-
Call Now
1800-102-2727
Surface Energy - Its relation with Surface Tension, Practice Problems, FAQs
There is an interesting property related to liquids which can be realised through countless day to day examples. Like raindrops are spherical, a steel needle when placed gently on a water surface may rest on it, though steel is denser than water, oil rises up a cotton wick in spite of gravity, etc. All these occur due to the phenomenon of surface tension related to liquids. It can be understood with the help of surface energy. So in this article let’s study surface energy and also see how it is related to surface tension.
Table of Contents
- Surface Energy
- Work done by Surface Tension
- Relation between Surface Tension and Surface Energy
- Practice Problems
- FAQs
Surface Energy
The molecules of fluid stay together due to attraction between them. For a molecule well inside the liquid, it is attached in all directions by the other molecules surrounding it. This makes such molecules have negative potential energy.
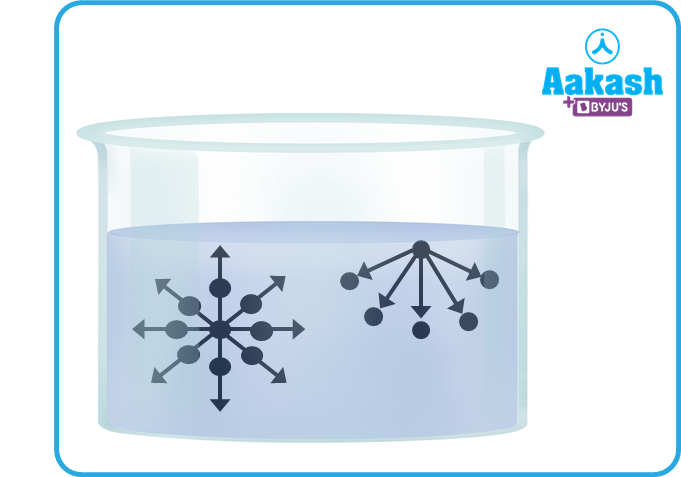
For a molecule which is present on the surface of the liquid, only its lower half side is surrounded by liquid molecules. It also has a negative potential energy due to attraction of liquid molecules in its lower half. But this is less negative as compared to the potential energy of a molecule fully inside. It is approximately half of the latter. It is half the energy required to remove a molecule completely away from the liquid.
This is because the energy required to remove the molecule completely from a liquid is equal to its potential energy. This energy is known as its heat of evaporation. For molecules on the surface, only half of this energy is required to make it free. Thus we can say that the molecules at the surface have extra energy as compared to the molecules in the interior. We call this energy the surface energy.
More the surface area of the liquid surface, more is the number of molecules present on the surface. Thus, more is the extra energy possessed by the surface molecules. Thus, the surface of a liquid is always under a tension so as to have a minimum surface area and hence the minimum surface energy, so as to attain maximum stability.
Surface energy is the potential energy of surface molecules per unit area of the surface.
Note:
CGS unit of surface energy is and SI unit is Joule m-2.
The dimensional formula is .
Surface energy depends on the number of surfaces.
Work done by Surface Tension
Consider a system consisting of a U-shaped wire and a slider of length l, which can move frictionlessly on the U-frame. Immerse the system in a soap solution and a soap film will be formed within the system as shown in the figure.
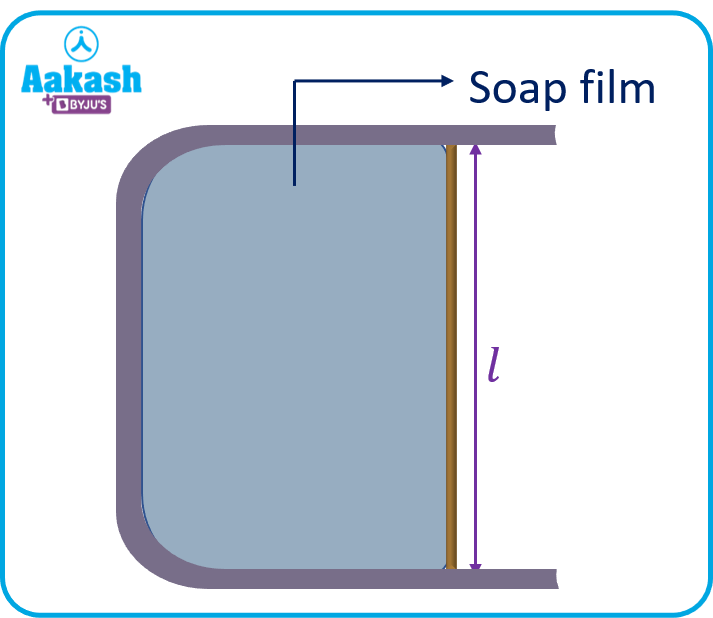
We know that surface tension is defined as force per unit length. Here, the force on the slider due to the surface tension of the soap film is equal to 2Sl towards the left. (Factor 2 is arising because of the two faces of the soap film.) As we start moving the slider away, quasi-statically (slowly slowly), the molecules between two surfaces shift to increase the surface area. So, work is required. This work is stored on the surface in the form of surface potential energy.
