-
Call Now
1800-102-2727
Bonding in Coordination compounds- Werner's theory, Valence Bond Theory, Crystal field theory, Back Bonding in Metal Carbonyl, Practice Problems and FAQs
Coordination chemistry is an essential and demanding topic of current inorganic chemistry. Chemical bonding and molecular structure theories have provided new insights into how these substances work as crucial components of biological systems. Chlorophyll, haemoglobin, and vitamin B12 are magnesium, iron, and cobalt coordination molecules, respectively. Coordination compounds are used in a variety of metallurgical processes, industrial catalysts, and analytical reagents. Electroplating, textile dyeing, and pharmaceutical chemistry are just a few of the fields where coordination chemicals are used. So, let us discuss how we can explain the bonding in coordination compounds.
Table of Contents
- Werner's theory of Coordination compounds
- Valence Bond Theory
- Crystal field theory
- Back Bonding in Metal Carbonyl
- Practice Problems
- Frequently asked Questions
Werner's theory of Coordination compounds
Werner developed a theory in 1893 to explain the structures, formation, and nature of bonding in coordination compounds. First of all, let's discuss the experiment performed by Werner.
Postulates of Werner’s Theory
Werner proposed his theory of coordination compounds and the following are the main postulates:
- Metals in coordination compounds have two types of linkages (valences): primary and secondary.
- Negative ions satisfy the primary valences, which are normally ionisable. For metals, it is equal to the oxidation number of metals.
- The secondary valences cannot be ionized. These are generally neutral molecules or negatively charged ions. For a metal, the secondary valence is equal to the coordination number and is fixed.
- The ions/groups bound to the metal by secondary linkages have distinct spatial arrangements that correspond to different coordination numbers.
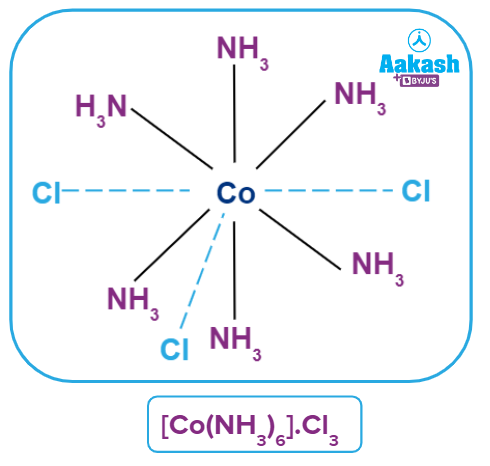
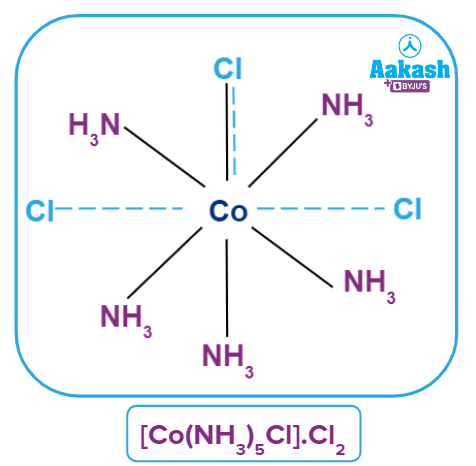
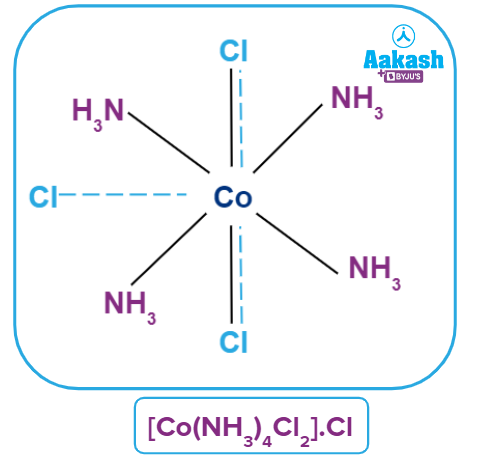
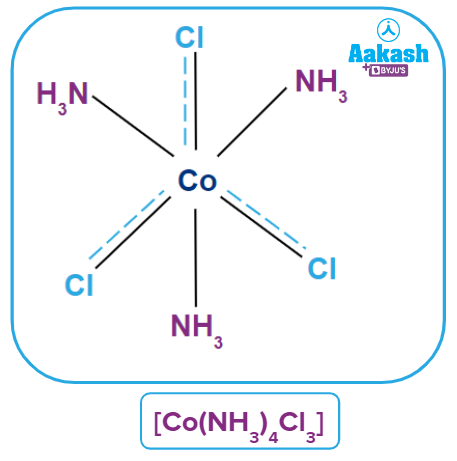
Coordination Polyhedra
Such spatial arrangements are known as coordination polyhedra in modern formulations. Coordination entities or complexes are the species within the square bracket, and counter ions are the ions outside the square bracket. In the spatial arrangement, primary valency is shown by a dotted line and secondary valency is shown by a solid line.
