-
Call Now
1800-102-2727
Amines Chemical Properties: Basic Nature of Amines, Carbylamine Reaction, Schotten Baumann Reaction, Practice Problems and FAQ
Amines are one of the most useful organic compounds. Amines are also of many types. It will be interesting to know what makes the amines so reactive and the type of products they can give.
Imagine there is a laboratory practical going on in your school. Two amines were given to you and asked you to distinguish between them.

How will distinguish which one is a primary amine or a secondary amine?
Well, you can’t decide on the basis of appearance because they both look similar.
Hence, to identify them properly you have to conduct several tests to confirm their existence.
Let’s understand how amines react with different compounds.
Table of content
- Definition of Amines
- Chemical Properties of Amines
- Basicity of Amines
- Reaction With Water
- Reaction With Acids
- Reactions With Electrophile
- Reactions with Metal Ions
- Alkylation Reaction with Alkyl Halides
- Schotten Baumann Reaction
- Carbylamine Reaction or Isocyanide Test
- Practice Problems
- Frequently Asked Questions
Definition of Amines
Amines are organic compounds of alkyl or aryl substituted ammonia. So they can be considered organic compounds derived from ammonia. One or more hydrogen atoms of ammonia can be replaced by organic hydrocarbons.
The substitution results in many different classes of amines:
Alkyl Amines: Amines with only alky groups are alkyl amines. The alkyl amines can be further classified into primary, secondary, tertiary and quaternary amines depending upon the number of alkyl groups attached to the nitrogen. Further, the alkyl groups can be identical or different. Accordingly, they are called simple or mixed amines respectively. Quarternary amine is salt between the tertiary amine and an acid
Aryl Amines: When the hydrocarbon substituents in ammonia are benzene derivatives, they are referred to as aryl amines. Here also there can be simple (containing the same aryl group) or mixed aryl groups (different aryl groups).
Heterocyclic amines: The nitrogen could be a part of a cyclic ring of carbon, which is called a heterocyclic amine.
Chemical Properties of Amines
Amines can undergo three different types of reactions:
- Based on the alkyl or aryl groups
- Based on the available hydrogen atoms attached to nitrogen and
- Based on the lone pair electron nucleophilicity of nitrogen
Following are the reactions of amines:
Basicity of Amines
Because of the lone pair of electrons on Nitrogen, amines have basic nature. They are more basic than water and less basic than hydroxide ions.
Amines react with acid to form salts called quarternary salts, which can be reversed by treatment with a base. Amines turn red litmus paper blue.
aliphatic or aromatic amine]
Reaction with Water
Due to the presence of a lone pair of electrons on the N-atom, amines like NH3 are stronger bases than H2O. Therefore, these amines act as Bronsted bases in water. In other words, they accept a proton from water to form alkyl or aryl ammonium hydroxides which ionize to furnish hydroxyl ions.
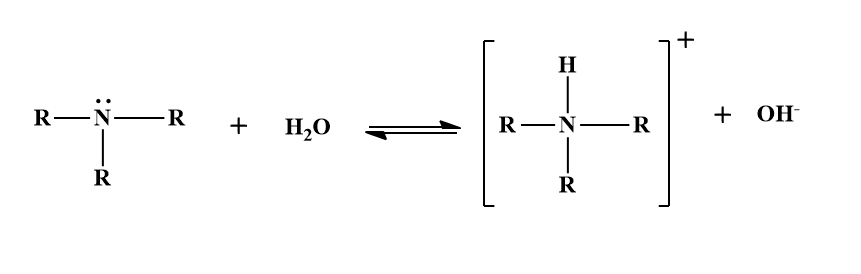
Due to the formation of hydroxide ions, the aqueous solutions of amines are basic in nature. The presence of the hydroxide ions in the aqueous solutions of amines is detected by the precipitation of metal hydroxides such as aluminium hydroxide (gelatinous white ppt.) or ferric hydroxide (brown ppt.) when AlCl3or FeCl3 is added to their aqueous solutions.
