-
Call Now
1800-102-2727
Addition of Halogens - Addition of Dihalogen, Hypohalous Acid, and Hydrogen Halides to Alkenes & Alkynes, Practice Problems and FAQs
Have you ever seen clear marble-style glass gems in craft stores and wondered if people wear them plain or customize them in some way when wearing them as pendants, and if so, how?
This is just one of many ways to customize a glass pendant, but it is by far our favourite! Look at how well the alcohol inks blend and mix to create beautiful swirling effects. But how can we prepare for them?
The answer is an alkene. To convert alkenes to alcohol, three different chemical methods can be used. An alkene is used as a starting material in a number of reactions, some of which are discussed below.
Table of Contents
- Addition of dihalogen to alkenes
- Addition of Hypohalous acid to alkene- Halohydrin
- Addition of Hydrogen Halides (HX) to alkenes
- Addition of dihalogen to alkyne
- Addition of hypohalous acid to alkynes
- Addition of hydrogen halides to alkynes
- Practice Problems
- Frequently Asked Questions
Addition of dihalogen to alkenes:
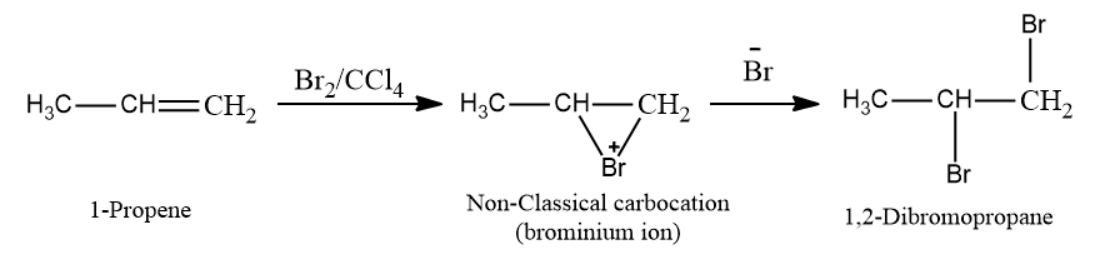
Halogen addition (mainly Cl2 and Br2) entails the addition of chlorine or bromine in the presence of an inert solvents such as CCl4. An electrophilic addition reaction involving the formation of a cyclic halonium ion is the addition of halogens to alkenes.
In the presence of an inert solvent (CCl4), 1-propene reacts with bromine to form a non-classical carbocation (bromonium ion) as an intermediate. Now, Br- attacks the cyclic intermediate, and 1,2-dibromopropane is formed as the desired product.
Addition of Hypohalous acid to alkene- Halohydrin:
Halohydrins are formed by the electrophilic addition of other halogen-containing reagents to an alkene, such as hypohalous acids, or HOX. In each of these reagents, the electrophilic element is halogen. Oxygen has a higher electronegativity than chlorine because O has δ− charge and Br has δ+ charge.
Example 1: Addition of HOBr to ethene to form 2-Bromoethanol

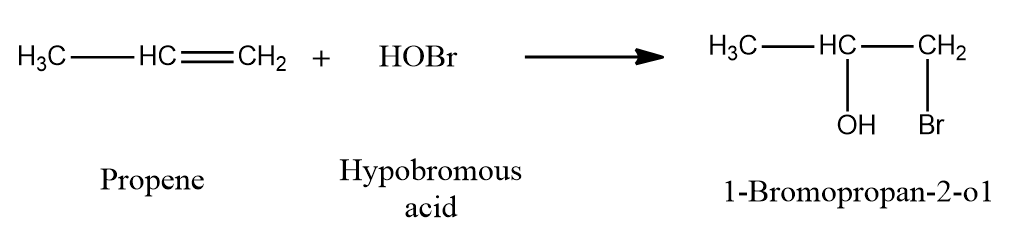
Example 2: Addition of HOBr to propene to form 1-Bromopropan-2-ol
Halohydrins, on the other hand, generally are not generated by adding a hypohalous acid directly to an alkene; instead, the alkene is reacted with Br2 or Cl2 in the presence of water. Let us discuss in detail by mechanisms.
Mechanism:
Step 1: The first step is the same as it is for halogenating alkenes. When a halogen, in this case bromine, interacts with an alkene, a cyclic bromonium ion is formed.
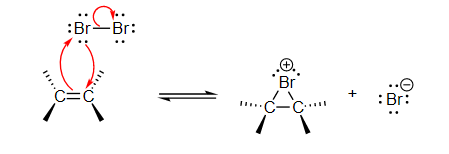
Step 2: In step two, the bromide ion is not attacked; instead, water acts as the nucleophile. Water is more abundant than bromide ions, so it is more likely to be available in the appropriate position for nucleophilic attack. Water's nucleophile property outperforms that of the bromide ion. The stronger a nucleophile, the more likely it is to attack. Nucleophiles attack is preferred on more substituted carbon.
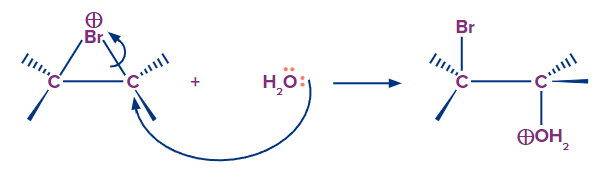
Step 3: It is the final acid-base step in which a proton is lost from oxygen to the solvent (water), yielding neutral halohydrin. In this case, the addition reaction produces bromohydrin.

