-
Call Now
1800-102-2727
Actinoids-Discovery, General Characteristics, Uses, Practice Problems, FAQs
Have you noticed that there are two rows kept out of the periodic table at the bottom as you see it? You could wonder why these are not a part of the periodic table to be kept as two independent rows below the periodic table. Is it a drawback of the periodic table not being able to include it?
These two rows are f-block elements. The Lanthanides include the element lanthanum and a collection of elements with atomic numbers 57 to 71. The Actinoids are a set of elements with atomic numbers 89 to 103 that are arranged in a different row starting from the element actinium.
Actinoids are radioactive elements which radiate energy and have a very less half-life. They are used in nuclear reactors. Most nuclear fuels are made of fissile actinide elements, which can undergo and sustain nuclear fission.

We will learn more about the Actinoids series in this topic and talk about some of their key characteristics.
TABLE OF CONTENT
- Discovery of Actinoids
- General Characteristics of Actinoids
- Uses of Actinoids
- Practice Problems
- Frequently asked questions-FAQs
Discovery of Actinoides:
The first actinoids, to be discovered naturally were uranium and thorium, by Klaproth in 1789 and Berezelius in 1829, respectively. However, the majority of actinoids were discovered, as part of research in laboratories in the 20th century. As breakdown products of 253 and 238-uranium, actinium and protoactinium respectively can be found in trace levels in nature. Small amounts of plutonium are also naturally produced when uranium undergoes neutron capture. Monazite is the primary thorium mineral. It is a phosphate ore containing a sizable amount of lanthanide. The principal uranium ore, U3O8, is also known as pitchblende due to its appearance as masses of black, pitch-like substance. Other than uranium, all elements are artificial.
General Characteristics of Actinoids:
Actinides or actinoids are the names given to elements that are produced by sequentially filling up 5f orbitals with electrons. They are known by this name because, according to the periodic table, they are the next element after actinium (Ac). The actinide series, which contains 14 elements ranging from Th(90) to Lw(103), is also known as the second inner transition series (103). Despite not having any 5f electrons, actinium (Z=89) is examined within the f-block category and actinides are frequently employed to study it due to its similarities in properties to other elements.
Let's look at some important tendencies of actinoids in the periodic table:
a) Electronic Configuration of Actinoids:
All actinoids are assumed to have the same electronic configuration of 7s2, with varying occupancy of the 5f and 6d subshells. Though not in thorium (Z = 90), the fourteen electrons are formally introduced to the 5f orbitals from protactinium (Pa) onwards at element 103.
The anomalies in the electronic configurations of actinoids, as they were in lanthanoids, are related to the stabilities of the f0, f7, and f14occupancies of the 5f orbitals. [Rn]5f77s2 and [Rn]5f76d17s2 are the configurations for Am and Cm, respectively. The angular component of the wave function of the 5f orbitals is similar to that of the 4f orbitals, but because they are less buried, the 5f electrons have a much higher bonding potential.
