-
Call Now
1800-102-2727
Adsorption: Definition, Types and Application of Adsorption, Adsorption Isotherm, Practice Problems & FAQs
Which is the most polluted city in India?
Well, the data changes frequently but Delhi has been the constant name on this list for a very long time.
The reason for pollution is very subjective. Industries, excessive usage of vehicles powered by fossil fuels and burning of agricultural waste. They all are equally responsible for the hazardous air quality index of Delhi.

The pollution level in winter rises so much that it becomes impossible to inhale direct air.
You must have seen a lot of people have started using air purifiers. But it’s not affordable for everyone.

Can you think of any cost-effective air purifier which is affordable for everyone?
Activated charcoal, even if you are unaware of the function of this product but i am pretty sure that you are quite familiar with activated charcoal. Starting from toothpaste brand to soap brand everyone is frequently using this word for their advertisement.

Activated charcoal is produced by heating charcoal in the presence of a gas. The charcoal develops numerous internal pores as a result of this process. Activated charcoal can trap chemicals to its pores. To treat poisoning, activated charcoal is frequently employed. These activated charcoal can be used as an air purifier to trap dust and harmful polluted air. This process is called adsorption. I haven’t mistyped it, yes it is adsorption.
Let’s unfold the concept of adsorption and understand how it is different from absorption!
TABLE OF CONTENTS
- Definition of Adsorption
- Mechanism of Adsorption
- Thermodynamics of Adsorption
- Types of Adsorption:
- Adsorption Isotherm:
- Factors Affecting Adsorption:
- Applications of Adsorption:
- Differences between Absorption and Adsorption:
- Practice Problems
- Frequently Asked Questions-FAQs
Definition of Adsorption:
Adsorption is a process in which a substance builds up in molecular species at higher concentrations on a surface. Hydrogen, Nitrogen and Oxygen are three gasses that adsorb on activated charcoal. A distinction between adsorption and absorption must be made in the interim. The two processes use entirely distinct mechanisms.
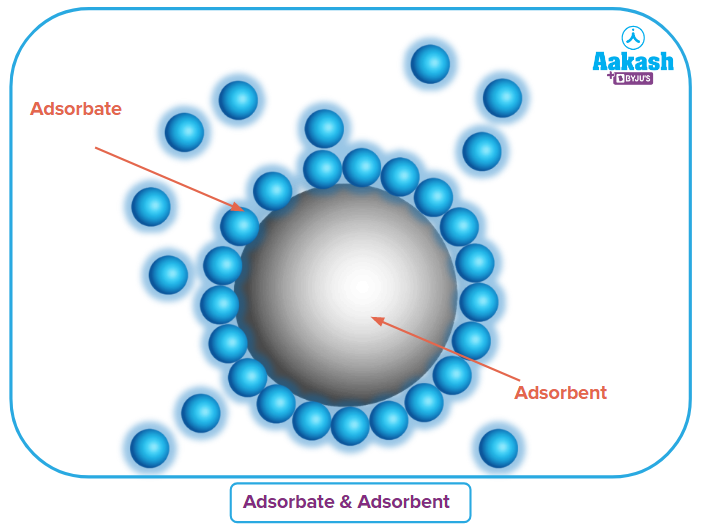
The molecules that are adsorbed are typically referred to as the adsorbate, and the solids used to do so are known as adsorbents. The charcoal found in gas masks, which is used to filter poisons or other impurities from an air stream, is an excellent example of an adsorbent.
Interface & Surface:
A region where two phases meet & interact is called an interface. If one of the faces is gas then the interface is commonly known as surface.
In 1881, Heinrich Kayser, a German physicist, first used the term "adsorption." Adsorption is frequently explained as a surface phenomenon where particles stick to the material's top layer. It typically involves molecules,ions , or even atoms of a liquid, gas, or solid that are attached to the surface in a dissolved state.
Surface energy is primarily responsible for adsorption. The particles that can be left uncovered on the surface usually attract other particles to where they are. It's interesting to note that adsorption occurs naturally in numerous physical, biological, chemical, and natural systems and is used extensively in industry.