Addition of Hydrogen Halides (HX) to alkenes:
Hydrogen halides (HCl, HBr, HI) when added to alkenes give alkyl halides. This is known as the hydrohalogenation process. It is an electrophilic addition reaction.
Alkene + HX Alkyl halide
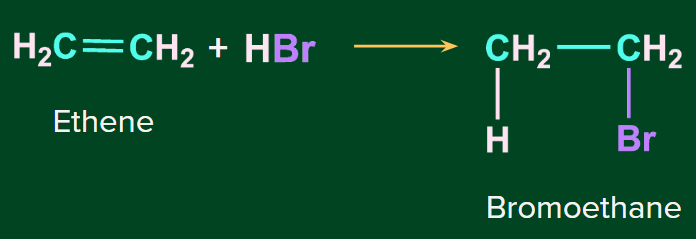
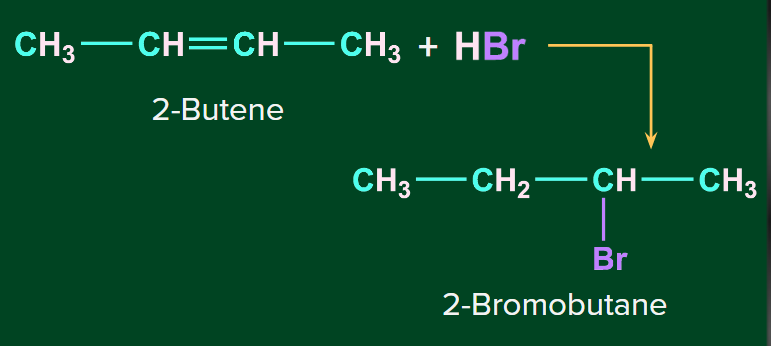
Addition of HBr to symmetrical alkenes:
In the addition reaction of HBr to symmetrical alkenes, the electrophilic addition mechanism occurs, in which the hydrogen ion attaches to one carbon and bromide attaches to the second carbon of the double bond.
Addition of HBr to unsymmetrical alkenes:
When HBr is added to unsymmetrical alkenes, double bonds can break in two ways, with one carbon receiving the negative charge and the other receiving the positive charge. Because the bond is broken, the bond-forming carbocation is the most stable of the three.
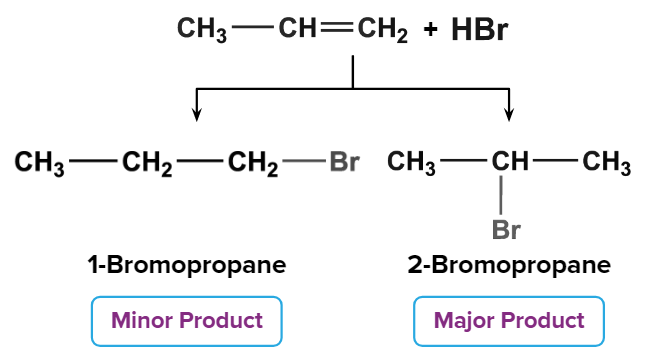
Markovnikov's rule (Rule states that the negative part of the addendum (adding molecule) gets attached to the carbon atom with the least number of hydrogen atoms) is followed to determine the major product formed upon the addition of HBr to an unsymmetrical alkene.
Mechanism (Markovnikov Addition):
Step 1: The H+ ion attacks the double bond (electrophilic addition of H+) and gets attached to the carbon having more hydrogen atoms.
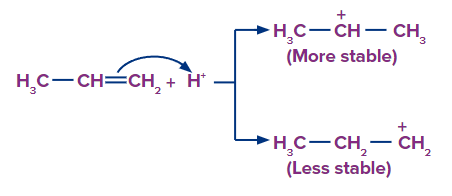
Step 2: Since, the 2o carbocation is more stable than 1o carbocation, so the stable carbocation will be available more for the attack of Br- and the product formed from 2o Carbocation is the major product.
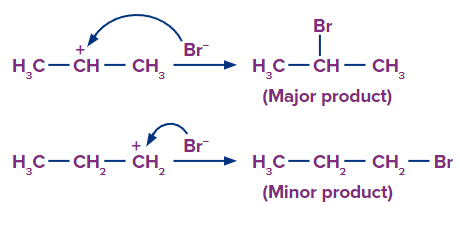
Rearrangement of Carbocation:
The H+ ion attacks the double bond and it gets attached to the carbon having more hydrogen atoms. The 2o carbocation undergoes rearrangement (1,2-hydride shift) to form a more stable 3o carbocation if there is an option to rearrange. Finally, Br- ion attacks on the 3o carbocation to form the major product.
