-
Call Now
1800-102-2727
Davisson Germer Experiment, experimental apparatus, working observations, FAQs
Now we know that matter has dual characteristics of waves and particles as per the de-Broglie hypothesis. But it was just a theoretical hypothesis until Davisson and Germer performed a famous experiment that proved the hypothesis and Davisson received the Nobel prize for his work in 1937.
Table of contents
- Davisson Germer Experiment
- Experimental apparatus
- Working
- Bragg's law
- Observations
- G.P. Thomson’s Experiment
- Heisenberg’s Uncertainty Principle
- FAQs
Davisson Germer Experiment
Clinton J. Davisson and Lester H. Germer performed an experiment to prove the wave nature of electrons by studying the diffraction of an electron beam being scattered by the crystals of a nickel. The experiment also proved the early theoretical de-Broglie hypothesis that matter also possesses wave nature apart from the particle nature.
Experimental apparatus
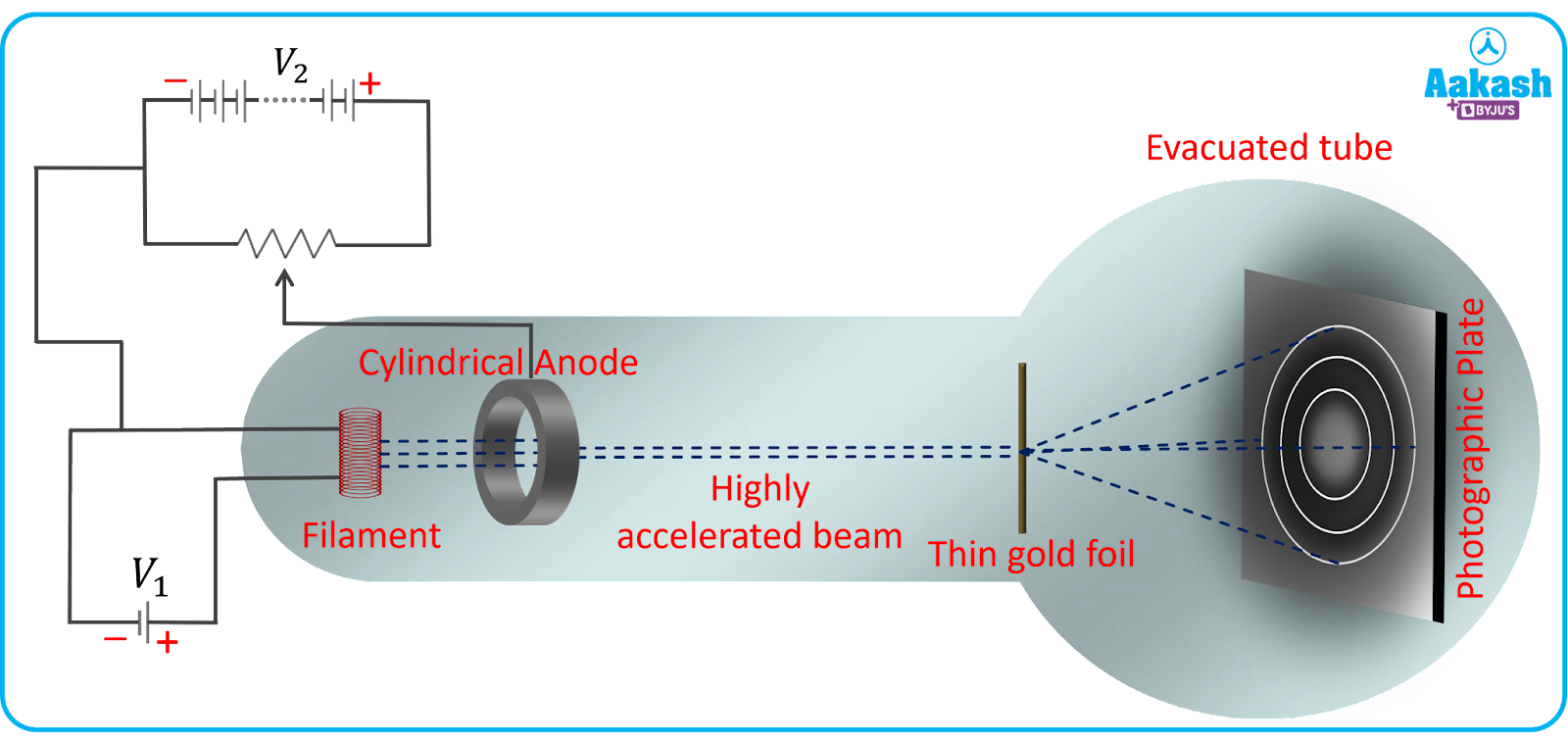
The arrangement used by Clinton J. Davisson and Lester H. Germer to prove the wave nature of electrons is shown in the image below. The arrangement consists of an electron gun having a tungsten filament which is heated using a Low tension voltage source. The gun consists of an accelerator which is responsible for accelerating the thermally excited electrons from the filament by creating the required potential difference using a high tension voltage source. Thus the velocity of the electrons can be varied using the accelerator. The hole in the accelerator ensures a fine beam of electrons