Valence Bond Theory
Pauling’s valence bond theory, (VBT), was applied to explain the bonding in coordination compounds also.
The Important points of VBT are:
- The coordination complex is formed by the interaction between a Lewis base (considered as ligand) and a Lewis acid (either a metal or thea cation) to form a coordinate bond..
- Necessary s, p and d atomic orbitals of the atom(s) or ion(s) of the metal undergoes hybridization to produce, same number of equivalent orbitals but in specific directions such that the compound exhibit a specific geometry.
- The hybridised orbitals of the metal or metal ions overlap with the ligand orbitals to form coordinate bonds.
- Generally the orbitals containing free lone pair of electrons of the ligand overlap with the hybridised orbital of the metal or metal ion and form coordinate bonds..
- The orbitals of metal or metal ions utilised for hybridization is identified from the experimentally measured magnetic moment of the compound.
The hybridisation of coordination compounds
|
C.N. of metal |
4 |
4 |
5 |
6 |
6 |
|
Type of hybridisation |
sp3 |
dsp2 |
sp3d |
sp3d2 |
d2sp3 |
|
Shape of complex |
Tetrahedral |
Square Planar |
Trigonal Bipyramidal |
Octahedral |
Octahedral |
|
Type of d-orbital |
- |
Note: Inner orbital complex: In complex formation, the inner d-orbitals are used in the hybridisation.
Outer orbital complex: In complex formation, the outer d-orbitals are used in hybridisation.
Example
The cobalt ion is in the +3 oxidation state and has the electronic configuration 3d6 in the diamagnetic octahedral complex
Information:
n=4
The diagram depicts the hybridization scheme.
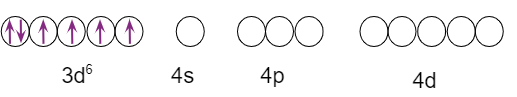
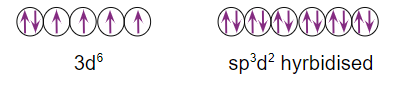
In hybridisation, the paramagnetic octahedral complex employs outer orbital (4d) sp3d2 ). As a result, it is known as the outer orbital, high spin, or spin-free complex.
Crystal field theory
The interaction of metal ions with ligands is treated as a purely electrostatic event in crystal field theory, which treats the ligands as point charges in the region of the central atom's atomic orbitals. There are some basic assumptions which are listed below as
Assumption-1: The crystal field theory (CFT) is an electrostatic model.
The metal-ligand bond must be ionic, and the interactions must be entirely electrostatic, according to the electrostatic model.
Assumption-2: The transition metal is said to be a positive point charge that serves as the complex's core atom.
Assumption-3: The ligands are arranged around the core metal ion in such a way that there are fewer repulsions between these sites.
Assumption-4: It addresses the impact of ligands on the relative energies of the core metal atom/d-orbitals.Five d-orbitals (energy-identical orbitals) are degenerate in an isolated gaseous metal ion.
Assumption-5: If a spherically symmetrical field of ligands surrounds the metal atom/ion. This degeneracy is still maintained.
