-
Call Now
1800-102-2727
Calcination - Introduction, Purpose, Working, Examples, Difference Between Roasting and Calcination, Practice Problems & FAQs
Let’s scan the interior of the house. Have you ever considered the commonality between the sterling stud you are wearing and the utensils you are using to eat? They all contain metal or metal alloys and are essential to our daily lives. Iron is probably also used in the legs of the tables and chairs in your classroom. As a chemist, you can provide a lot more intriguing explanation than "from a shop" if someone asks where this iron came from.
The origin of iron and how it came to be used in our furniture illustrates how the chemistry of metals affects us on a daily basis. To make the legs for chairs and tables, iron ore had to be mined, crushed, and moulded.
Metals make up about 75% of all known chemical elements. Aluminium, iron, calcium, sodium, potassium, and magnesium are the ones that are most plentiful. The bulk of metals is found in ores, although a small number including copper, gold, platinum, and silver occur naturally in the native state since they do not easily react with other elements. Few metals occur naturally on the earth’s crust in the form of compounds.
So, for the extraction of metals from their ores, several processes take place and out of which is calcination which we will discuss in this article.
Table of Content
- Introduction to Calcination
- Working of Calcination Process
- Difference between Roasting and Calcination
- Practice Problems
- Frequently Asked Questions- FAQs
Introduction to Calcination
In the simplest terms, calcination is the procedure of heating a substance in a controlled atmosphere. Usually, the process also controls the temperature. The purpose of calcination is to alter a substance's physical or chemical properties.
Substances are heated to extremely high temperatures during calcination. Generally, this is done to get rid of water, and volatile compounds and to oxidise the substance. This procedure is occasionally referred to as a purifying procedure. The Latin term “calcinare”, which means "to burn lime," is also the root of the English word "calcination."
So, calcination is defined as the process of heating concentrated ore at a temperature slightly below its melting point in a limited supply of air or in the absence of air.
Examples of calcination:
Calcination of aluminium hydroxide: The aluminium hydroxide precipitate obtained in the preceding step is calcined in a rotating kiln at 1500°C to obtain pure aluminium oxide, Al2O3 (commercially referred to as alumina).
- It transforms metal carbonates into their respective oxides.



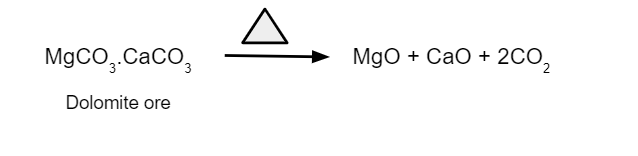
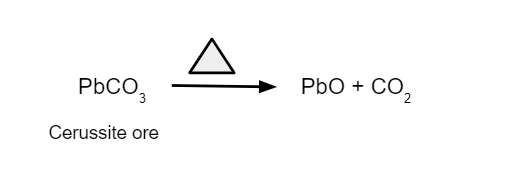
2. Removes moisture, which is water in the form of crystallisation, from the hydrated oxide.
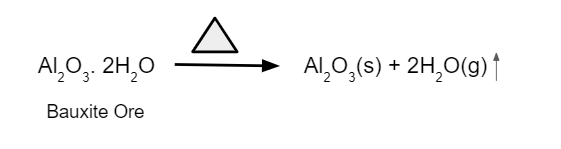



