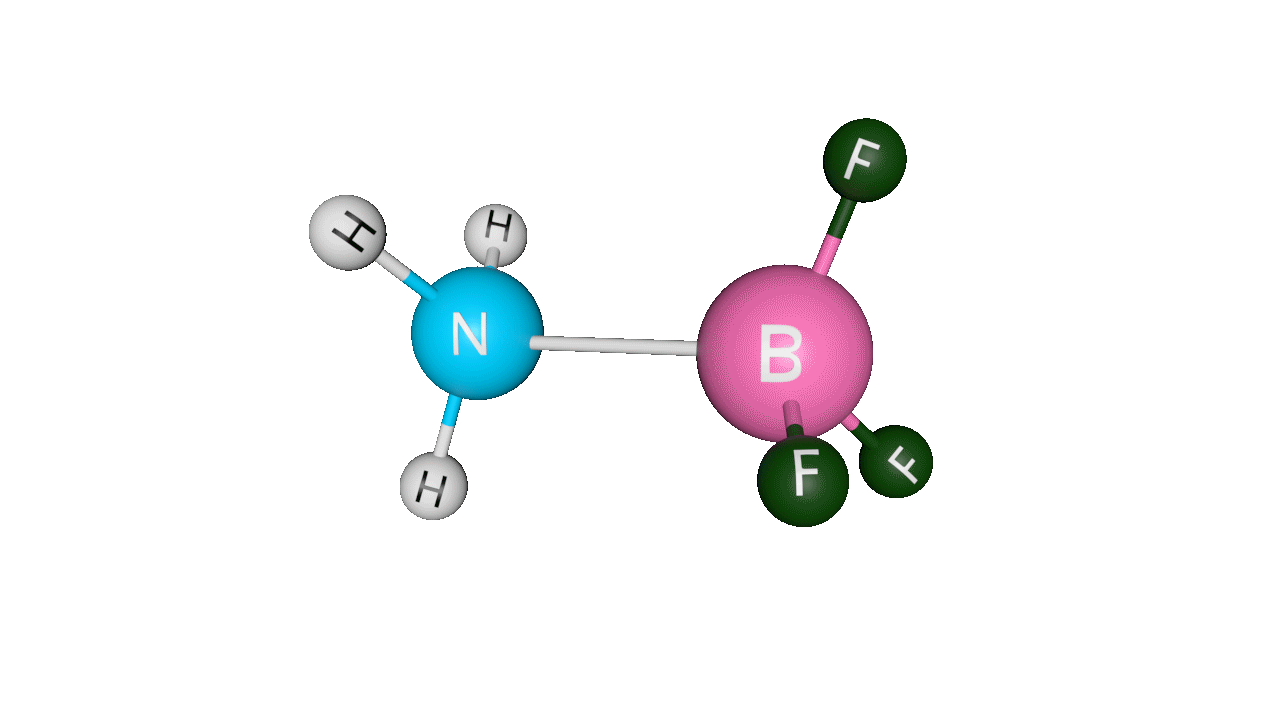
Formation of ammonia-boron trifluoride:
Boron trifluoride (BF3) is a compound in which the boron (B) atom is not accompanied by a noble gas structure. The valence shell of a boron only has three pairs of electrons, and one pair is required to complete the octet. As a result, BF3 is electron-deficient and lewis acid.
To overcome the deficiency, the lone pair on the nitrogen (N) of the ammonia (NH3) molecule is utilised, and a complex is formed via a coordinate covalent bond. The Lewis acid in this case is BF3, which is electron-deficient, whereas the Lewis base is NH3, which is electron-rich.

Formation of carbon monoxide:
The valence shell of carbon (C) has four electrons, while the valence shell of oxygen (O) has six. Two electrons, one from each atom, are shared by carbon and oxygen. The octet rule is satisfied for oxygen atoms in carbon monoxide (CO), however the carbon atom has two electrons missing. In addition to the two regular (double) covalent bonds, oxygen shares two electrons with carbon to form a coordinate covalent bond. As a result, the electron-rich oxygen atom works as the donor in carbon monoxide, while the carbon atom acts as the acceptor. And here in the carbon monoxide molecule, a coordinate covalent bond is formed within the molecule.

