-
Call Now
1800-102-2727
Preparation of Aspirin – Introduction, Structure, Preparation, Uses, Practice Problems and FAQ
Have you ever read the text on the tablet covers that are written on the back?
The nutritional data, nutritional profile, additional components, consumption, storage, etc. are typically included. A component known as "Aspirin" is found in the majority of painkilling medications that are sold on the medical market.

Aspirin is an analgesic (a substance that reduces pain) that has anti-inflammatory properties. Fever can also be treated with it. On this concept page, we will get to know the structure, preparation and uses of aspirin.
TABLE OF CONTENTS
- Aspirin – Introduction
- Aspirin – Structure
- Aspirin – Preparation
- Aspirin – Uses
- Practice Problems
- Frequently Asked Questions – FAQ
Aspirin – Introduction
- Aspirin is a salicylate that is used to treat headaches, discomfort, fever, and inflammation, and to lower the risk of serious adverse cardiovascular events.
- It appears as a crystalline, colourless white powder. Generally, it has no odour, but when exposed to damp air, it starts to smell like acetic acid. It has a 482° F flashpoint. The most common uses of it in medicine are to alleviate fever, inflammation, and pain.
- Aspirin, which is listed on the WHO's List of Essential Medicines, is one of the safest and most widely used medications in the world.
Aspirin – Structure
Acetylsalicylic acid has the chemical formula C9H8O4. Similar to that, the extended formula is given as CH3COOC6H4COOH. Its IUPAC name is 2-acetoxybenzoic acid.

Aspirin – Preparation
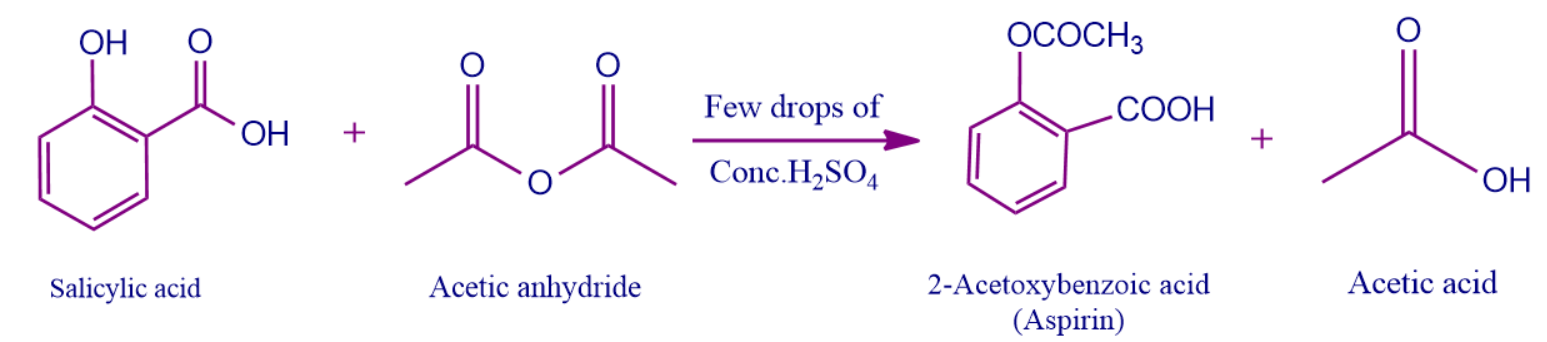
Salicylic acid is easily converted into acetylsalicylic acid by esterification with acetic anhydride. As a result, the hydroxyl group in salicylic acid is replaced by the ester group. Sulphuric acid can be used to catalyse the process.
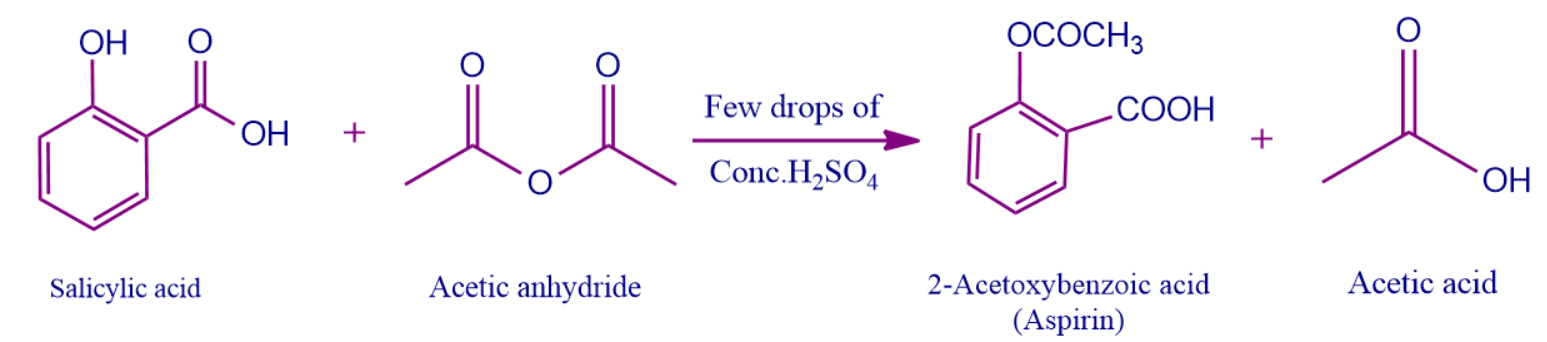
Due to aspirin's ability to hydrolyse in damp circumstances and produce salicylic and acetic acid formulations, high amounts of aspirin frequently smell like vinegar.