3. By calcining, further volatile and organic contaminants can be eliminated.
Working of Calcination Process
To ensure a uniform product, calcination is frequently done in furnaces, retorts, or kilns. Materials are frequently turned over or stirred during this process. This often takes place in a reverberatory furnace. The ore becomes porous as a result, making subsequent processing stages much easier.
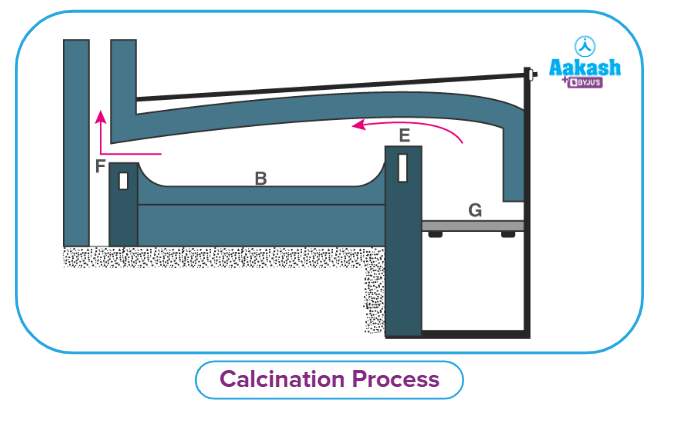
Reverberatory furnaces can be built in a variety of ways, but they all include direct contact between the material to be calcined with the flames and hot gases from the fire. The fuel is kept separate from the material, though.
The fire is burning on the grate at G in the image above. Now, the flames that are crossing the bridge at E are diverted downward by the furnace's low roof, passing directly over the charge or material that is being calcined and is placed on platform B. The heated gases and fumes subsequently vent into the chimney through the throat F. A thin layer of the charge is dispersed uniformly across the bed.
Difference between Roasting and Calcination
Below is a list of some of the key distinctions between roasting and calcination.
|
Calcination |
Roasting |
|
In the process of calcination, the air may be given in limited amounts or ore may be heated below its melting point without the presence of air. |
Roasting is a process of heating ore below its melting point while it is exposed to air or oxygen. |
|
Carbonate ores are thermally decomposed during calcination. |
For sulphide minerals, roasting is primarily used. |
|
During calcination, moisture from the ore is expelled. |
Dehydration of ore does not take place during roasting. |
|
During calcination, carbon dioxide is released. |
A significant amount of poisonous, acidic, and metallic chemicals are emitted during roasting. |
Recommended Link: https://www.youtube.com/watch?v=i54zcRBaFJo
Practice Problems
Q. Which of the following is true for the calcination of metal ore?
- Carried out in presence of air.
- The ore is heated to a temperature when fusion just begins.
- Hydrated salts lose their water of crystallization.
- Impurities of S, As and Sb are removed in the form of their volatile oxides.
Answer: (C)
Solution: Calcination is basically the process of heating ore to a high temperature that is inadequate to melt the ore without the presence of air. Generally, carbonates and hydrated ores are calcined and remove the water of crystallisation present in the hydrated oxide in the form of moisture to form metal oxides. Impurities of S, As and Sb are removed in the form of their volatile oxides during roasting not during calcination.
Q. Which gas releases during the calcination process?
- Oxygen
- Carbon dioxide
- Hydrogen
- Nitrogen
Answer: (B)
Solution: The calcination process is used to convert metal carbonates into metal oxides and carbon dioxides.

Where ‘M’ is a metal. So, option (B) is the correct answer.
Q. Which of the following processes makes the ore porous?
- Distillation
- Roasting
- Calcination
- None of the above
Answer: (C)
Solution: Calcination is basically the process of thermally decomposing a substance by heating it to a high temperature that is below its melting point in the absence of air. The ore is made porous by this treatment. So, option (C) is the correct answer.
Q. Which of the following reactions is an example of the calcination process?
Answer: (D)
Solution: In order to remove CO2 from the carbonate ore and water or moisture from hydrated oxides, the concentrated ore is heated in the absence of air at a temperature below their melting points. Calcination is the name of this procedure, which is often done in a reverberatory furnace. So, option (D) is the correct answer.
Frequently Asked Questions - FAQs
Q. Why does calcination occur without the presence of oxygen?
Answer: The carbonate ores undergo calcination. The calcination process involves intensely heating the ores without oxygen (air). This transforms the metal carbonates into metal oxides and carbon dioxide. These ores won't react when heated in the presence of oxygen or air, making the entire procedure pointless.
Q. What distinguishes calcination from smelting?
Answer:
|
Calcination |
Smelting |
|
Calcination is basically the process where the substance is heated to a high temperature below its melting point in absence of oxygen, bringing in thermal decomposition of ores. |
Smelting is defined as the process of melting substances to extract metal from their ore. |
|
Calcination is majorly used in the process to obtain gypsum and limestone. |
The smelting process is used to extract metals like silver, copper, iron, etc |
Q. In which furnace calcination and roasting processes carried out?
Answer: Calcination and roasting are done in a reverberatory furnace depending upon the conditions required in each case.
Q. What is the calcination temperature of limestone?
Answer: In limestone calcination, a thermal decomposition process usually occurs at the temperature range between 900 to 1050 °C.



