-
Call Now
1800-102-2727
Bromine Water Test- Bromine Water, Bromine Water Test, Identification of Unsaturation in Organic Compounds, Practice Problems and Frequently Asked Questions
Assume you are completing your graduation practical. The chemistry teacher says as she offers you two test tubes with the letters A and B. "See, my buddy, there are two hydrocarbons present in each test tube. How can you find out which compound is an unsaturated out of these two?"
If you are familiar with the bromine water test, you can identify test tubes containing saturated and unsaturated hydrocarbons.
If you are not, let's learn about it

Table of Contents
- Bromine Water
- Bromine Water Test
- Identification of Unsaturated organic compounds
- Practice Problems
- Frequently Asked Questions
Bromine water
An aqueous solution containing bromine molecule is the bromine water.
Bromine water solution can be prepared by two ways-
- Shake a few (2 to 6) drops of bromine liquid with 50mL of water in a brown bottle. Addition of very small amounts of sodium bromide and sodium bromate will help in the test for unsaturation.
- Mix sodium bromide and sodium bromate salts in the ratio of about 10:1 and add dil.sulphuric acid slowly.
Bromine water has yellow to red colour depending on the bromine concentration.
Inhaling the vapours may cause lung problems. May cause eye irritation and severe skin burns when it comes in contact with. Having a 5% sodium thiosulphate for washing is advisable first aid for external bromine contact.
Bromine Water Test
Bromine water is an oxidizing agent and reacts with reducing compounds like glucose-containing aldehydes, and as brominating agents with aromatic compounds like phenols and amines. It is used primarily to test the unsaturation present in organic compounds.
Identification of unsaturation in organic compounds
It is commonly used as a reagent in chemical recognition tests for substances that, in an aqueous environment, react with bromine via the halogenation mechanism, primarily for unsaturated carbon compounds (with a double or triple bond(s)).
It can be used for both alkenes and alkynes.
This test can be used for detecting the presence of unsaturated hydrocarbons.
This test can be used to distinguish between unsaturated hydrocarbons and saturated hydrocarbons.
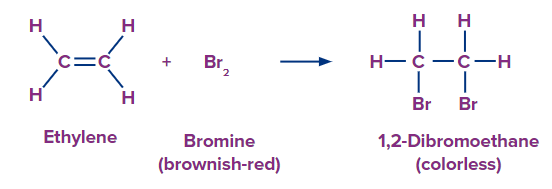
Bromine addition to alkene and alkyne is anti-addition.

![]()
Bromine solutions are brown. In this test, when a bromine solution is added to an unsaturated hydrocarbon, the brown colour disappears if the hydrocarbon is unsaturated. Bromine forms an addition product when it reacts with the unsaturated hydrocarbon.
Practice Problems
- The bromine water test is used to distinguish between
- Pentyne and Pentanol
- Pentanol and Pentane
- Pentene and Pentane
- Pentene and Pentanol
Solution: The bromine water test is used to distinguish between unsaturated and saturated hydrocarbons. Both pentene and pentyne are unsaturated hydrocarbons and Pentane is a saturated hydrocarbon. Pentanol does not fall into either category. Hence, the correct option should be (C) in which Pentene being an unsaturated hydrocarbon can be distinguished from Pentane being a saturated hydrocarbon.
- The bromine water test distinguishes between
- Hexene and Butene
- Butane and Heptyne
- Pentene and cyclopentene
- Ethylene and Acetylene
Solution: The bromine water test is used to distinguish between unsaturated and saturated hydrocarbons.
In option (A), Hexene and Butene both are unsaturated hydrocarbons and give the same result with the bromine water test. Hence can not be distinguished.
In option (B), Butane is a saturated hydrocarbon and Heptyne is an unsaturated hydrocarbon. Hence, they can be distinguished by using this test.
In option (C), Pentene and Cyclopentene both are unsaturated hydrocarbons and give the same result with the bromine water test. Hence can not be distinguished.
In option (D), Ethylene and Acetylene both are again unsaturated hydrocarbons and give the same result with the bromine water test. Hence can not be distinguished.
Hence option (B) will be the correct answer.
- Which of the following compound give a colourless solution on reaction with reddish-brown coloured bromine water?
- Propane
- Propanone
- Propanoic acid
- Propene
Solution: If an unsaturated hydrocarbon is treated with a bromine solution, the brown colour disappears. Propene is an unsaturated hydrocarbon among all the given options. Propanol, Propanone and Propanoic acid do not fall into the category of unsaturated hydrocarbons. As a result, the correct option is (D).
- The bromine water test allows for the distinction of
- C2H4 and C2H2
- C3H4 and C2H4
- C3H6 and C2H4
- C3H8 and C2H4
Solution:
Saturated hydrocarbon has a general formula CnH2n+2. Hydrocarbons not having the same C:H ratio will be unsaturated.
The bromine water test is used to distinguish between unsaturated and saturated hydrocarbons.
In option (A), C2H4 (CH2=CH2) and C2H2(CHCH) both are unsaturated hydrocarbons and give the same result with the bromine water test. Hence can not be distinguished.
In option (B),C3H4 (CHC-CH3) and C2H4(CH2=CH2)both are again unsaturated hydrocarbons and give the same result with the bromine water test. Hence can not be distinguished.
In option (C), C3H6 (CH3-CH=CH2) and C2H4(CH2=CH2)both are unsaturated hydrocarbons and give the same result with the bromine water test. Hence can not be distinguished.
In option (D), C3H8 (CH3-CH2-CH3) is saturated hydrocarbon and C2H4(CH2=CH2) is again unsaturated hydrocarbons and give result with the bromine water test. Hence can be distinguished.
Hence option (D) will be the correct answer.
Frequently Asked Questions
1. Is alkyne used as fuel?
Answer: The most fundamental member of the alkyne series is acetylene. Because it generates a lot of heat when burned, it's commonly used as a fuel for welding torches.
2. What are the uses of alkynes in everyday life?
Answer: Alkynes and compounds containing alkynes have a wide range of applications in a variety of industries. Alkynes such as propyne and acetylene, for example, are used as starting materials in the manufacture of plastic products in the fuel and plastics industries.
3. What does a bromine water test for lipids or vegetable oils achieve?
Answer: When shaken with unsaturated vegetable oil, bromine water turns colourless, but when shaken with saturated vegetable fat, it remains orange-brown. Bromine water can also be used to test the saturation level of vegetable oil.
4. What purposes do unsaturated hydrocarbons serve?
Answer: In various industrial processes for the creation of insecticides, gasoline, paints, lacquers, and other products, unsaturated hydrocarbons are frequently employed. They are additionally utilised in numerous chemical processes to create new molecules. Alkenes, alkynes, and aromatic hydrocarbons are the three different forms of unsaturated hydrocarbons.