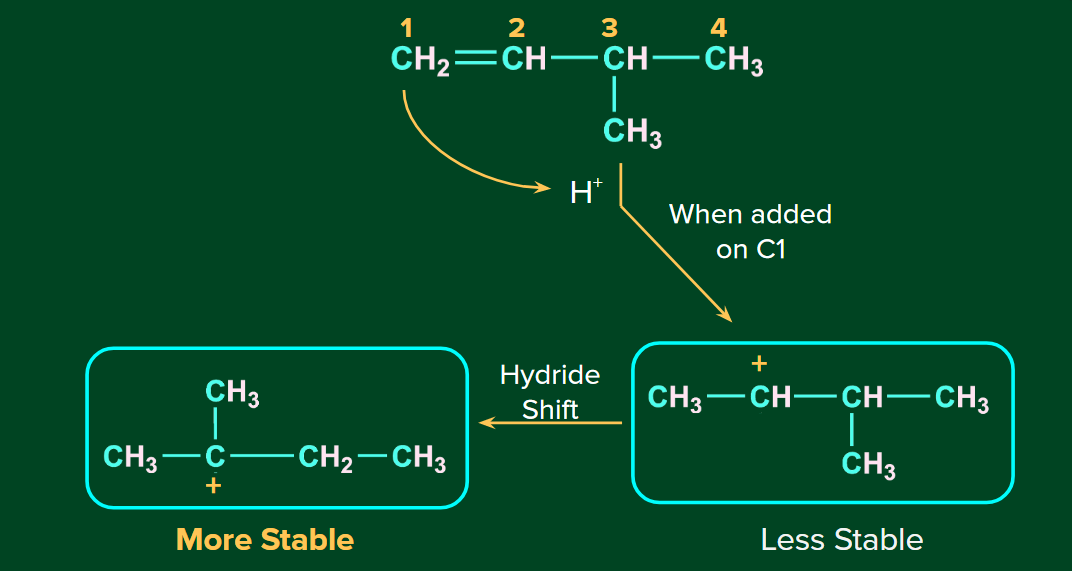
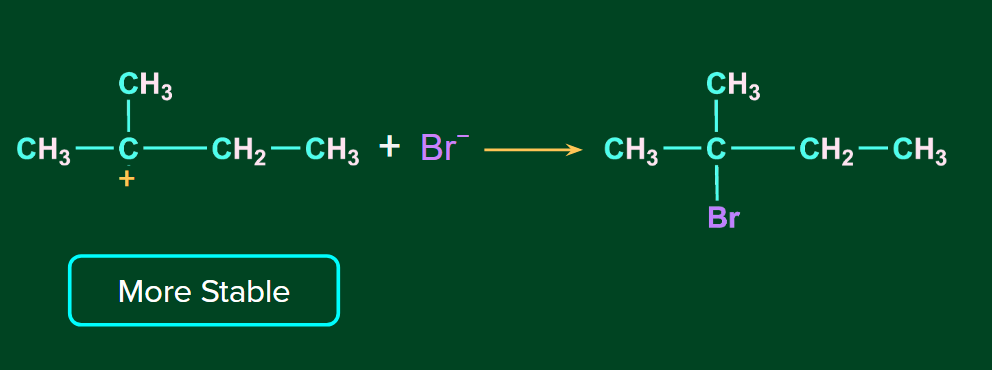
Order of reactivities of hydrogen halides:
The bond length of HX increases as we move down the group, while the bond strength decreases (i.e., the H-X bond becomes weak and can be easily dissociated). As a result, the reactivity of hydrogen halides towards addition reactions increases as one moves down the group.
HF < HCl < HBr < HI
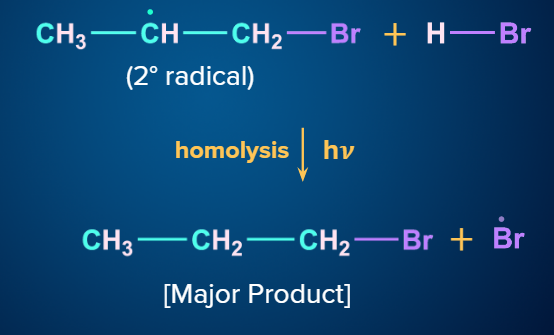
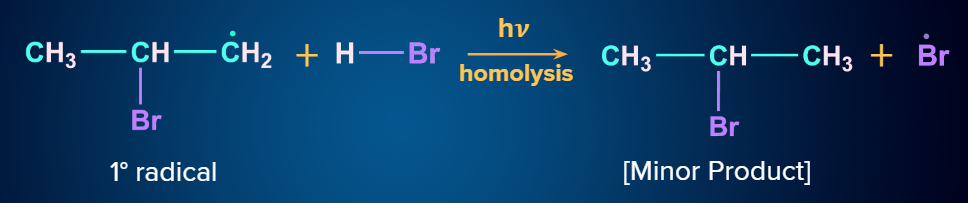
Mechanism (Anti-Markovnikov Addition):
When unsymmetrical alkenes are treated with HBr in the presence of peroxide, they follow Anti - Markovnikov’s rule. The addition occurs via a free radical process in the presence of peroxide. There is a formation of less substituted haloalkanes as a major product. This is known as the Peroxide effect. The Kharasch effect is another name for the peroxide effect.
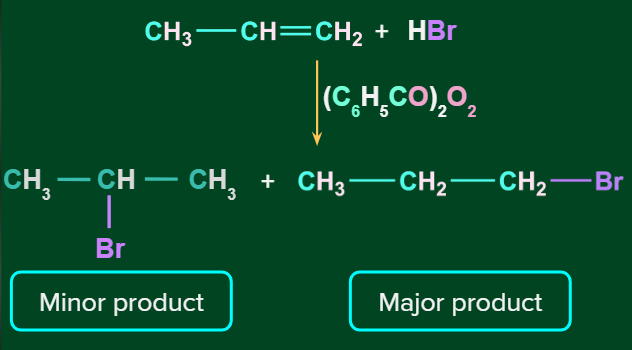
Note: Where (C6H5CO)2O2 is benzoyl peroxide.