Reaction with Acids
Being basic, all types of amines react with mineral acids such as HCl, HNO3, H2SO4, etc. to form soluble salts.
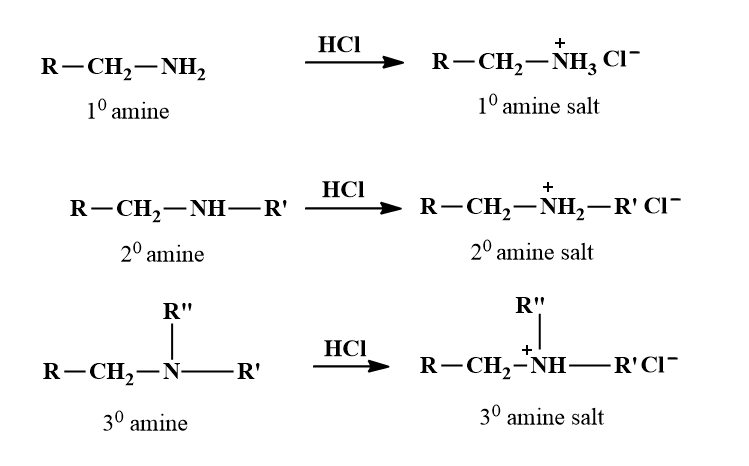
These amine salts are ionic compounds. They are non-volatile solids and generally decompose before their melting points are reached. They are soluble in water but are insoluble in non-polar solvents such as benzene, chloroform, ether, etc. This difference in solubility behaviour of amines and their salts is often used to detect amines and separate (or purify) them from non-basic compounds.
Reactions with an Electrophile
Due to the presence of a lone pair of electrons on the nitrogen atom, amines like ammonia are good nucleophiles and hence react with a variety of electrophiles (electron-deficient compounds) such as metal ions, alkyl halides, acid chlorides, acid anhydrides, chloroform, etc. as discussed below:
Reaction with Transition Metal Ions.
- Like ammonia, amines also form soluble coordination complexes with transition metal ions such as Ag+ , Cu2+ ions, etc. Thus, silver chloride dissolves in methylamine solution due to the formation of a soluble complex salt.
- Similarly to ammonia, amines such as methylamine, ethylamine, etc. react with Cu2+ ions to form a deep blue solution due to the formation of the following soluble coordination compound.
Alkylation Reaction with Alkyl Halides
The process of introducing an alkyl group into any molecule is called alkylation.
(a) With aliphatic amines.
- All three classes of amines undergo alkylation through nucleophilic substitution reactions on alkyl halides.
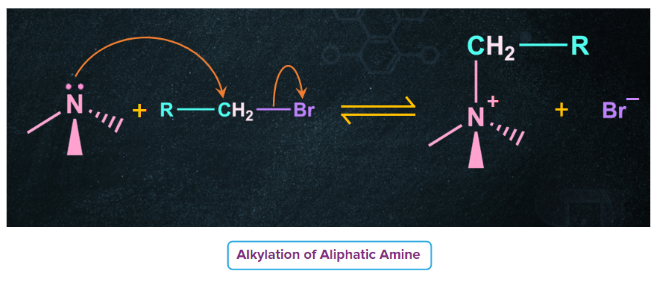
- Thus, a primary amine can be converted into secondary and tertiary amines and finally into quaternary ammonium salts. For example, when treated with an excess ethyl bromide, ethylamine undergoes alkylation to produce a mixture of diethylamine, triethylamine and tetraethylammonium bromide (quaternary ammonium halide).

- Quaternary ammonium halides on treatment with moist silver oxide or methanolic KOH are converted into their corresponding hydroxides. These quaternary ammonium hydroxides are white deliquescent crystalline solids which are as strongly basic as NaOH or KOH.