is incident on the nickel crystal placed in its path. The apparatus is placed in a vacuum chamber for avoiding any collisions of electrons with atoms in the way of an electron beam. As it can be seen from the image that a movable collector with a galvanometer that can be rotated is placed in the path of the diffracted electron beams. The collector is basically a Faraday cup electron detector to measure the number of electrons being diffracted after striking the nickel target at different angles since it is movable.
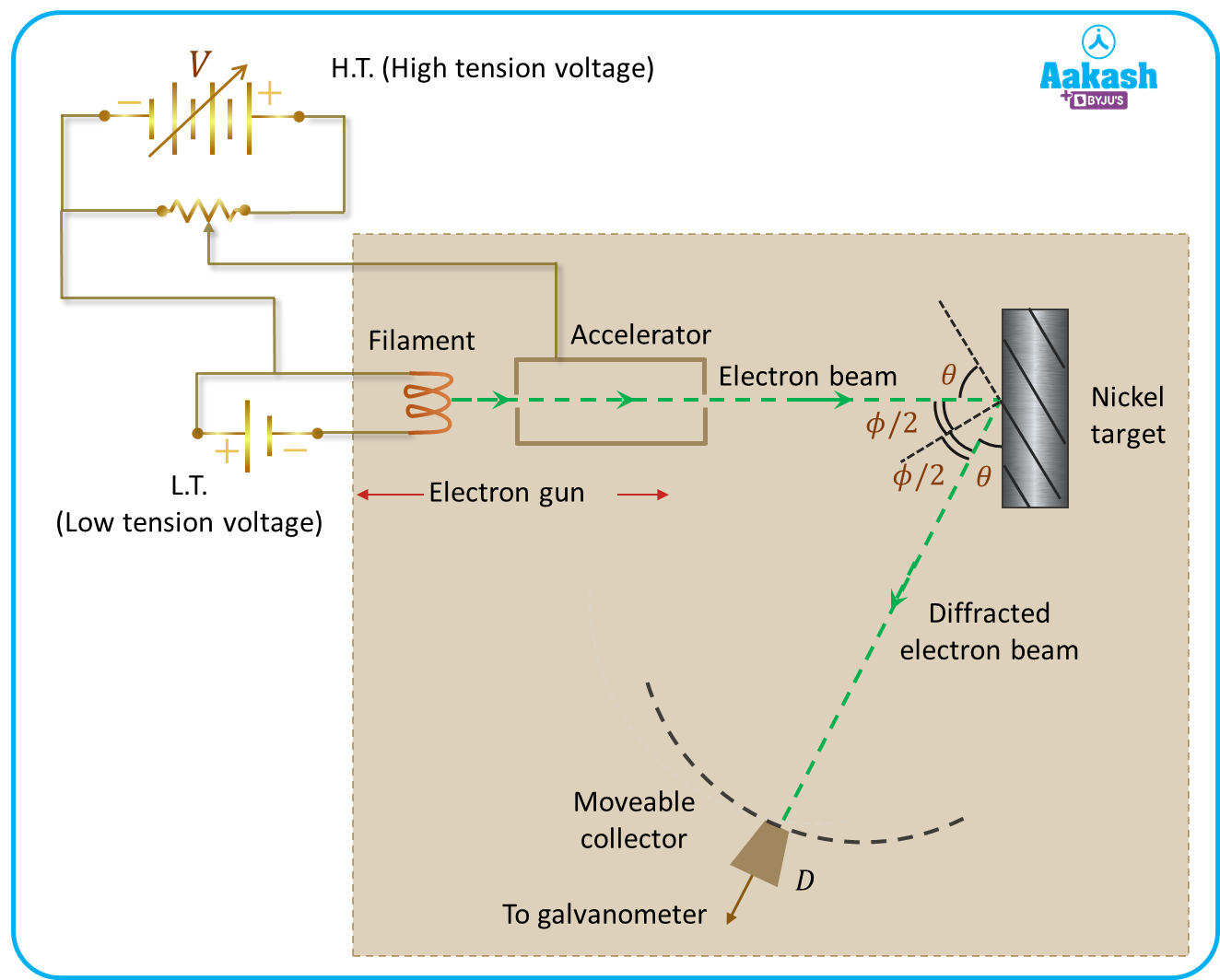
Working
When the filament is heated, thermionic emission of electrons takes place; the electrons are then accelerated using an accelerator which produces an electron beam. This electron beam is then incident normally on a nickel