Mechanism:
The mechanism containing 3 steps is given as follows:
Step - 1: Chain initiation:
The peroxide bond of RCO–O–O–COR breaks into radicals (i.e., RCOO) in the presence of sunlight, which is then followed by the removal of carbon dioxide and the formation of alkyl radical (i.e., R).
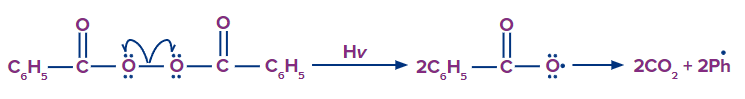
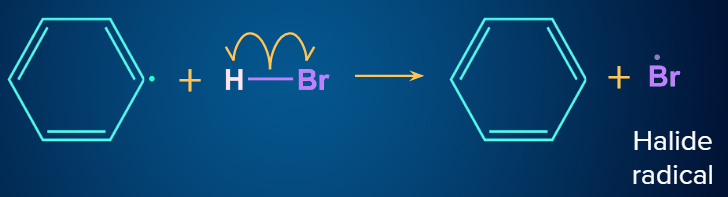
Now, the phenyl radical (Ph) attacks on HBr and results in the formation of R−H (i.e., benzene in benzoyl peroxide) and Br radicals.
Step - 2: Chain propagation:
The order of stability of the free radicals is 3oradical>2oradical>1oradical. The two intermediates formed are given and we know that a 2o free radical is more stable than a 1o free radical.
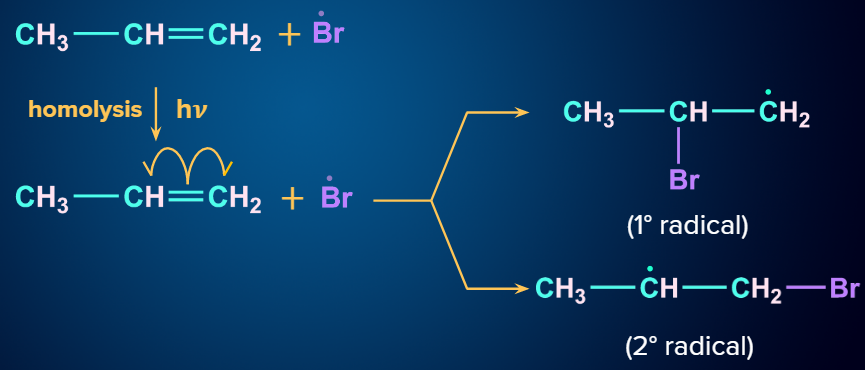
Step - 3: Chain Termination:
The hydrogen radical from HBr will attack the 2o free radical to give the major product. Hence, in the major product, Br will be attached to the carbon with more hydrogen atoms, which is in contradiction to Markovnikov’s rule. Hence, it is an anti-Markovnikov addition.
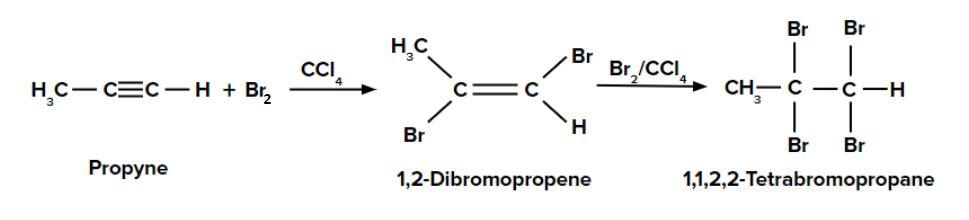
Addition of dihalogen to alkyne:
- Alkynes react with a molecule of X2 (in CCl4) to form 1,2-dihaloalkenes.
- Further, the reaction of one more molecule of X2 to 1,2-dihaloalkene produces tetra halogenated products.
Addition of Br2 (in CCl4) to propyne:
When the first molecule of Br2 in CCl4 is added to propyne, an anti-addition reaction happens since the two substituents are added on the opposite sides of the bond.
Test for unsaturation with Br2 in CCl4:
When bromine adds up to an unsaturated site, the reddish-orange colour of bromine solution in carbon tetrachloride is discharged. As a result, this reaction is used to test for unsaturation. The reddish-brown colour of Br2 in CCl4 is decolourised by alkenes and alkynes.
We have hexane, hexene, and hexyne in three test tubes. When Br2 / CCl4 is added to hexane, the reddish-brown colour of Br2 / CCl4 is retained. When Br2 / CCl4 is is added to hexene, the reddish-brown colour is discharged (decolourised) and 1,2-dibromohexane is formed. Similarly, when Br2 / CCl4 is added to hexyne, the reddish-brown colour is discharged (decolourised) and 1,1,2,2-tetrabromohexane is formed.