There are two components necessary for the adsorption process,
|
Adsorbate |
Adsorbent |
|
substance that is deposited onto another substance's surface. for instance, the gasses N2,H2, and O2. |
Surface of a material on which an adsorbate binds. Charcoal, alumina, and silica gel are a few examples. |
Desorption:
Adsorbed molecules are removed from a surface in a process known as desorption. This is a reverse of process adsorption.
Example: Have you ever noticed how water droplets adsorb on the glass surface during winter in cars? Now to remove them instantly we quickly turn on the blower of the car in heating mode. After a couple of seconds, you can observe a clear glass. This is reversing the effect of adsorption.
Sorption:
The process in which adsorption & absorption takes place simultaneously is called sorption.
Example: Dying in fabrics. As we know if the colors on fabrics are only adsorbed at the surface then definitely after a certain wash color will fade. To avoid this situation during the dying process of fabrics, both the adsorption and absorption process happens simultaneously.
Mechanism & Process of Adsorption:
All forces in action within the adsorbent are mutually balanced between the particles. The adsorbent's surface particles are not completely encircled by the same particles on every side. Therefore, they have residual or unbalanced forces of attraction.
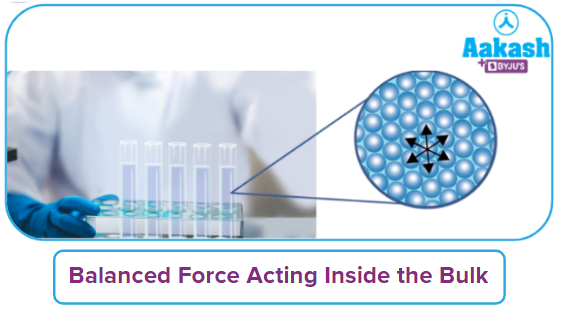

The adsorbate particles are drawn to the surface of the adsorbent by these unbalanced forces of attraction.
Thermodynamics of Adsorption:
Adsorption always results in a reduction in the surface's residual forces, or in the surface energy, which is visible as heat. Therefore, during adsorption, heat is released (exothermic process). Adsorption is therefore always exothermic. ( H <0)
As the adsorbate particles are bound to the surface, or when their freedom is constrained, the degree of randomness of the particles decreases with adsorption. As a result, entropy drops( ΔS < 0).
For a reaction be to be spontaneous, Gibbs free energy, ΔG< 0.
According to the equation ΔG = ΔH - TΔS, ΔG can be negative only if ΔH is significantly negative, as - TΔS is positive.
We know that for adsorption, H <0, ΔS < 0
If ΔH is initially significantly negative at constant temperature and pressure, then the sum of these two causes ΔG to be negatively polarized.


As the process of adsorption continues, ΔH becomes less and less negative, until eventually it equals TΔSand ΔG is zero. Equilibrium has now been attained.
Enthalpy of Adsorption (∆HAdsorption):
The amount of heat produced when 1 mol of an adsorbate is adsorbed on a specific adsorbent at equilibrium with adsorption is called as Enthalpy of adsorption (∆HAdsorption) . It is dependent upon the characteristics of the adsorbent and adsorbate.
Types of Adsorption:
Adsorption is divided into two categories based on the forces that interact between the adsorbent and the adsorbate.