target. The incident electrons are scattered in different directions by the atoms of the nickel target. The intensity of the electron beam, scattered in a given direction, is found by rotating the movable collector in a circle. The movable collector is attached to a galvanometer which gives the reading of the photocurrent and hence the intensity of the electron beam can be found.
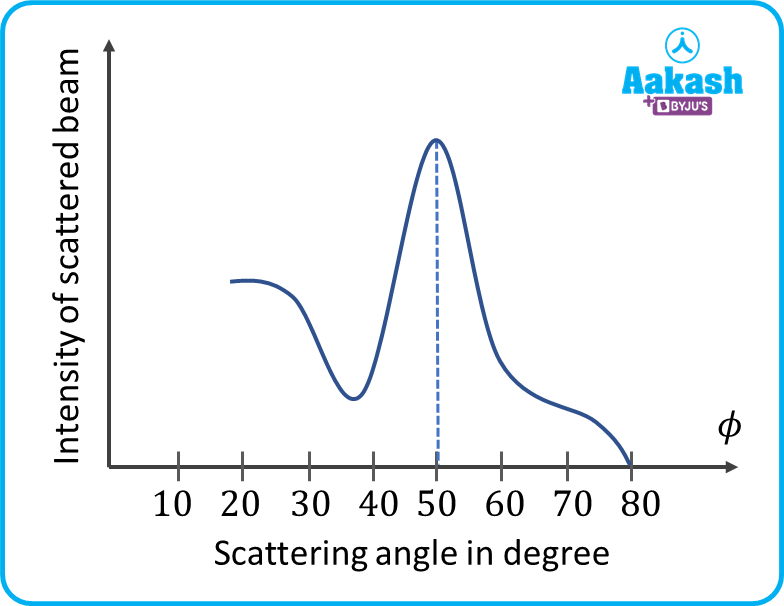
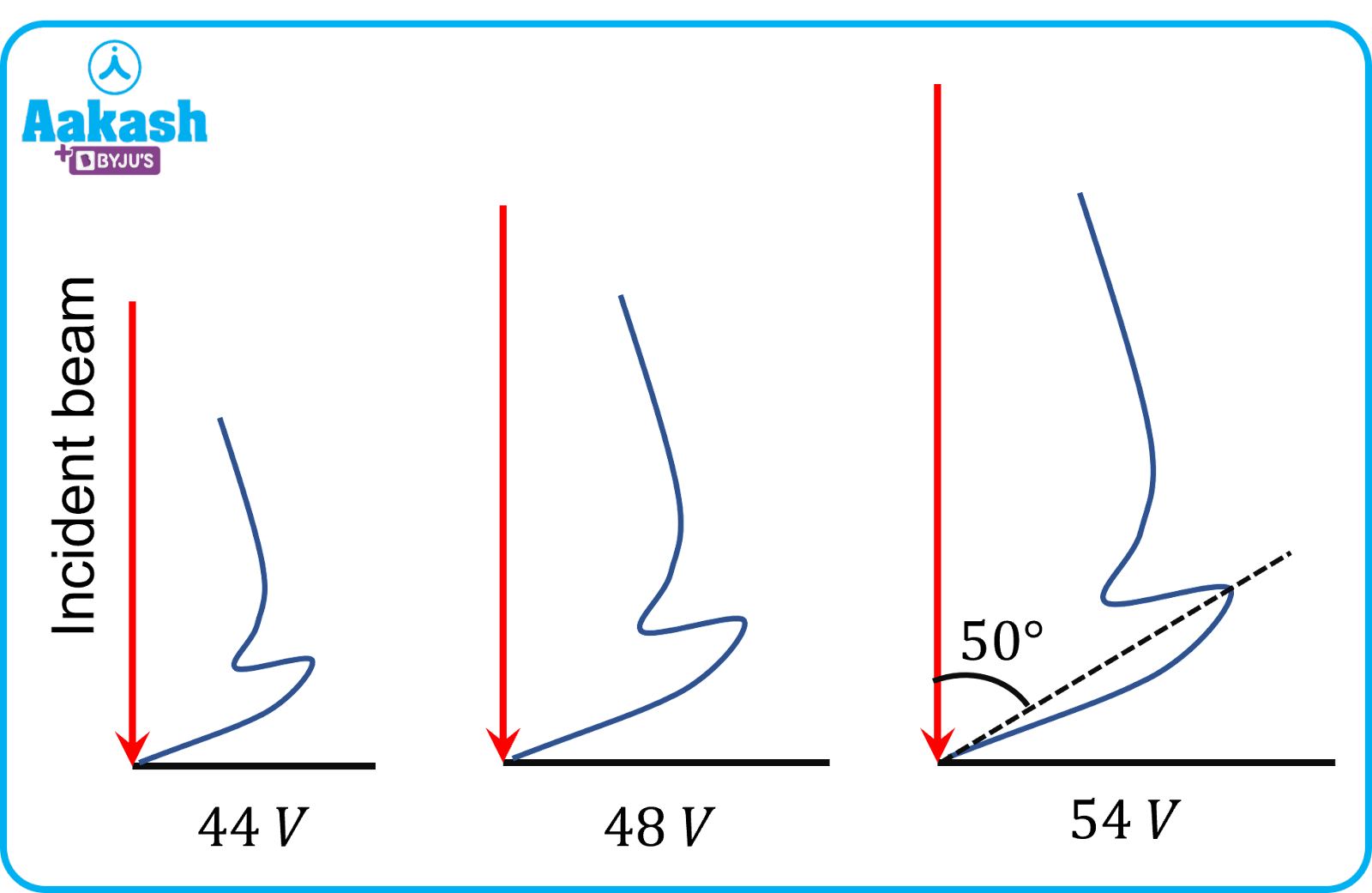
Graph shows the variation of intensity of the scattered electrons with the angle for the accelerating voltage of 54 V. The scattered wave shows maximum intensity at an angle of 50° to the incident electron beam. The maximum intensity occurs due to constructive interference electrons being diffracted from the nickel target atomic layers.