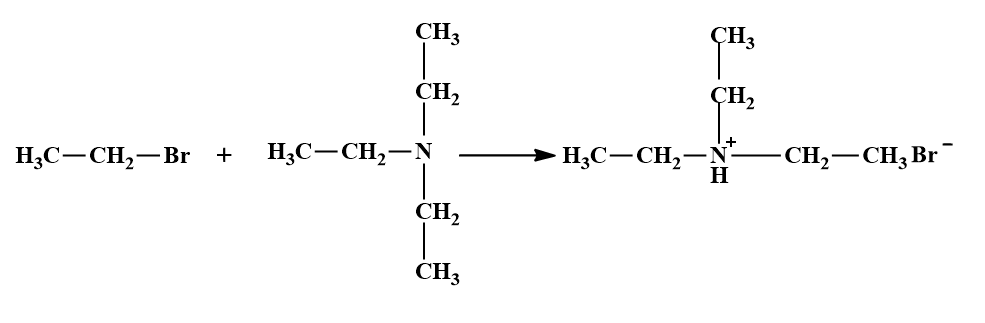
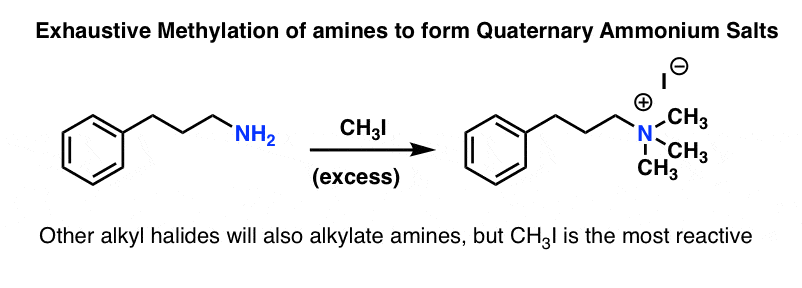
(b) With aromatic amines.
Aniline when heated with excess methyl iodide under pressure gives first N-methyl aniline, then N. N-dimethylaniline and finally trimethyl anilinium iodide. This process of converting an amine (10, 20, 30) into its quaternary ammonium salt on treatment with an excess of an alkyl halide is called exhaustive alkylation. However, if the alkyl halide used is methyl iodide, the process is commonly known as exhaustive methylation.
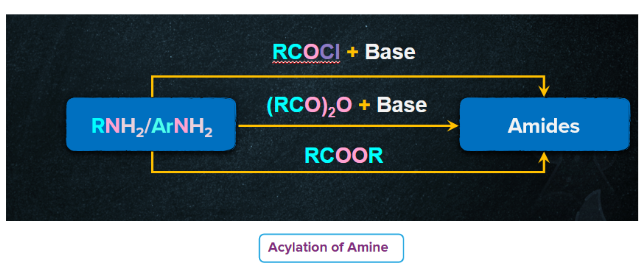
Acylation Reaction with Acid Chlorides and Acid Anhydrides.
The process of introducing an acyl group into any molecule is called acylation.
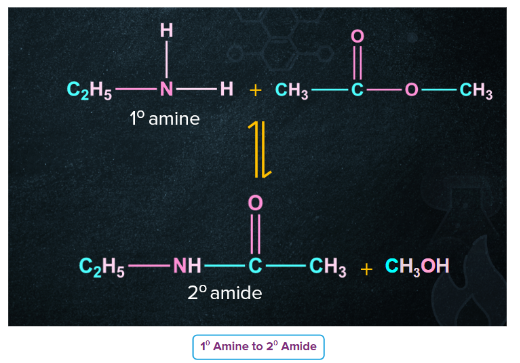
- Primary and secondary amines (but not tertiary amines because they do not contain an H-atom on the N-atom) undergo nucleophilic acylation reactions when treated with acid chlorides or acid anhydrides or esters to form N-substituted amides.