Coordination compounds:
A coordination complex is made up of a central atom or ion, which is usually metallic and is known as the coordination centre, and a ring of attached molecules or ions, which are known as ligands or complexing agents.
For example;
Properties of coordinate compounds:
- This type of bonding is typically formed by a metal and an electron-rich ligand.
- Coordinate compounds have substantially greater melting and boiling points than covalent compounds but much lower melting and boiling points than ionic compounds.
- These compounds are only moderately soluble in water.
- Coordinate compounds do not ionise in water completely and are weak electrical conductors.
- The compounds are directional, polar, and rigid.
- This bond is less strong than ionic bonding.
Practice problems:
Q.1. Which of the following forms a dative bond?
(A) AlCl3
(B) HCl
(C) NaCl
(D) None of the above
Answer: (A)
Solution: In AlCl3, each aluminium (Al) atom has a deficit of two electrons in its valence shell, and chlorine (Cl) has a lone pair of electrons. Al forms a coordinate covalent bond with the Cl atom on an adjacent AlCl3 group. The Al atom acts as the acceptor and the neighbouring Cl atom acts as the donor. As each of two Al atoms does this, then aluminium chloride is a covalent dimer molecule with the formula Al2Cl6.
Q.2. BF3 forms an adduct with NH3 because:
(A) nitrogen has a high electronegativity
(B) boron has a high electronegativity
(C) boron has an empty p-orbital and nitrogen has one lone pair of electrons
(D) boron has an electropositive character
Answer: (C)
Solution: In BF3, boron is electron deficient and has vacant p-orbital and in ammonia, nitrogen has a lone pair of electrons and is electron rich, Hence nitrogen will donate the lone pair of electrons and boron will accept it and forms a coordinate covalent bond.
Q.3. When a cation is hydrated, the bond formed between the cation and the water molecule is typically
(A) Dative bond
(B) Covalent bond
(C) Ionic bond
(D) Hydrogen bond
Answer: (A)
Solution: Cation is a positively charged ion with fewer electrons. It forms a dative bond with the lone pair of electrons of the oxygen atom of the water molecule because it is electron deficient.
Q.4. What are the types of bonds present in copper sulphate pentahydrate ()?
(A) Electrovalent, covalent and coordinate covalent
(B) Covalent and coordinate covalent
(C) Electrovalent and coordinate covalent
(D) Electrovalent, covalent
Answer: (A)
Solution: Sulphur and oxygen make covalent bonds since they are both nonmetals, but copper and sulphate form an ionic bond because one is metal and the other is nonmetal. Hydrated copper sulphate is a complex that is bonded to water molecules, which act as ligands in the formation of a coordinate bond.
Frequently asked questions(FAQs):
Q1. What exactly do you mean by a lone pair and a shared pair of electrons?
Answer: Non-bonded pairs of electrons on a molecule's atom that can be shared with other atoms are referred to as lone pairs of electrons, whereas bonded pairs of electrons that are shared equally between two bonded atoms are referred to as shared pairs of electrons.
Q2. Are dative bonds polar or nonpolar?
Answer: When an atom on one molecule with an empty orbital interacts with an atom on another molecule with a lone pair, a dative bond is formed. The atom with an empty orbital has a low core charge (and a low number of valence electrons), whereas the atom with a lone pair of extra electrons has a high core charge (and a high number of valence electrons). Generally, the one with the lone pair has a stronger electronegativity than the one with the empty orbital. As a result, dative bonds are polar.
Q3. Why are coordinate compounds melting and boiling points higher than covalent compounds but lower than ionic compounds?
Answer: A coordinate covalent bond is a union of one electrovalent and one covalent bond; the volatility of these substances is intermediate between covalent and ionic compounds. As a result, their melting and boiling temperatures are higher than those of covalent compounds but lower than those of ionic compounds.
Q4. What is the Difference Between a Coordinate Bond and an Ionic Bond?
Answer: The electrostatic force of attraction between positively charged cations and negatively charged anions forms ionic bonds. However, even if the electron pair is shared by only one atom in the case of coordinate bond formation, no ions are formed since none of the atoms totally lose or gain electrons.