Diffraction
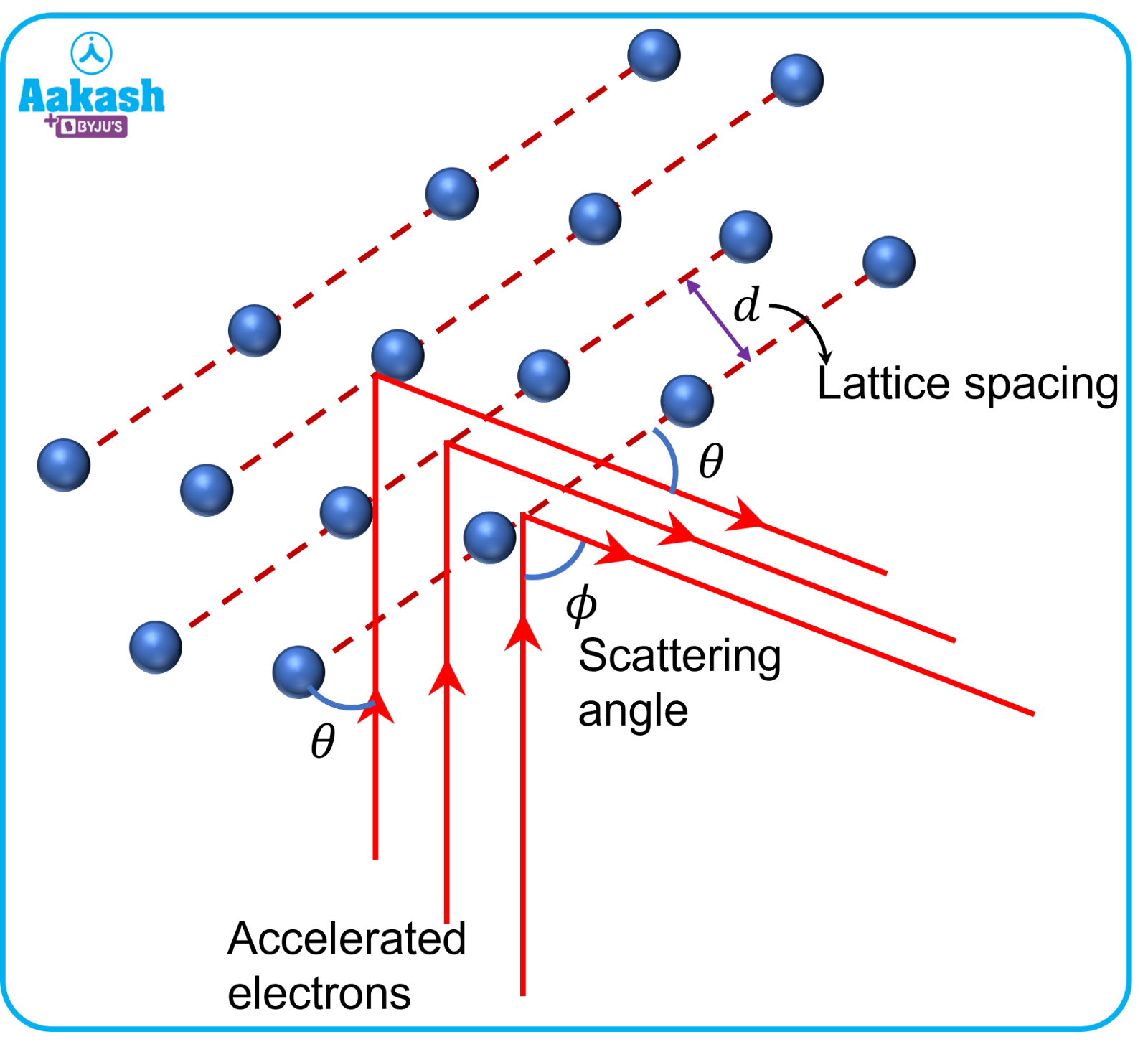
When a stream of fast particles impinges on the atoms of a crystal, their paths are bent into a regular pattern, which can be recorded by directing the diffracted beam onto a photographic film. In the Davisson Germer experiment, the electrons get scattered from a nickel crystalline surface and thus matter waves produce diffraction patterns. Since the nickel target used has a regular crystalline surface, the scattered electron beam exhibits a clear diffraction pattern and a clear maximum at a specific angle. The diffraction pattern was similar to that obtained in Bragg’s X-ray diffraction pattern.