|
Parameter |
Physical adsorption |
Chemical adsorption |
|
Definition |
Physical adsorption refers to the adsorption of a gas onto the surface of solid by weak intermolecular forces of attraction (van der Waals forces) without the creation of a chemical bond between the adsorbent and the adsorbate. |
Chemical adsorption, also known as chemisorption, is the process that occurs when the forces of attraction between the adsorbent and the adsorbate are chemical forces of attraction or chemical bonds. Chemisorption occurs when an adsorbate monolayer forms on an adsorbent. It has a high adsorption enthalpy. It can occur at any temperature. Chemisorption first rises with temperature increases and then falls. |
|
Cause |
Physical forces are the root cause of this kind of adsorption. |
Chemical forces are what lead to this kind of adsorption. |
|
Strength |
Physisorption is a weak phenomenon as compared to chemical adsorption. |
Chemical adsorption is a very strong process. |
|
Layer |
This adsorption process involves multiple layers. |
This kind of adsorption resembles a single-layered phenomenon. |
|
Occurrence |
Physical adsorption occurs everywhere on the adsorbent and is non-specific. |
Chemisorption occurs at reaction centers on the adsorbent and is very specific. |
|
Factors affecting |
Effects of physisorption include surface area, temperature, pressure, and adsorbate type. |
Chemisorption is influenced by surface area, temperature, and the type of adsorbate. |
|
Activation energy |
Low energy (20–40 kJ/mol) is required for activation. |
The activation energy ranges from 40 to 400 kJ/mol. |
|
Example |
|
|
Adsorption Isotherm:
Isotherms are frequently used to describe adsorption. It is because temperature affects the entire process greatly or plays a significant role in both. Furthermore, the adsorption method is described using a number of isotherm models. These consist of;
Freundlich Isotherm Theory:
Freundlich adsorption isotherm establishes the relationship between the extent of adsorption with pressure at constant temperature.
Where,
x= amount of gas adsorbed on the 'm' gram of adsorbent
P=Pressure
K & n=adsorption constant
The Freundlich adsorption isotherm's major flaw is that it breaks down at high pressure. The process of multi-layered adsorption could not be explained by it.
Langmuir Theory:
In 1916, Langmuir proposed the idea that the variation of extent of adsorption takes place at a constant temperature when pressure varies. The ability of a gas molecule to bind to any one site is presumed to be independent of whether or not the neighboring sites are occupied, and all adsorption sites are thought to be equivalent. It is also assumed that there is a dynamic equilibrium between gas molecules that have been adsorbed and those that have not.
Postulates:
These principles can be drawn from the Langmuir adsorption isotherm.
- Adsorbent contains a fixed number of active sites on which adsorption takes place.
- Adsorption only happens at the first layer, it is a monolayer phenomenon.
- Adsorbed molecules don't interact with each other & have the same heat of adsorption.
- Adsorbed and desorbed molecules exist in equilibrium.
BET Theory:
In 1938, Brunauer, Emmett, and Teller put forth the BET theory. According to this theory, multilayer adsorption forms during physisorption. This theory also discusses the uniformity of solid surface adsorption sites. It is assumed that when adsorption takes place at one site, it won't have an impact on adsorption at nearby sites.
Factors Influencing Adsorption:
Temperature:
Temperature has a direct impact on absorption but it is only applicable for physical adsorption.
With the increase in temperature adsorption decreases. Temperature is inversely proportional to adsorption.
Adsorbent (s) + Adsorbate (g) ⇌ [Adsorbent---Adsorbate] + ΔH
We are aware that the processes of adsorption and desorption are both endothermic. Le Chatelier's principle states that a rise in temperature favors desorption.
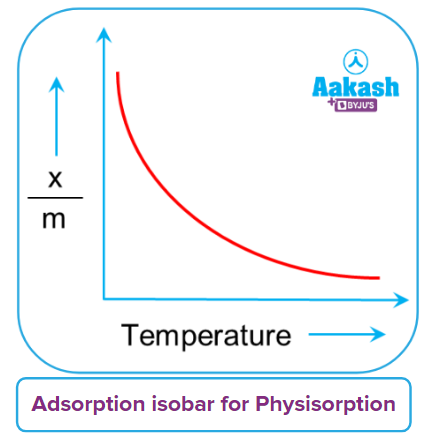
A curve known as the adsorption isobar can be used to express how the amount of gas adsorbed by the adsorbent varies with temperature. It shows the relationship between xm and temperature at a fixed pressure. Here, x is the mass of the gas that has been adsorbed, m is the mass of the adsorbent, and xm is the mass of gas that has been adsorbed as a percentage of its mass.