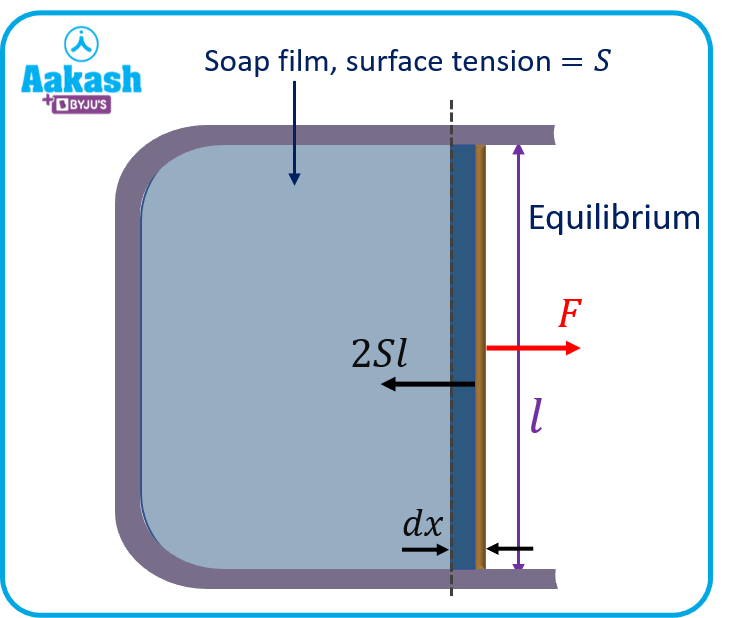
Let a force F be applied quasi-statically on the slider to move it through distance dx.
The work done by the external force is,
Surface tension is the work done by an external agent that is trying to increase the area of the film per unit area. Mathematically,
Relation between Surface Tension and Surface Energy
According to the work-energy theorem, the sum of work done by all forces is equal to the change in kinetic energy. Let Wext be the work done by an external force to stretch a soap film. If the applied force is quasi-static, then the change in kinetic energy of the soap film will be zero.
because of quasi-static process
Here U is the change in internal energy of film. Hence the work done by external agents increases the internal energy of soap film. This change in internal energy is known as surface energy.
Surface tension is numerically equal to increase in surface energy per unit increase in surface area.
Examples:
- Liquid drops acquire spherical shape in space. This is because for a particular volume the spherical surface has the least surface area. As the surface area is less, liquid drop will have low potential energy. We know that because of stability, particles try to lower their potential energy.
- When a bigger spherical drop of a liquid is broken to form smaller spherical droplets, the total surface area of the system would increase. Due to this, the energy of the system due to surface tension will increase. This increase in energy is taken from the system and therefore the temperature of the system will fall. That’s why the air is colder near the fountain.
Practice Problems
Q1. How much work will be done in increasing the radius of a soap bubble from r1 to r2? The surface tension of the soap solution is S.
Answer. The change in surface area when radius of bubble increases from r1 to r2 is,
as soap bubble is having two surfaces,
We know that,
Hence, the work done in increasing the radius of the soap bubble from r1 to r2 is given by,
Q2. A water drop of radius R is broken into 1000 equal droplets. Calculate the gain in surface energy. The surface tension of water is S.
Answer. Consider that a big water drop of radius R is broken into 1000 equal droplets of radius r, where r < R.
The mass of water before and after breaking the drop is conserved.
Thus, we can write,
Here, is the density of water.
The surface area of the big water drop is,
The total surface area of the droplets is,
The increase in surface area is,
Hence, the required work done is,
Q3. Between two straight parallel wires, each 10 cm long and separated by 0.5 cm, a layer of water is formed. Calculate the work required to increase 1 mm distance between the wires. Surface tension of the water is .
Answer. Initial surface area = 2 length separation
The required work,
Q4. Calculate the effort required to blow a soap bubble from a radius of 10 cm to 20 cm against surface tension, if the surface tension of soap bubble solution is .
Answer. Original total surface area
Factor of 2 is multiplied as the bubble has two surfaces.
Final total surface area
Therefore, extension in area
Now work done,
FAQs
Q1. Does surface energy depend upon the temperature?
Answer. Yes, the surface energy depends on the net inward cohesive force and so surface energy decreases with increasing temperature and so does surface tension.
Q2. Does adding salt in water increase surface tension?
Answer. The surface tension of water increases when salt is added to it. Although the strong interactions between sodium cations and partial negative oxygen, and chloride anions and partial positive hydrogens disrupt some hydrogen bonding between water molecules, they actually strengthen the surface tension of water.
Q3. Does the potential energy of molecules on the surface of liquid and inside it have the same potential energy?
Answer. No, molecules which are present on the surface of liquid have half of the potential energy as compared to the molecules which are inside the liquid.
Q4. How surface energy and surface tension relate to each other?
Answer. Surface tension is numerically equal to surface energy per unit surface area.