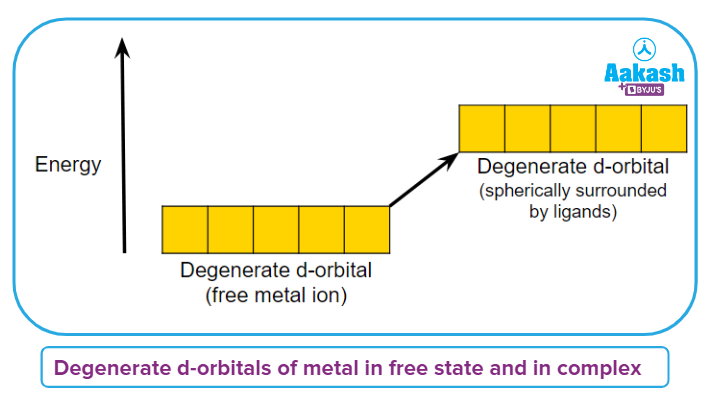
Crystal Field Splitting
Crystal field splitting is the conversion of five degenerate d-orbitals of a metal ion into multiple sets of orbitals with varying energies in the presence of an electrical field of ligands.
Crystal field splitting in octahedral complex
Ligands are approaching from the axes,as a result, the dz2, dx2-y2 orbitals that point toward the axes along the ligand's direction will suffer more repulsion and gain energy than the dxy, dyzand dxz orbitals that point between the axes.
Due to ligand electron-metal electron repulsions in the octahedral complex, the degeneracy of the d orbitals has been lifted, yielding three lower-energy t2g orbitals and two higher-energy eg orbitals.
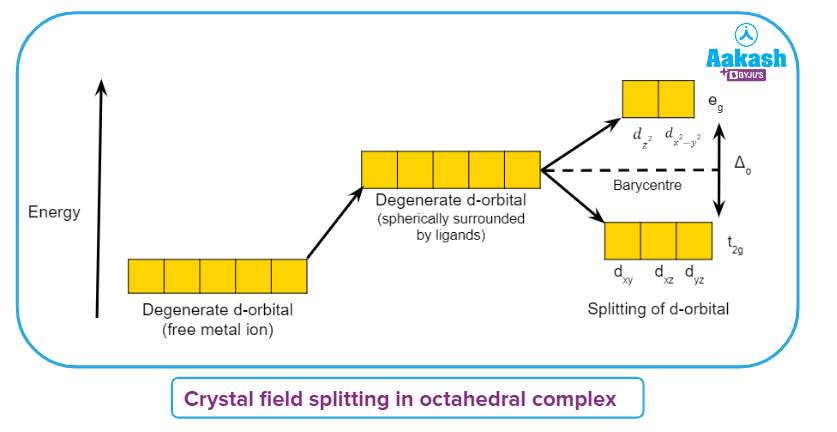
The energy difference between the two sets of energy levels is known as crystal field splitting energy and is written as o (the octahedral subscript 'o'). The average energy of the d-orbitals does not change as a result of the crystal field splitting. This is referred to as the barycentre rule.
Crystal field splitting in tetrahedral complex
As ligands are approaching away from the axes, the dz2, dx2-y2 orbitals that point towards the axes will suffer lesser repulsion from ligands than the dxy, dyzand dxz orbitals that point between the axes in case of the tetrahedral complex.
Due to ligand electron-metal electron repulsions in the tetrahedral complex, the degeneracy of the d orbitals has been reduced, yielding three higher-energy t2 orbitals and two lower-energy eorbitals.
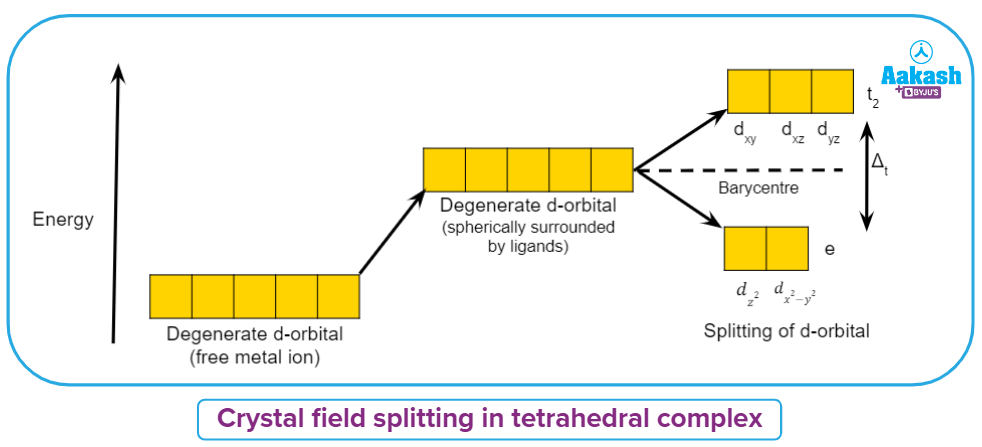
The d orbital splitting is smaller in tetrahedral coordination entity generation than in octahedral field splitting due to less number of ligands.