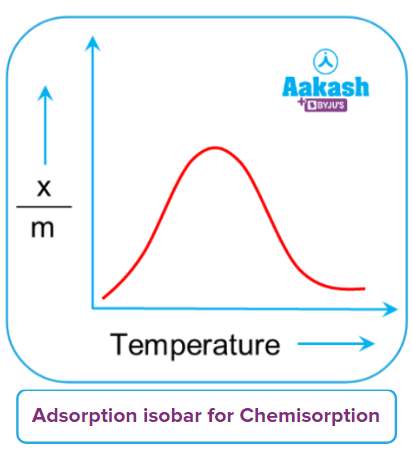
For physisorption and chemisorption, separate adsorption isobar plots are displayed. Physical adsorption continuously decreases with temperature increase, or the gas is removed from the surface.
Chemical adsorption initially reaches a maximum then decreases continuously. The initial increase in chemisorption is caused by the heat supplied acting as activation energy, and more and more adsorbate molecules gain energy and possess energy greater than activation energy. As a result, adsorption increases as temperature rises.
Pressure:
With the increase in pressure the phenomenon of adsorption increases but after a certain interval pressure has no role on adsorption. As we know there must be a specified number of sites where adsorption can actually take place. Once these sites are occupied further adsorption doesn’t happen.