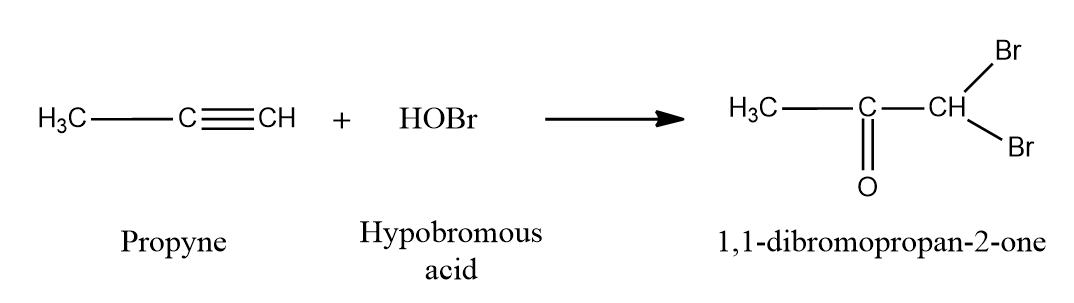
Addition of hypohalous acid to alkynes:
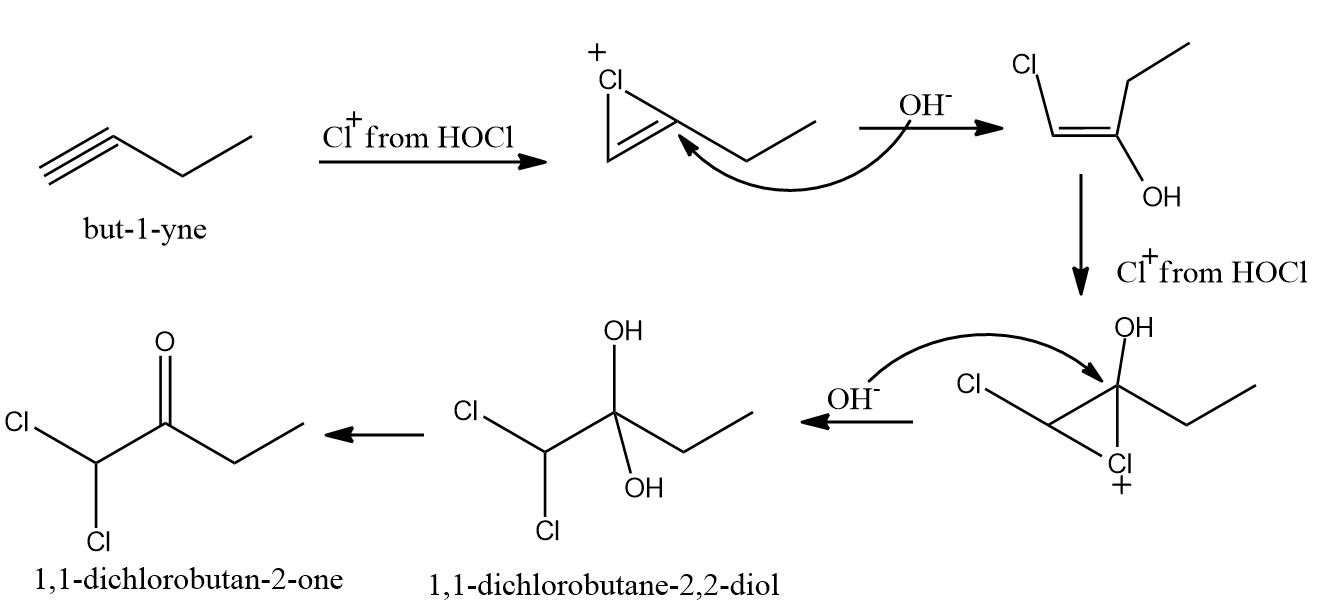
Except for fluorine, HOX breaks down into OH- and X+ because oxygen has a higher electronegativity than halogens. When two molecules of HOX (HOCl, HOBr, or HOI) are added to alkynes, geminal diols are formed. On the removal of H2O, these geminal diols form dihalo ketones (two halogen groups are present at the -position with respect to the carbonyl group). The geminal diols are extremely unstable. As a result, dehydration easily converts them to their respective ketones or aldehydes (loss of one water molecule).
In general, the -OH group is added to the carbon atom of an alkyne where positive charge is more stabilized.
 .
.