Electronic configurations of actinoids are mentioned below:
|
Atomic Number |
Element |
Symbol |
Configuration |
|
89 |
Actinium |
Ac |
[Rn]6d17s2 |
|
90 |
Thorium |
Th |
[Rn]6d27s2 |
|
91 |
Protactinium |
Pa |
[Rn]5f26d17s2 |
|
92 |
Uranium |
U |
[Rn]5f36d17s2 |
|
93 |
Neptunium |
Np |
[Rn]5f46d17s2 |
|
94 |
Plutonium |
Pu |
[Rn]5f67s2 |
|
95 |
Americium |
Am |
[Rn]5f77s2 |
|
96 |
Curium |
Cm |
[Rn]5f76d17s2 |
|
97 |
Berkelium |
Bk |
[Rn]5f97s2 |
|
98 |
Californium |
Cf |
[Rn]5f107s2 |
|
99 |
Einsteinium |
Es |
[Rn]5f117s2 |
|
100 |
Fermium |
Fm |
[Rn]5f127s2 |
|
101 |
Mendelevium |
Md |
[Rn]5f137s2 |
|
102 |
Nobelium |
No |
[Rn]5f147s2 |
|
103 |
Lawrencium |
Lr |
[Rn]5f146d17s2 |
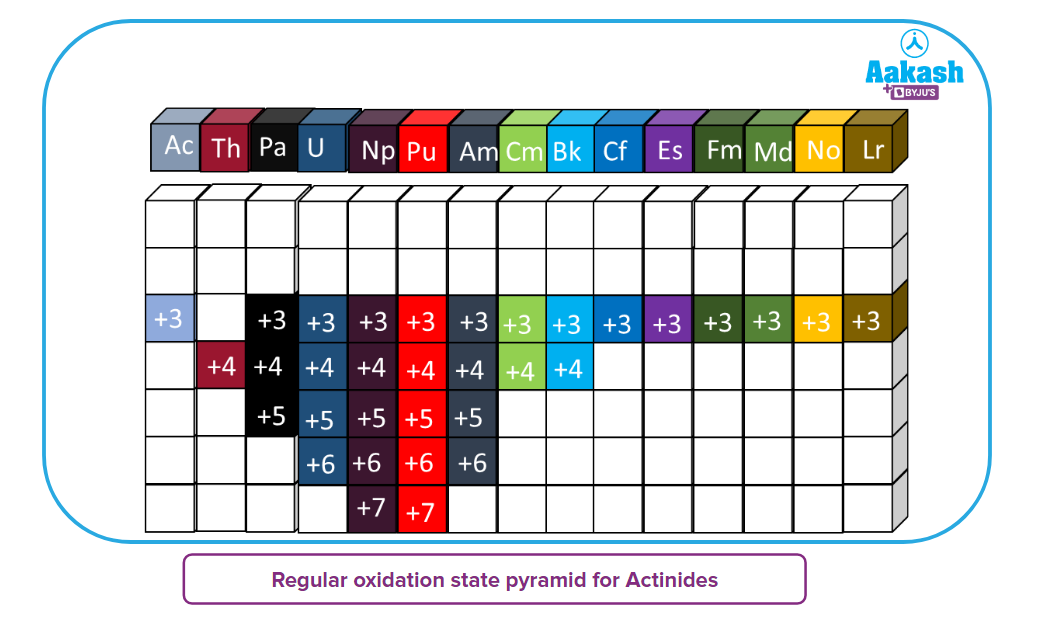
b) Oxidation states of Actinoids:
- The most prevalent oxidation state for actinides, like lanthanides, is +3. This condition isn't usually the most stable, though, in contrast to the first four constituents ( Th, Pa, U, Np).
- U3+ is readily oxidised, for instance, in solution in the air. But, the most stable state for the later elements AmLr, is the +3 state (except No).
- The most stable oxidation states for the first four elements are Th(+4), Pa(+5), and U(+6) In these high oxidation states, all of the outer electrons, including the f- electrons, are utilised for bonding.
- Neptunium-Np exhibits a maximum oxidation state of +7, however, it is oxidizing in nature, and its most stable state is +5.
- Plutonium-Pu shows all oxidation states from +3 to +7, with +4 being the most stable.
- Amercium-Am represents the +2 to+6 range of oxidation states. The Am2+ is set up in the f7configuration. However, it only exists as fluoride in solid form and is the equivalent of Eu2+. The most stable state, however, for Am and essentially all of the other elements is +3.
- From, thorium to berkelium ( Th to Bk), all elements exist in the +4 oxidation state. Cf2+, Es2+, Fm2+, Md2+ and No2+ exist as ions in solutions.
c) Physical Properties of Actinoids:
- All actinides appear like silvery metals.
- Although the melting points of transition elements are higher, Actinoids are somehow lower in their melting points.
- The size of the ions steadily decreases along the series because the increased charge on the nucleus is insufficiently veiled by the f electrons.
