-
Call Now
1800-102-2727
Chemical Reactions of Alkenes - Reaction With Dry Silver Oxide (Ag2O) and Moist silver Oxide (Ag2O), Practice Problems and FAQs
As we have seen many times, a car engine can overheat and stop working in extreme temperatures. To solve this problem, antifreeze or engine coolant mixed with water is used to help regulate your engine during extreme temperatures.
The most common antifreeze is ethylene glycol, which is produced industrially from ethylene oxide (epoxide). Because epoxides are the raw material for engine coolants, we should understand how they are formed.

Table of Contents
- Reaction of an alkene with dry silver oxide
- Reaction of an alkene with moist silver oxide
- Practice Problems
- Frequently Asked Questions
Reaction of an alkene with dry silver oxide:
The majority of epoxides are formed by reacting alkenes with peroxide-containing reagents, which donate a single oxygen atom. Metal complexes are also effective epoxidation catalysts. When alkene reacts with dry Ag2O which is prepared in situ from silver and oxygen, they undergo Epoxidation.
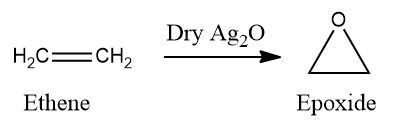
High temp is used to form nascent oxygen ([O]) which follows a free radical mechanism to form an epoxide.
Epoxyethane is a heterocyclic compound. These compounds are cyclic structures in which one (or more) of the ring atoms is a hetero atom, that is, an atom of a non-carbon element.
Reaction of an alkene with moist silver oxide:
When alkene reacts with moist Ag2O, They undergo the first epoxidation but due to the presence of water, the ring opens to form diols. Hence here the final product is diols.

The condition of reaction is basic here, so always remember that ring opens from a less hindered site through SN2 mechanism.
Practice Problems:
Q1. What will be the preferred product for the given reaction?

a. 1,2-epoxy butane
b. 2,3-epoxy butane
c. Butane-1,2-diol
d. Butane-2,3-diol
Answer: (D)
Solution: When an alkene reacts with moist silver oxide, the first epoxidation occurs, but because of the presence of water, the ring opens to form diols. As a result, the final product is diols instead of an epoxide. Hence, the option (A) and (B) can not be an appropriate answer.
The correct answer is option (D) Butane-2,3-diol.

Q2. What will be the preferred product for the given reaction?

a. 1,2-epoxy hexane
b. 1,2-epoxy cyclohexane
c. Cyclohexane-1,2-diol
d. 2,3-epoxy cyclohexane
Answer: (B)
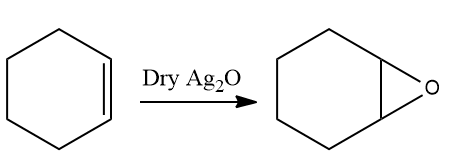
Solution: Metal complexes can also be used as epoxidation catalysts. Epoxidation occurs when alkene reacts with dry silver oxide produced in situ from silver and oxygen. Hence the diol can not be a correct answer.
The correct answer should be 1,2-epoxy cyclohexane. Hence, the correct option is (B).
Q3. When 1-methyl cyclopentene is treated with moist silver oxide and dry silver oxide respectively, What will be the preferred product for the reaction?

a. 1-methylcyclopentane-1,2-diol, 1-methyl-1,2-epoxy cyclopentane
b. 2-methylcyclopentane-1,2-diol, 1-methyl-1,2-epoxy cyclopentane
c. 2-methylcyclopentane-1,2-diol, 2-methyl-1,2-epoxy cyclopentane
d. 1-methylcyclopentane-1,2-diol, 2-methyl-1,2-epoxy cyclopentane
Answer: (A)
Solution: The first epoxidation occurs when an alkene reacts with moist silver oxide, but due to the presence of water, the ring opens to form diols. As a result, instead of an epoxide, the final product is diols.
While alkene reacts with dry silver oxide produced in situ from silver and oxygen, epoxidation occurs.
Hence the product formation can be expected as:

The correct answer is hence, option (A).
Q4. What will be the preferred product for the given reaction?
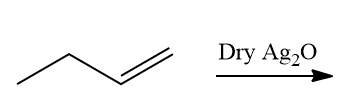
a. 1,2-epoxy butane
b. 2,2-epoxy butane
c. 1,3-epoxy butane
d. Butane-1,2-diol
Answer: (A)
Solution: Metal complexes can be used as epoxidation catalysts as well. When alkene reacts with dry silver oxide produced in situ from silver and oxygen, epoxidation occurs. As a result, the diol cannot be the correct answer.

1,2-epoxy butane is the correct answer. As a result, the correct option is (A).
Frequently Asked Questions-FAQs:
1. What are the applications of an alkene?
Answer: They are used to make polythene for buckets, bowls, and bags, as well as polystyrene for car battery cases and refrigerator parts. They are used in the production of ethane-1,2-diol, which is used in automobile radiators as an antifreeze agent.
2. Where can you find alkene?
Answer: Thermal cracking of ethane can result in the production of ethene and hydrogen molecules. Alkenes are the raw materials for many plastics, including polyethene, PVC, polypropylene, and polystyrene. Alkene chemistry can be found in unsaturated fats, beta-carotene, and light through vision.
3. What are the properties of alkenes?
Answer: Some of the physical properties of alkenes and alkanes are similar: they are colourless, nonpolar, and combustible. The physical state is determined by molecular mass: At room temperature, gasses behave similarly to the corresponding saturated hydrocarbons, the simplest alkenes, ethene, propene, and butene.
4. Why are alkanes referred to as paraffin?
Answer: The term "paraffin" comes from Latin. Because alkanes have a very low affinity for a general reagent, they are referred to as paraffin. In other words, alkanes are toxic compounds. They have reactions only under extreme conditions.