Metal Carbonyl
Metal carbonyls are a set of different coordination compounds in the sense that here metals are in zero oxidation state and the metal and carbon monoxide ligands are held together by either metal to carbon monoxide or carbon monoxide to metal donation of electron pair.
Back bonding in Metal Carbonyl
The two types of bonding found in metal carbonyls are described here:
- Type-1: A metal-carbon σ bond is created as a result of the carbonyl molecules donating electrons from non-bonding molecular orbital (NBMO) to metal's empty orbitals.
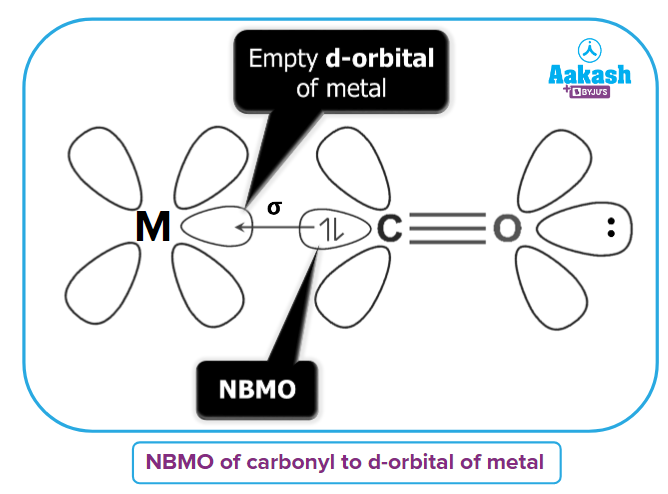
- Type-2: A metal-carbon bond is created when a pair of electrons from a filled d orbital of metal are donated to the unoccupied antibonding π* orbital of the carbonyl ligand.
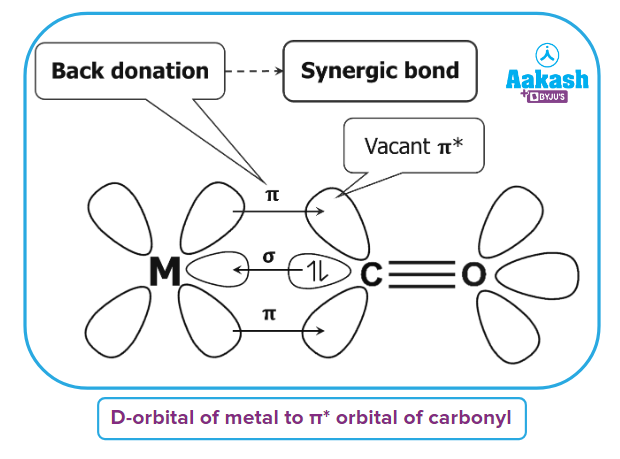
Synergic Bond
A synergic bond is a π-bond established between a metal atom/ion and a ligand by reverse donation of a metal atom/ion.
Synergic Effect
Synergic bonding causes the internal bond length of a ligand and the metal-ligand bond length to vary, which is known as the synergic effect.
The synergic effect is caused by a self-strengthening link that involves electron transfers from
Ligand to metal
Metal to ligand
Synergic Bonding in the case of Alkenes
The metal's filled d-orbitals overlap with the ethylene molecule's vacant π* molecular orbital to generate a back bond/synergic bond.
Related Video Link: Coordination Compounds Class 12 Chemistry (Ch-9) (L-1) | JEE Main Important Topics | JEE 2022 Exam
Practice Problems
Q1. Calculate the number of unpaired electrons and CFSE in the complex?
- 4, -4Dq
- 5, -8Dq
- 4, -4 Dq
- 0, -24 Dq
Solution: The configuration of . The ligand CN- is a strong field ligand, Hence pairing will occur in this case. So, this complex is a low spin octahedral complex. There are no unpaired electrons present in this complex.