- An "actinide contraction" is produced as a result, similar to the lanthanide contraction. High densities are found in actinides.
- Actinoids are highly reactive metals, especially when finely separated.
- When boiling water is applied to them, for example, a mixture of oxide and hydride is formed.
The majority of non-metals combine with them at moderate temperatures.
- Below is a table listing some of the properties of actinides up to berkelium. Heavy actinides are a subject about which little is known.
|
Element |
Melting Point(℃) |
Density (g cm-3) |
Radius M3+ (ppm) |
Radius M4+ (ppm) |
|
Thorium |
1750 |
11.8 |
108 |
94 |
|
Protactinium |
1552 |
15.4 |
104 |
90 |
|
Uranium |
1130 |
19.1 |
102.5 |
89 |
|
Neptunium |
640 |
20.5 |
101 |
87 |
|
Plutonium |
640 |
19.9 |
100 |
86 |
|
Americium |
1170 |
13.7 |
97.5 |
85 |
|
Curium |
1340 |
13.5 |
97 |
85 |
|
Berkelium |
986 |
14.8 |
96 |
83 |
d) Colour of the Ions :
- The number of electrons in 5f-orbitals has an impact on the ions' colour. The ions with zero 5f-orbital electrons (i.e., 5f0) or seven 5f-orbital electrons (i.e., 5f7 ) are colourless.
- Ions with 2 to 6 electrons in 5f-orbitals show colour in both crystalline structures and aqueous solutions. What imparts colour is the f-f transition.
- The coloured ions in different charges are shown in the below table:
|
Ions |
Inner configuration |
colour |
|
Th4+ |
5f0 |
Colourless |
|
U3+ |
5f3 |
Red |
|
Np3+ |
5f4 |
Purple |
|
Pu3+ |
5f5 |
Vilot |
|
Am3+ |
5f6 |
Pink |
|
Cm3+ |
5f7 |
Colourless |
|
U4+ |
5f2 |
Green |
|
Np4+ |
5f3 |
Yellow green |
e) Actinoid contraction:
The atomic sizes of the tri-positive actinides are observed to decrease continuously from Thorium (Th) to Lawrencium (Lr). The causes of this are the growing nuclear charge and the electrons entering the inner orbital (n-2)f. Actinide contraction is the term used to describe the element's shrinkage brought on by a rise in atomic number.
f) Magnetic behaviour:
- The majority of the actinide ions are paramagnetic in nature because they have unpaired electrons present in their shell.These ions like etc. are showing paramagnetic character.
- Actinide cations that only have paired electrons are diamagnetic in nature.A few examples .
g) Formation of Complexes:
- Actinides are marginally more likely to produce complex compounds than lanthanides. Their smaller ions and higher charge are the cause for this.
- The majority of the actinide halides form complex combinations when they interact with alkali metal halides.
- Actinides combine with organic substances like oxime and EDTA to generate chelates.
- For the ions M4+, MO22+, M3+and, MO2+ the degree of complex formation declines in the order.M4+>MO22+>M3+>MO2+
h) Radioactivity:
- The radioactivity in all actinide elements is inherent.
- Actinides are heavy elements that contribute to the radioactivity of spent fuel in addition to the fission products present in nuclear reactor cores. Because many actinides are radioactive and/or unstable, they need to be handled carefully. The chemistry and arrangement of the particles in crystals are significantly influenced by the radiation in actinides.
- They are able to undergo nuclear reactions.
- The most prevalent isotope of the plutonium actinide, which is by far the most frequently generated, is plutonium (239). 'Minor' actinides like neptunium (238) and americium (241) and (243), as well as curium 244 and 245, are also produced in lower amounts by nuclear reactors.
Uses of Actinoids:
Some major uses of a few actinides are discussed below:
- Uses of thorium:
- When burnt in a gas flame, thorium dioxide with 1% CeO2 generates a dazzling white light.
- It is utilised to create incandescent gas mantles as a result.