The salicylate ion is the therapeutic component of aspirin. Until aspirin enters the alkaline environment of the intestines, it does not ionise and produce the salicylate ion. Once it has been hydrolysed, the acetate and salicylate ions are produced. The latter ion is absorbed after passing through the intestinal wall.
Aspirin – Uses
- Aspirin is used to treat a variety of ailments, such as fever, pain, rheumatic fever, and inflammatory disorders like Kawasaki illness, pericarditis, and rheumatoid arthritis.
- Small concentrations of aspirin have also been found to lower the chance of dying from a heart attack or a stroke in persons who are at high risk or who have cardiovascular disease, but not in older people who are otherwise healthy.
- Although it is typically regarded as less effective than ibuprofen because it is more likely to result in gastrointestinal bleeding, aspirin is an excellent analgesic for acute pain.
- It helps in reducing high fever, relieving mild to high pain, and preventing the clotting of blood.
Practice Problems
1. What smell does aspirin have?
a. Vinegar
b. Sweet smell
c. Foggy smell
d. Gasoline
Answer: A
Solution: Generally, it has no odour, but when exposed to damp air, it starts to smell like acetic acid. Due to aspirin's ability to hydrolyse in damp circumstances and produce salicylic and acetic acid formulations, high amounts of aspirin frequently smell like vinegar.
So, option A is the correct answer.
2. 2-Acetoxybenzoic acid is the IUPAC nomenclature for ___________.
a. Vinegar
b. Paracetamol
c. Aspirin
d. Loperamide
Answer: C
Solution: Acetylsalicylic acid, usually referred to as aspirin, is a salicylate drug that is frequently used to alleviate pain, fever, and inflammation. Its IUPAC name is 2-Acetoxybenzoic acid.
So, option C is the correct answer.
3. Which among the following is the correct statement regarding the use of aspirin?
a. It helps in reducing high fever
b. It helps in relieving mild to high pain
c. It prevents the clotting of blood
d. All of these
Answer: D
Solution: Aspirin is used to treat mild to moderate pain from headaches, menstrual cramps, arthritis, toothaches, and muscular pains as well as to lower fever. It also prevents the clotting of blood.
So, option D is the correct answer.
4. Synthesis of aspirin is an important example of ____________ reaction?
a. Hydrogenation
b. Esterification
c. Aromatisation
d. Acid base reaction.
Answer: B
Solution: In organic chemistry, the process that creates aspirin is known as an esterification reaction. Salicylic acid's -OH group, an alcohol in nature, interacts with acetic anhydride in a substitution process to create aspirin, an ester.
So, option B is the correct answer.
Frequently Asked Questions – FAQ
1. Why is sulphuric acid used in the production of aspirin?
Answer: Without a catalyst, the esterification reaction to produce aspirin would happen very slowly. In this instance, adding a few drops of sulphuric acid to the mixture causes some free, positively charged hydrogen ions to be present in the mixture, which may then attach to the ethanoic anhydride. It becomes more active as a result, quickening the response.
2. What safety measures are required during aspirin synthesis?
Answer: Avoid the contact of acetic anhydride with your skin, clothes, and eyes. In the event of contact, thoroughly rinse that part. Clean up any spillage. The vapours of acetic anhydride irritate the eyes causing tears to flow, as acetic anhydride is a lachrymator.
3. How can the yield of aspirin production be increased?
Answer: Aspirin crystals should start to form after a short period of air cooling in the flask. To speed up crystallisation and maximise the amount of product, place the flask in an ice bath. Scratching the interior of the flask with a glass rod could be beneficial if crystals are taking a while to form.
4. What are the things that should be avoided while taking aspirin?
Answer: Medications such as warfarin, rivaroxaban, dabigatran, edoxaban, apixaban, and clopidogrel prevent blood clots. Combining the aforementioned drugs with aspirin may result in bleeding issues.