The pressure of the gas affects the amount of adsorption (xm). A curve known as the adsorption isotherm can be used to express variations in the quantity of gas that the adsorbent can bind to under pressure and at constant temperature.
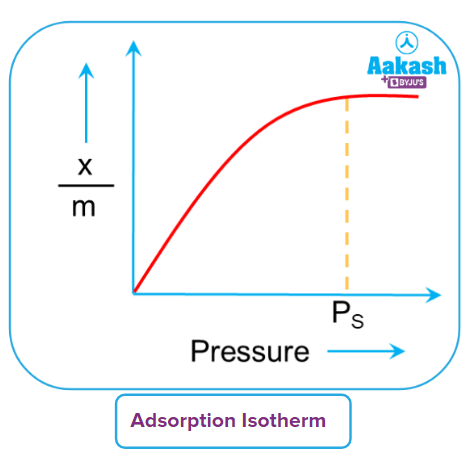
Pressure causes an increase in adsorption's (xm) extent. The saturation pressure, which is the highest value it can reach and corresponds to PS pressure, is reached.
Consider the following illustration of a beaker with a piston attached. In the beaker, NO2 gas is added after placing charcoal at the bottom.
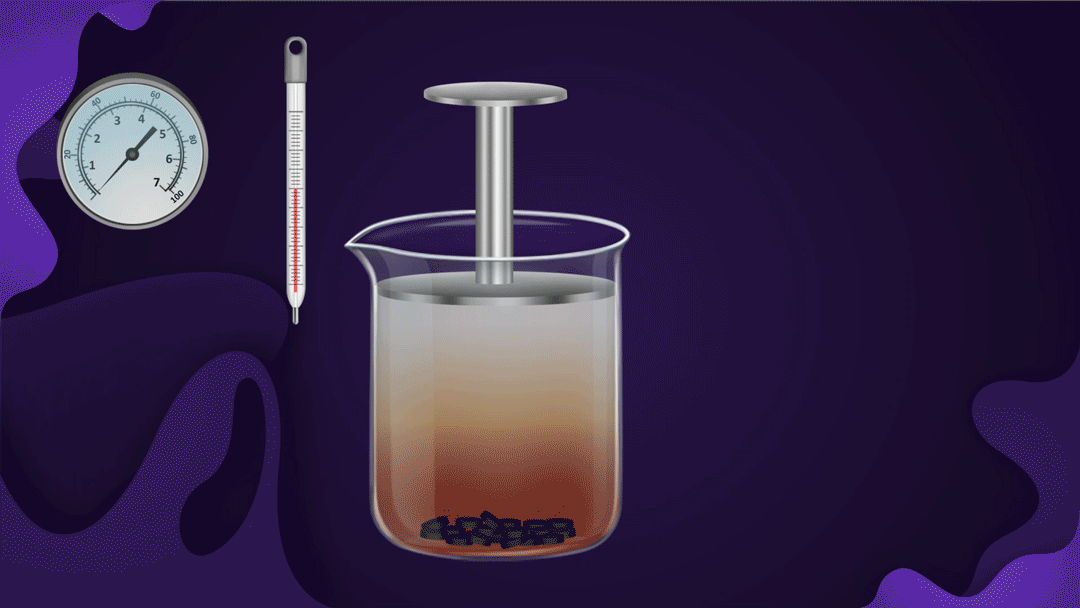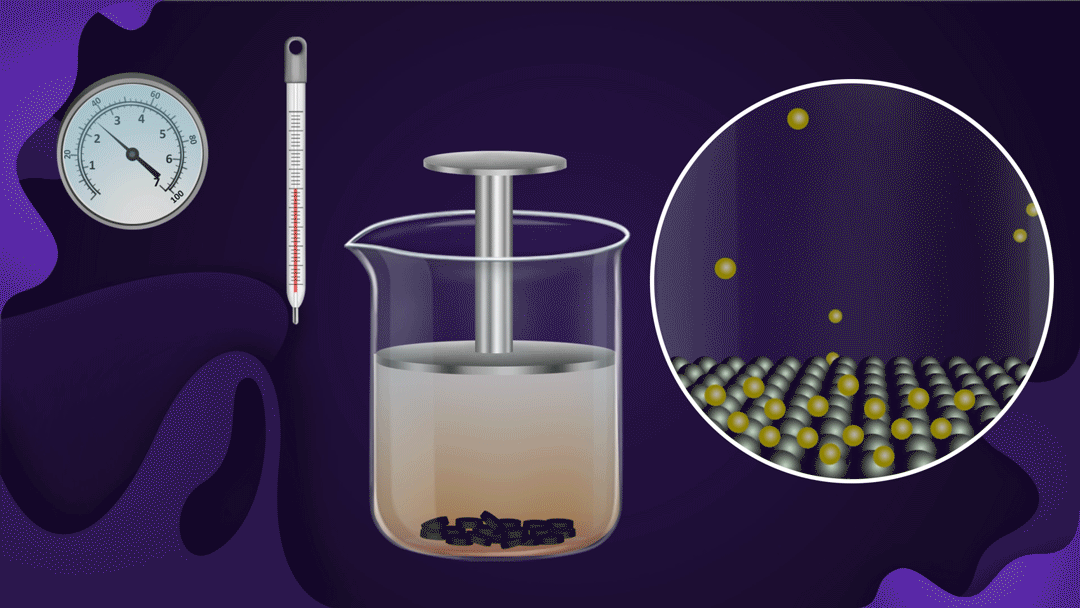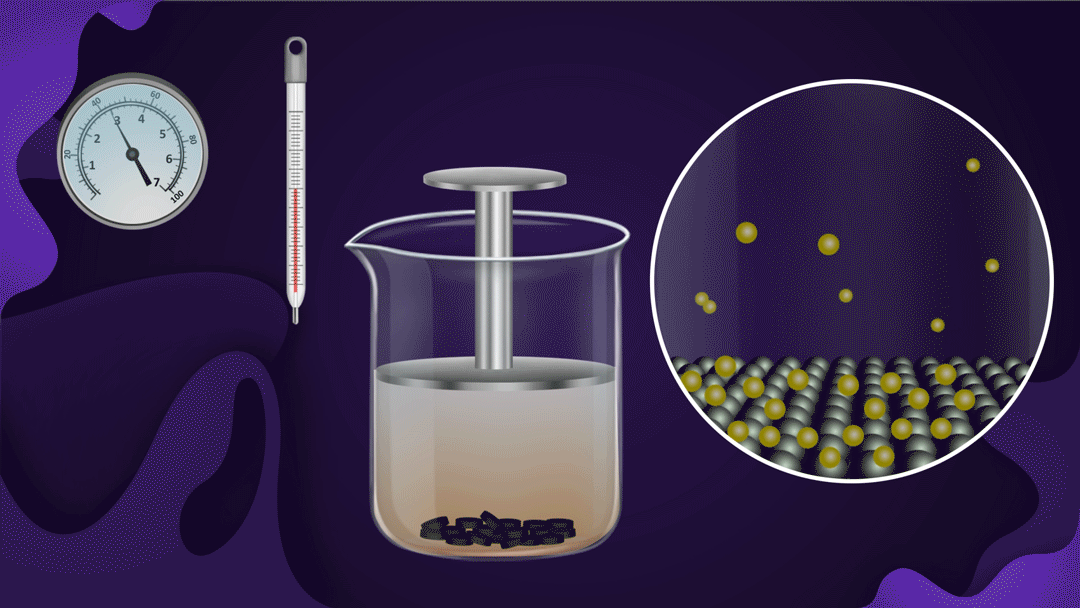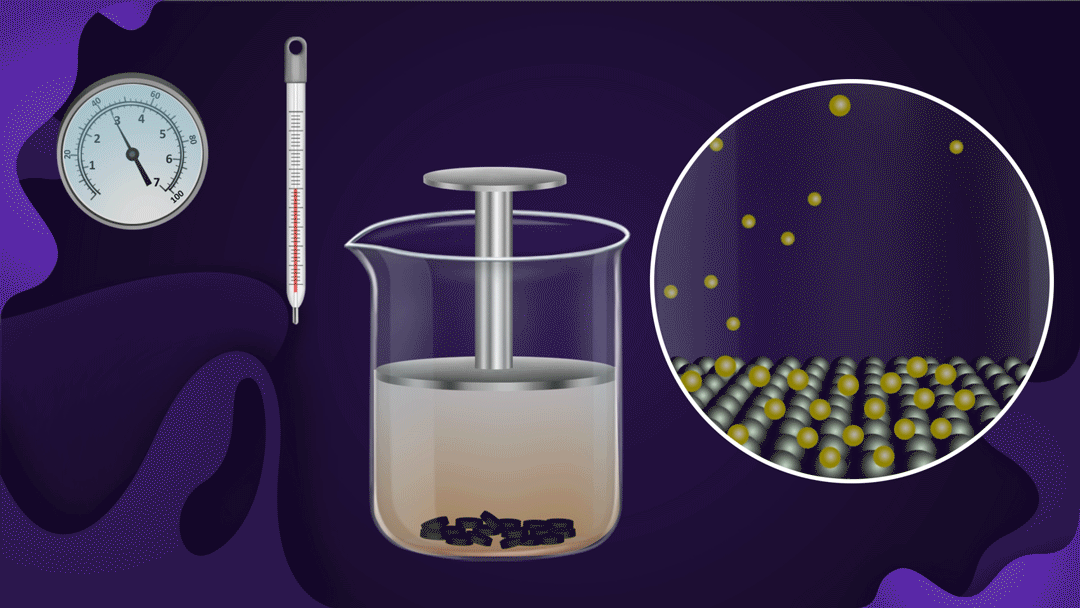

The pressure of the gas rises when the piston is pressed while the temperature remains constant, which speeds up the adsorption of NO2 gas onto the charcoal. The reduction in NO2 gas's colour intensity as the piston moves inwards can be used to detect increased adsorption.
Surface Area of Adsorbent:
Larger the surface area of the adsorbent greater will be the number of sites available for adsorption. Due to this reason powdered charcoal is much more effective than solid charcoal.

![]()
Applications of Adsorption:
Masks for air pollution:
These are made of silica gel or powdered activated charcoal, and when smoke or dust passes through them, the particles become adsorbent on their surface.
Noble gas separation:
In the presence of heated coconut charcoal, a mixture of the noble gasses Ne, Ar, and Kr is passed through a Dewar's flask. Neon remains after Argon and Krypton have been absorbed.
water purification:
Impurities are entrapped on the alum and removed from the water by the addition of an alum stone.
Elimination of moisture and humidity :
Moisture from the air is eliminated by putting silica gel on which water molecules get adsorbed.
Adsorption chromatography:
Hormones and pigments are separated using this method.