When HOX is added to the given alkyne, the electrons shift in such a way that the positive charge is stabilized by the alkyl group's +I effect or hyperconjugation. 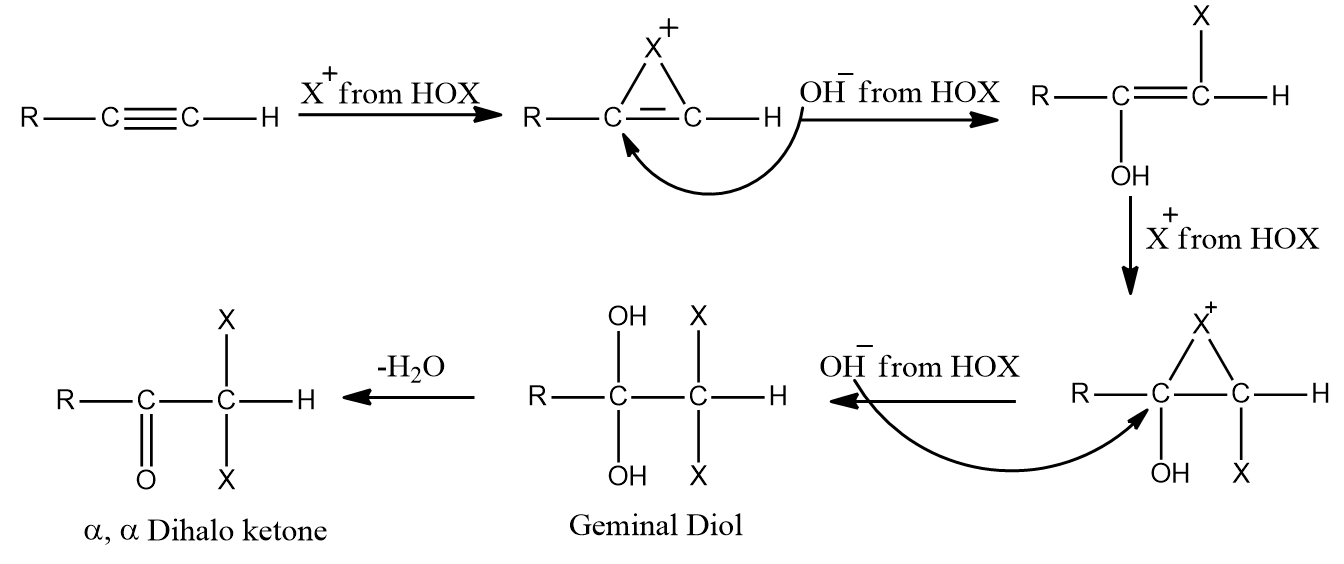
Addition of hydrogen halides to alkynes:
Geminal dihalides (two halogen atoms attached to the same carbon atom) are formed when two molecules of HX (HCl, HBr, or HI) are added to the alkynes.
Electrophilic addition of HX to the unsymmetrical alkynes occurs as
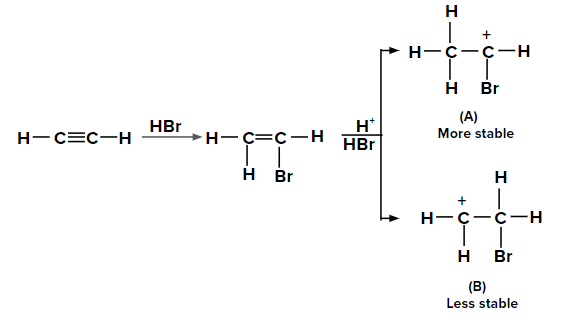
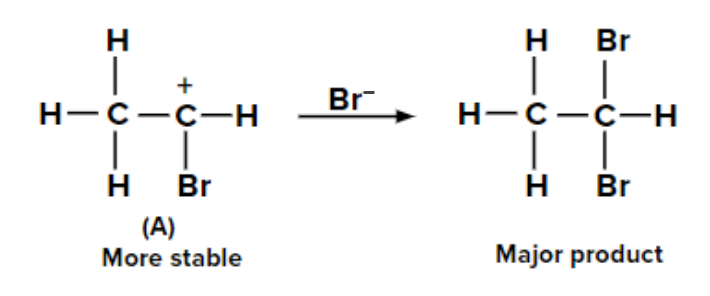
Two possible carbocations can be formed when the second molecule of HBr is added to bromoethene. Because of the +M effect of the -Br group and the hyperconjugation of three ⍺-hydrogen atoms, the carbocation labelled (A) is more stable than (B). As a result, carbocation produces the majority of the product (A).
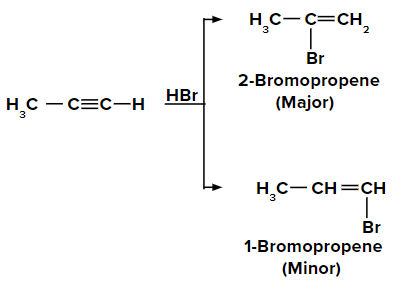
Adding HBr to propyne according to the Markovnikov rule:
On adding the first molecule of HBr to propyne, we obtain 2-bromopropene and 1-bromopropene as the major and minor products respectively, due to the Markovnikov rule.
When the second molecule of HBr is added to 2-bromopropene, we get the final product as 2,2-dibromopropane.
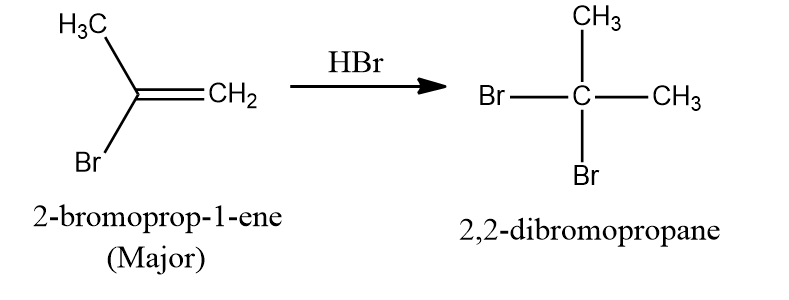
Practice Problems:
Q1. What would be the product formed in the given reaction?
- 1,1-dichlorobutane-2,2-diol
- 1,1-dichlorobutan-2-one
- 1,1-dichlorobutane
- Butane-2,2-diol
Answer: (B)
Solution: When HOCl is added to an alkyne, the electrons shift occurs in such a way that a positive charge is stabilised by the +I effect and hyperconjugation of the alkyl group. Geminal diols (1,1-dichlorobutane-2,2-diol) are highly unstable which undergo dehydration and convert to their respective ketones or aldehydes.