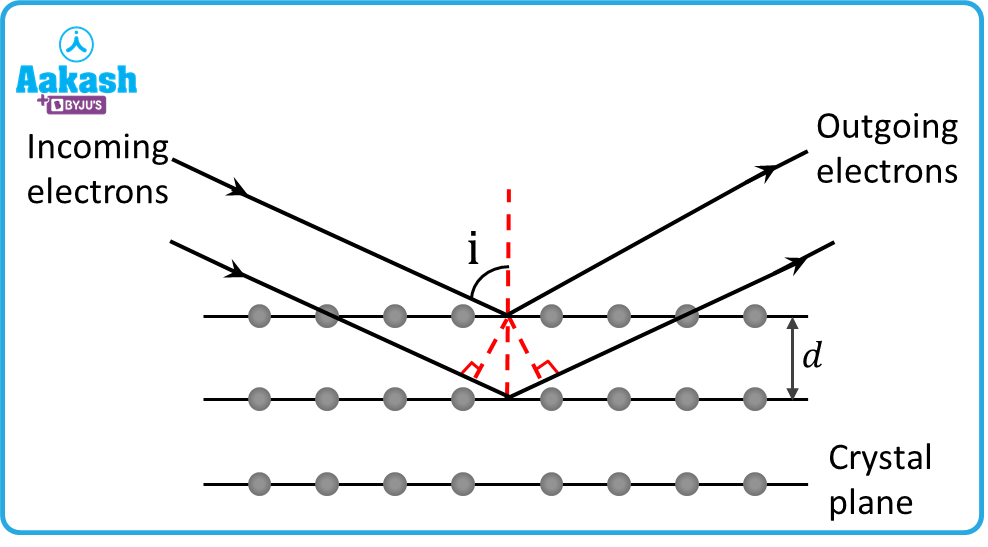
Bragg's law
When the crystal lattice scatters the incident rays, maxima are observed corresponding to the following conditions:
- Angle of scattering is equal to the angle of incidence.
- Path difference =n where n is an integer.