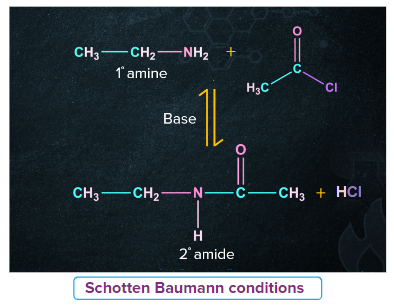
Schotten Baumann reaction
The Schotten Baumann reaction can also refer to the benzoylation of active hydrogen-containing compounds with the help of benzyl chloride and aqueous sodium hydroxide. Pyridine can also be used as an alternative to the sodium hydroxide base.

Sodium hydroxide or pyridine bases removes one of the product HCl and assist the forward reaction to completion.
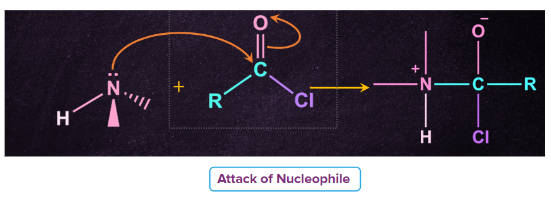
Mechanism
Step 1: Attack of nucleophile
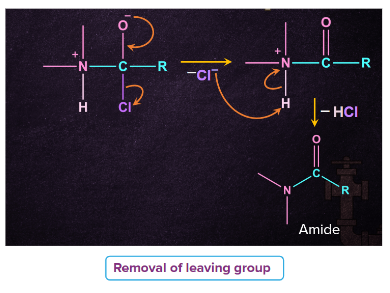
Step 2: Removal of leaving group
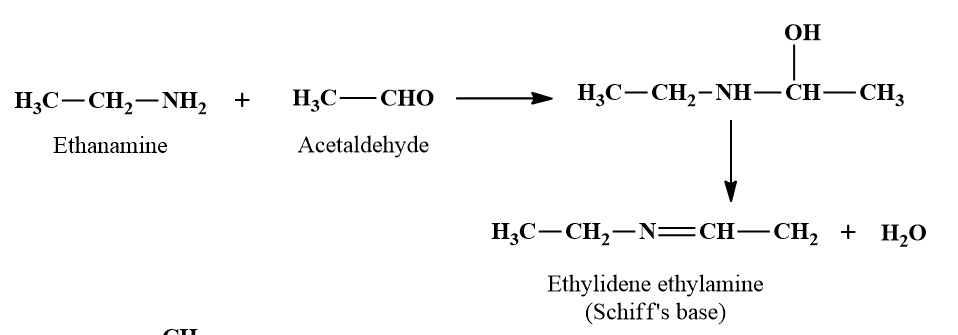
Reaction with Aldehydes and Ketones.
- Primary amines both aliphatic and aromatic react with aldehydes and ketones in presence of a trace of acid as a catalyst to produce azomethines called Schiff's bases or anils.
- Secondary amines both acyclic (i.e., N, N-dimethylamine) and cyclic (i.e., pyrrolidine, piperidine, morpholine, etc.) also react with aldehydes and ketones containing a-hydrogen atom/s to form first carbinolamines which being unstable readily lose a molecule of H2O to form stable a, B-unsaturated amines commonly called enamines (ene + amine - e = enamine).
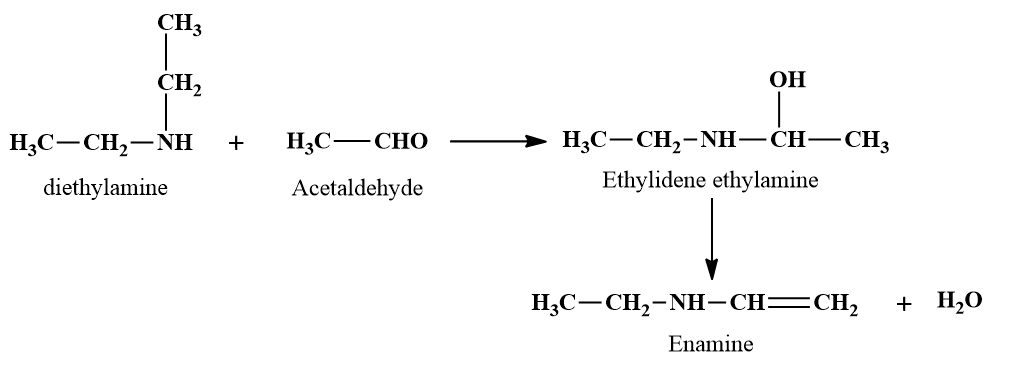
- The equilibrium is shifted in the forward direction by removing H2 as an azeotrope with benzene. Thus, enamine formation is usually carried out by refluxing the benzene solution of an aldehyde or a ketone with a secondary amine in presence of a trace amount of an acid such as p-toluenesulfonic acid (PTS).