Hence, the correct answer is Option (B) 1,1-dichlorobutan-2-one.
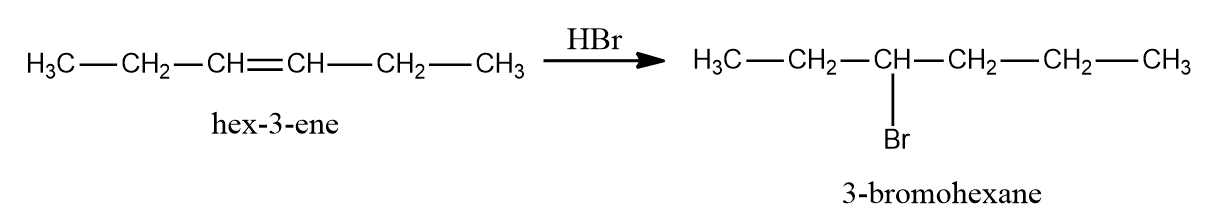
Q2. Which of the following given options give only product while reacting with halogen acid Hydrogen bromide?
- CH3-CH2-CH=CH-CH2-CH3
- CH3-CH2-CH2-CH=CH-CH3
- Both (a) and (b)
- CH2=CH-CH2-CH2-CH2-CH3
Answer: (A)
Solution: As CH3-CH2-CH=CH-CH2-CH3 (hex-3-ene) is a symmetrical alkene, only one product will be obtained on the addition of HBr, i.e., 3-Bromohexane.
As CH3-CH2-CH2-CH=CH-CH3 (hex-2-ene) is an unsymmetrical alkene, more than one product is formed. Thus, 2-Bromohexane and 3-Bromohexane will be formed as the products.
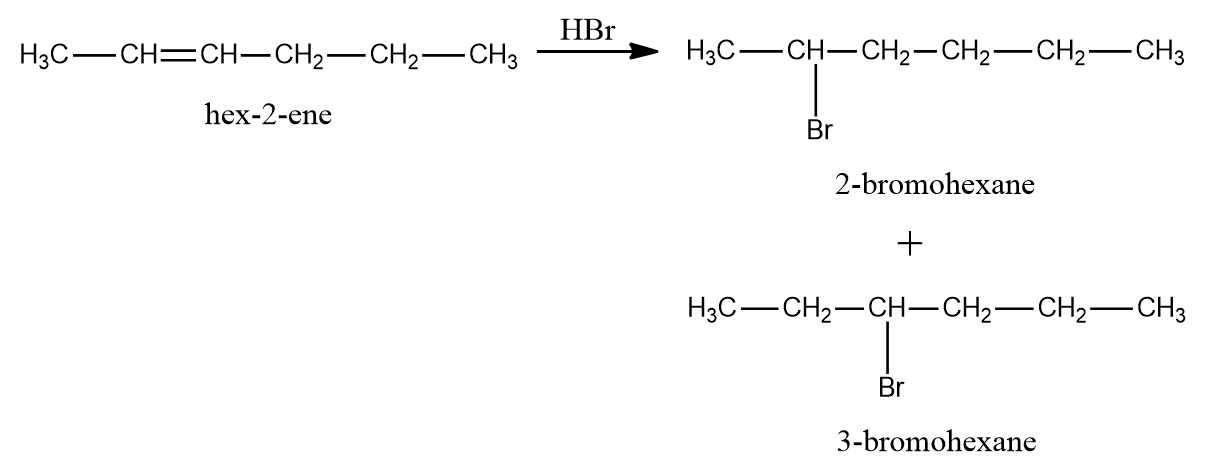
As CH2=CH-CH2-CH2-CH2-CH3 (hex-1-ene) is an unsymmetrical alkene, The principal product will be 2-Bromohexane, whereas the minor product will be 1-Bromohexane.
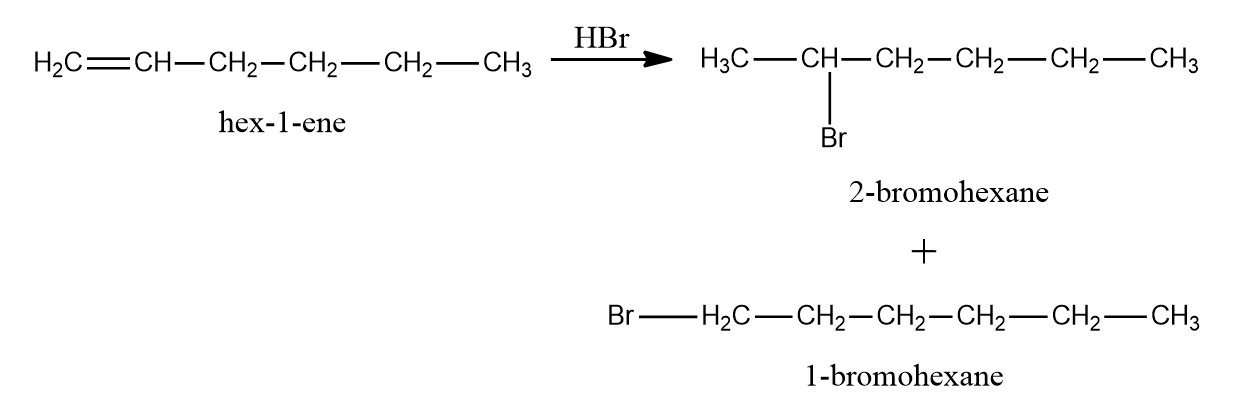
In the instance of hex-3-ene, just one bromo compound is generated as a result. As a result, choice (A) is the proper response.