Ion exchange method:
This method is generally employed to eliminate the hardness of water through resin.
In metallurgy:
The particle is adsorbed on the froth during the concentration of ore by froth floatation.
Differences between Absorption and Adsorption:
|
Parameter |
Adsorption |
Absorption |
|
Definition |
Adsorption is a process in which a substance builds up in molecular species at higher concentrations on a surface. |
Absorption is the total amalgamation of one substance into another. |
|
Phenomena |
Adsorption is a surface phenomena. |
Absorption is a bulk phenomenon. |
|
Spontaneity |
Adsorption is a continuous process. |
Absorption is not a continuous process. |
|
Uniformity |
Adsorption only occurs at the surface, hence, it is non-uniform throughout the system. |
It takes place uniformly throughout the system. |
|
Molecular Interaction |
During the adsorption process the interaction between the molecules is quite low as compared to absorption. |
During the absorption process the interaction between the molecules is quite high. |
|
Example |
When silica gel is placed in a watery environment, it adsorbs the water. At the surface of silica gel, there is a high concentration of water |
Calcium chloride, on the other hand, absorbs water when placed in a moist environment. The liquid is equally dispersed |
Recommended Video: Adsorption
Practice Problems:
Q1. Which one of the aforementioned forces contributes to physical adsorption?
- Gravitational Force
- Electrostatic Force
- Hydrogen bonding
- Van der Waals force
Answer: (D)
Solution: The Van der Waals force, which is very weak, is what causes the molecules of the adsorbate to adhere to the surface of the adsorbent during physical adsorption. The forces that lead to the condensation of gas into liquid and the Van der Waals force are similar.
Q2. Which of the following statements regarding the degree of physisorption is true?
- Increases with increasing surface area,
- decreases with increasing Van der Waals forces,
- increases with increasing temperature,
- increases with increasing surface area.
Answer: (D)
Solution: Physisorption is an exothermic process. The Le-Chatelier principle states that a drop in temperature favors exothermic reactions. As a result, physisorption decreases with temperature.
Q3. Which of the following statements about physisorption is false?
- Change in enthalpy is less than zero.
- Change in entropy is greater than zero.
- Physisorption is a spontaneous process.
- Physisorption is a reversible process.
Answer: (B)
Solution: Because only weak Van der Waals forces, which are easily broken, hold the molecules of the adsorbate to the adsorbent, physical sorption is reversible. Since physisorption produces heat, H is negative. Adsorption happens on its own. The movement of the adsorbed molecule is constrained during adsorption. The entropy change (S) is consequently negative.
Q4. With the increase in surface area, ____ also increases.
- Physisorption
- Chemisorption
- Both A and B
- None of the above
Answer: (C)
Solution: As the surface area of the adsorbent increases, more adsorbate molecules get adsorbed on the surface of the adsorbent. The extent of adsorption is directly proportional to the surface area of the solid for both physisorption and chemisorption. Therefore, with the increase in surface area, both physisorption and chemisorption increase. So, option C) is the correct answer.
Frequently Asked Questions-FAQs:
1. Why does physisorption result in no chemical bond being formed?
Answer: Low temperatures are typically favorable for physisorption to take place. The only van der Waals forces that exist at these temperatures are weak ones. Chemical bonds cannot be formed at low temperatures due to a lack of activation energy. In physisorption, therefore, no chemical bonds are created.
2. What is the critical gas temperature?
Answer: The temperature at and above which a substance's vapor cannot be liquefied, regardless of the amount of pressure used, is known as the critical temperature of the substance.
3. Why are gasses that are easily liquefiable and quickly adsorbed?
Answer: The van der Waals’ force of attraction are greater in easily liquefiable gasses like NH3, SO2, etc than in permanent gasses like H2, N2, etc. As a result, easily liquefiable gasses are absorbed readily.
4. Why does physisorption have a low enthalpy of adsorption?
Answer: As weak physical forces like van der Waals’ forces are involved in physisorption, the attraction between the adsorbate and the adsorbent is less. This is the reason why the enthalpy of adsorption is low for physisorption.