- A mixture of 99 % thorium nitrate and 1% cerium nitrate is used to treat the mantle made of silk fibre. A network of thoria (ThO2) and ceria (CeO2) is left behind after this mantle is mounted within the lamp and ignited.
- Th-232 makes up almost all naturally occurring thorium. This isotope is transformed into the fissionable U-233, which is not itself fissionable.
![]()
In order to create the fissionable material required for atomic reactors, thorium is employed.
- Uses of Actinium:
- For usage in targeted therapy (TAT), a treatment method that uses particle emissions to eradicate malignancies, certain prostate, brain, and neuroendocrine tumours are treated using tailored alpha therapy. The radionuclide actinium-225 has nuclear properties that are very promising.
- Uses of Uranium:
- The glass industry uses uranium salts to give the glass a green tint and is also used in the textile, ceramic, and pharmaceutical industries.
- Atomic reactors and bombs both utilise the U-235 isotope as nuclear fuel.
- Uses of plutonium:
- Nuclear fuel made of Pu-239 is utilised in nuclear reactors. It comes from the U-238.
Related links:
D and F block (1:06:38 to 1:18:25)
D and F Block Elements Class 12 Chemistry One Shot & Mind Maps L- 2 (Ep 29) | NEET 2022 Exam Prep
Practice Problems:
Q1. Which actinide oxidation state is the most stable?
A. +4
B. +2
C. +3
D. +5
Answer: C)
Solution: Because of the lesser energy difference between the 5f,6d and 7s orbitals, actinoids exhibit varying oxidation states. Other oxidation states are possible despite the fact that +3 is the most stable oxidation state for the actinides due to the efficient shielding of f-electrons.
Q2. Which among of given below is diamagnetic actinoid in nature?
A. Lr3+
B. Th4+
C. Am5+
D. Pa4+
Answer: A
Solution: Diamagnetic materials are those that are only faintly attracted to a magnetic field. Due to the existence of unpaired electrons, all actinoids exhibit paramagnetic behaviour. This indicates that a magnetic field only slightly attracts them. Lr3+ has all paired electrons
Q3. What type of plutonium is contained in nuclear weapons?
A. Pu-238
B. Pu-239
C. Pu-240
D.Pu-241
Answer: B)
Solution: The most frequent form of plutonium in a normal nuclear reactor is fissile Pu-239, which is created when a neutron is captured from U-238 and then undergoes beta decay. Fissioning Pu-239 produces roughly the same amount of energy as fissioning U-235.
Q4.Which trans uranium actinoid element was the first to be identified?
A.Protactinium
B.Americium
C.Actiniu
D.Neptunium
Answer: D
Solution: The first transuranium actinoid element to be identified as neptunium, was done in 1940. It is a silvery metal having to melt and boiling points of 637 ℃ and 4174℃ respectively. It has compounds with oxidation levels ranging from +3 to +7, making it suitable for chemical studies.
Frequently Asked Questions- FAQ
Q1. What makes actinoids special?
Answer: Since every actinoid isotope undergoes radioactive decay, the actinoids are remarkable in that they consist of a series of 15 elements with no stable isotopes. As a result, only a handful of the lighter, more stable members of the series (such as thorium and uranium) are found in nature.
Q2. Why are actinoids known for their instability?
Answer: Actinium is typically included in the series due to some chemical similarities. All actinides are radioactive because their big nuclei make them unstable and cause them to spontaneously fission, which releases a lot of energy.
Q3. Where can one find actinoids?
Answer: Thorium and uranium are the only actinides that are found in significant quantities in the earth's crust, however, uranium ores have occasionally contained trace amounts of neptunium and plutonium. Actinium and protactinium can be found in nature as decay byproducts of some thorium and uranium isotopes.
Q4. What procedures are used to prepare actinoids?
Answer: Vacuum evaporation, vacuum reduction-distillation, arc melting, levitation melting, rolling, electroplating, and loading precisely measured amounts of actinide materials into finely machined capsules are some of the methods utilised to manufacture such targeted actinides.