CFSE= 6(-4Dq) = -24 Dq
So, the correct answer is (D).
Q2. Why is Co4(CO)12 known as an organometallic compound?
- There are carbon and metal present in the coordination compounds
- Whenever there is a carbonyl present, that compound form organometallic compound
- There is a direct bond of Cobalt metal and carbon present in the compound
- All statements are true.
Solution: Organometallic compound has at least one bond between a metallic element and the carbon atom of an organic molecule.
In option (A), only the presence of carbon and metal is not enough to become an organometallic compound. So it is incorrect.
In option (B), Carbonyl alone is not a sign of an organometallic compound, metal should be attached to the carbon of carbonyl. So this option is also incorrect.
In option (C), the statement is correct as a direct bond of metal and carbon is a basic condition of an organometallic compound. So this option is correct.
The correct answer is (C).
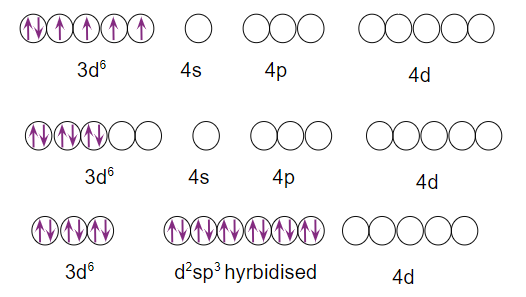
Q3. has a spin-only magnetic moment of 0 BM. Can you guess the geometry of the complex ion?
- Tetrahedral
- Trigonal Bipyramidal
- Inner orbital Octahedral
- Outer orbital Octahedral
Answer: Because the coordination number of the Fe2+ ion in the complex is 6, the hybridisation will be either inner orbital Octahedral (d2sp3 hybridisation) or outer orbital Octahedral (sp3d2 hybridisation). However, because the complex ion has a magnetic moment of 0 BM, it should be inner orbital Octahedral in shape rather than outer orbital Octahedral due to the absence of an unpaired electron in inner d orbitals
The correct answer is (C).
Q4. 0.04 moles of .Cl2 and 0.08 moles of .SO4are present in 500 mL of a solution X. The number of moles of the precipitate Y and Z that are formed when the solution X is treated with excess silver nitrate and excess barium chloride is respectively:
- 0.04, 0.08
- 0.08, 0.04
- 0.08, 0.08
- 0.04, 0.04
Solution: When 0.04 moles of [Co(NH3)5Br].Cl2 dissociates, it will give 0.08 mole of chloride ions.
0.04 moles 0.08 moles
0.08 moles of silver chloride precipitate will result from reaction with silver nitrate.
0.08 moles 0.08 moles
When 0.08 moles of [Co(NH3)5Cl].SO4 dissociates, it will give 0.08 mole of sulphate ions.
0.08 moles 0.08 moles
0.08 moles of barium sulphate precipitate will result from reaction with barium chloride.
0.08 moles 0.08 moles
So, the correct answer is an option (C).
Frequently Asked Questions
1. Why is VBT more successful than Werner's theory?
Answer: Werner's theory successfully describes the structures of many coordination compounds. It does not, however, explain the magnetic and spectral properties. Valence bond theory, primarily Linus Pauling's work, defined bonding as the overlap of atomic or hybrid orbitals of individual atoms.
2. Why is CFT better than VBT?
Answer: Valence Bond Theory detailed how orbitals combine during bond formation (VBT). The explanation was mostly based on principles of hybridization. While Crystal Field Theory describes how orbitals divide when ligands approach to metal, VBT failed to explain magnetic behaviour sufficiently. It couldn't explain how outer and inner orbital complexes formed. CFT, on the other hand, was quite clear.
3. Are the CFT and LFT the same?
Answer: The main advantage of CFT is its simplicity. The more advanced Ligand Field Theory (LFT) is based on molecular orbital theory. It's more difficult, but it's also more accurate. LFT will not only get you to the right solution more frequently but it will also be built on true concepts.
4. Who is the father of coordination chemistry?
Answer: Alfred Werner is the father of coordination chemistry because he provides the basis of modern coordination chemistry with experimental evidence.