Q3. When the given alkynes and halogen acid HBr reacts, which of the following products are formed?
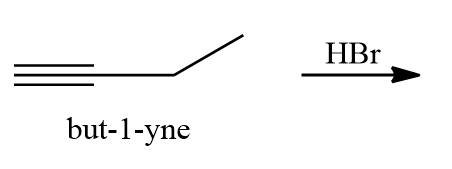
- 1,2-dibromobutane
- 1,1-dibromobutane
- 2,2-dibromobutane
- None of these
Answer: (C)
Solutions: Due to the Markovnikov rule, when the first molecule of HBr is added to Butyne, the major and minor products are 2-bromobutene and 1-bromobutene, respectively.

When the second molecule of HBr is added to 2-bromobutene, we get 2,2-dibromobutane as the end product.
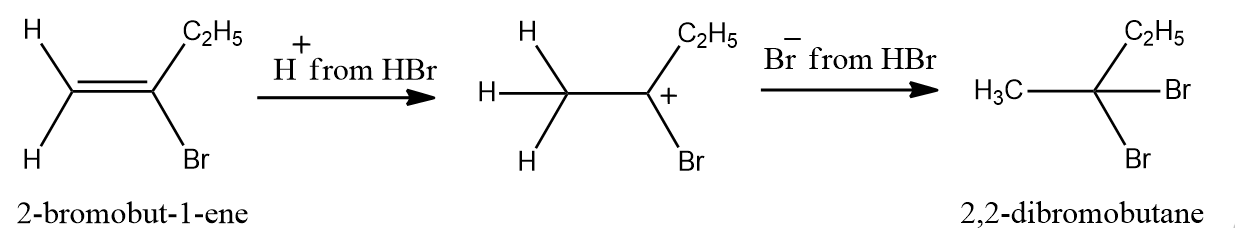
As a result, the correct answer is option (C).
Q4. What is the product of the following reaction?
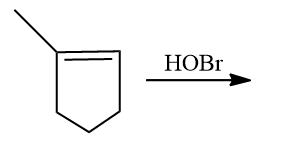
- 2-bromo-2-methylcyclopentanol
- 3-bromo-2-methylcyclopentan-2-ol
- 1-bromo-1-methylcyclopentanol
- 2-bromo-1-methylcyclopentanol
Answer: D)
Solution: HOBr, with the exception of fluorine, degrades into OH- and Br+ because oxygen has a higher electronegativity than halogens.
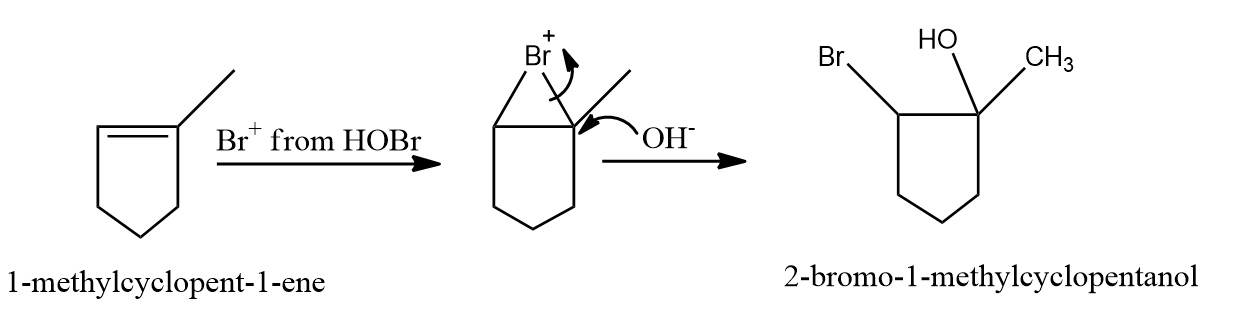
As a result, the correct option is (D).
Frequently Asked Questions-FAQs:
1. What type of reaction is very common for alkynes?
Answer: Alkenes and alkynes have very similar reactivity. They undergo electrophilic additions such as halogenation and hydrohalogenation. They can also be reduced with the help of a heterogeneous catalyst or oxidized using a variety of methods.
2. What applications does alkene have?
Answer: They are used in the production of plastics such as polythene for buckets, bowls, and bags and polystyrene for car battery cases and refrigerator parts. They are used in the production of ethane-1,2-diol, which is used as an anti-freeze agent in automobile radiators.
3. Is an alkyne polar or nonpolar?\
Answer: Alkynes are nonpolar, unsaturated hydrocarbons with physical properties similar to alkanes and alkenes. Alkynes dissolve in organic solvents, have little solubility in polar solvents, and are insoluble in water. In comparison to alkanes and alkenes, alkynes have significantly higher boiling points.
4. Do alkynes undergo substitution?
Answer: They do not prefer to substitute because it takes more energy to break existing bonds than it does to form new ones.