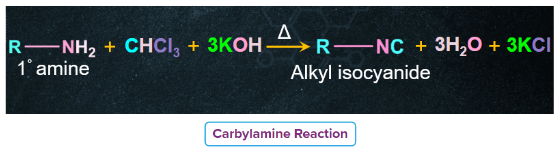
Carbylamine Reaction or Isocyanide Test
- Both aliphatic and aromatic primary amines when warmed with chloroform and an alcoholic solution of KOH produce isocyanides or carbylamines which have very unpleasant odours.
- In contrast, secondary and tertiary amines (both aliphatic and aromatic) do not give this test. Therefore, this test is used to distinguish primary amines from secondary and tertiary amines.
Practice Problems
Q1. Primary amines react with benzoyl chloride to give
A. Benzamide
B. Ethanamide
C. Imides
D. imines
Answer: (A)
Solution: Primary amines react with benzoyl chloride to give benzamide. This reaction is known as benzoylation. Benzoylation is a chemical reaction in which a benzoyl group is instigated by removing H– that is attached to O or N or any aromatic ring.
Q.2 What is obtained when benzoyl chloride reacts with aniline in the presence of sodium hydroxide?
A. Benzoic acid
B. Benzanilide
C. Acetanilide
D. Azobenzene
Answer:(B)
Solution: When benzoyl chloride reacts with aniline in the presence of sodium hydroxide it gives Benzanilide. This is known as Schotten Baumann's reaction. 
Q3. Acetylation of a secondary amine in alkaline medium yields
A. N, N. dialkylacetamide
B. N,N-dialkylamine
C. N,N-dialkylamide
D. acetyl dialkylamine
Answer: (A)
Solution: Primary and secondary amines (but not tertiary amines because they do not contain an H-atom on the N-atom) undergo nucleophilic acylation reactions when treated with acid chlorides or acid anhydrides or esters to form N-substituted amides.
Q4. Primary and secondary amines react with an acid chloride or acid anhydride to form
A. Tertiary ammonium salt
B. Substituted amides
C. Diazonium salt
D. Nitro compounds
Answer: (B)
Solution: Primary and secondary amines react with an acid chloride or acid anhydride to form substituted amides
Frequently Asked Questions (FAQs):
Q1. What is the importance of amines in our daily life?
Answer: Amines are used in making azo dyes and nylon apart from medicines and drugs. They are widely used in developing chemicals for crop protection, medication and water purification. They also find use in products of personal care. Ethanol amines are the most common type of amine used in the global market.
Q2. Why amines are soluble in water?
Answer: Aniline and other arylamines are generally colourless, but they get coloured if stored in open due to atmospheric oxidation. Lower aliphatic amines can form hydrogen bonds with water molecules hence they are soluble in water.
Q3. What is the pH of amine?
Answer: Most simple alkyl amines have pKa's in the range of 9.5 to 11.0, and their water solutions are basic (have a pH of 11 to 12, depending on concentration).
Q4. Suggest a test to identify the presence of a primary amine in mixed amines
- Both aliphatic and aromatic primary amines when warmed with chloroform and an alcoholic solution of KOH produce isocyanides or carbylamines which have a very unpleasant odour.
- In contrast, secondary and tertiary amines (both aliphatic and aromatic) do not give this test. Therefore, this test is used to distinguish primary amines from secondary and tertiary amines.