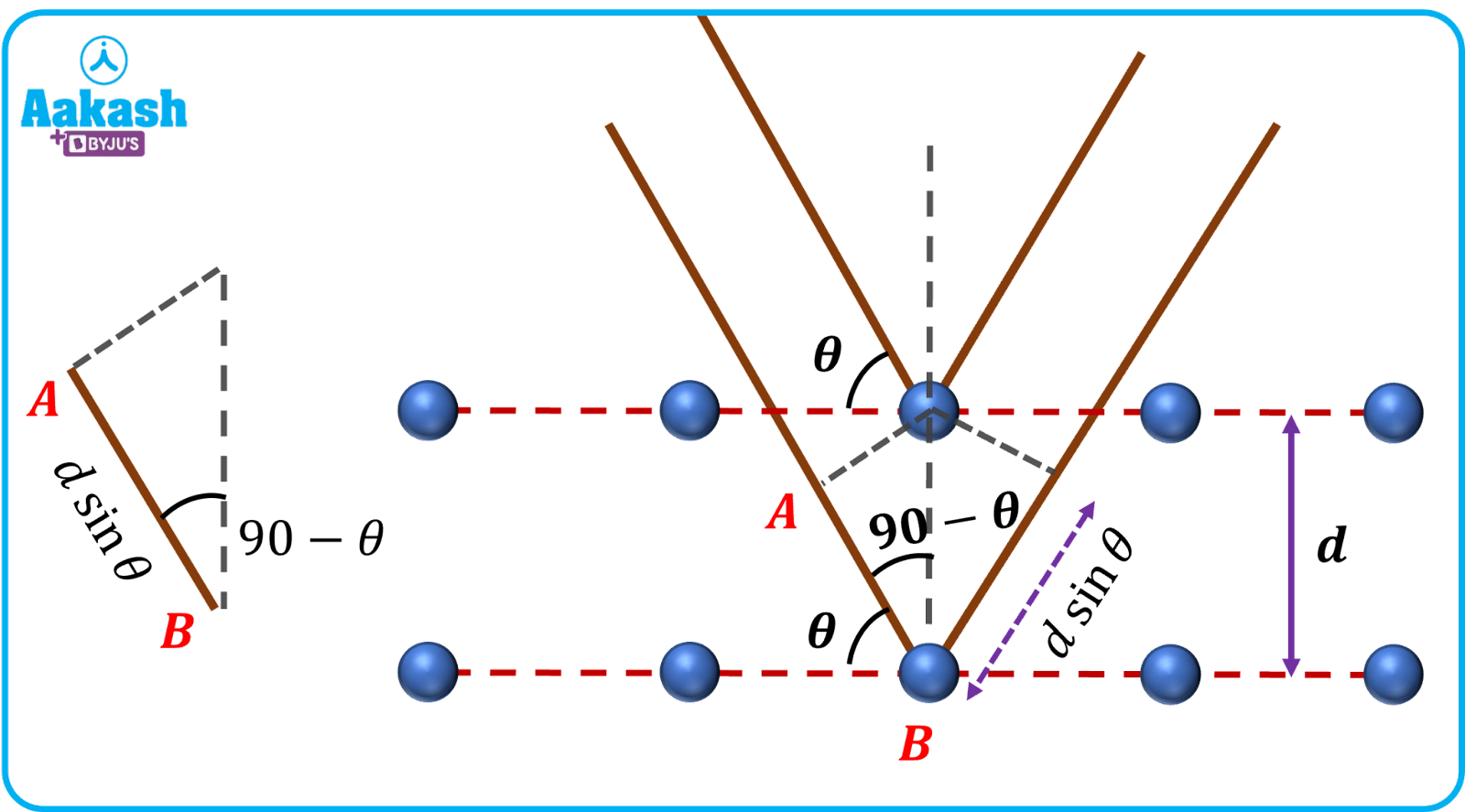
Extra path covered by second ray = AB+BC
From the figure, AB=d(90-)=d
Where d= interplanar spacing
= Angle between incident ray and crystals reflecting plane
Therefore the path difference between two rays, =AB+BC=d+d=2d
For constructive interference, 2d=n
For nickel, d=0.091 nm
For 1st order maxima , n=1
The experimental results shows that at an accelerating potential of about 54 V, the intensity maximum is registered at the scattering angle of about =50o.
The peak was found at an scattering angle of =50o
++=180
2=180-
2=180-50=130
=65o
For an angle of 65o between the incident ray and the reflecting plane of crystal,
20.09165o=1
=0.165 nm=1.65Å
Considering the electron to be associated with wave, the above value gives the experimental value of wavelength of the wave.
Observations
- The intensity (𝐼) of the scattered electrons variation was obtained by changing the angle of scattering (𝜃).
- The experiment was performed by varying the accelerating voltage from 44 𝑉 to 68 𝑉. The variation between the intensity of electrons detected at a particular voltage is shown in the graph below.
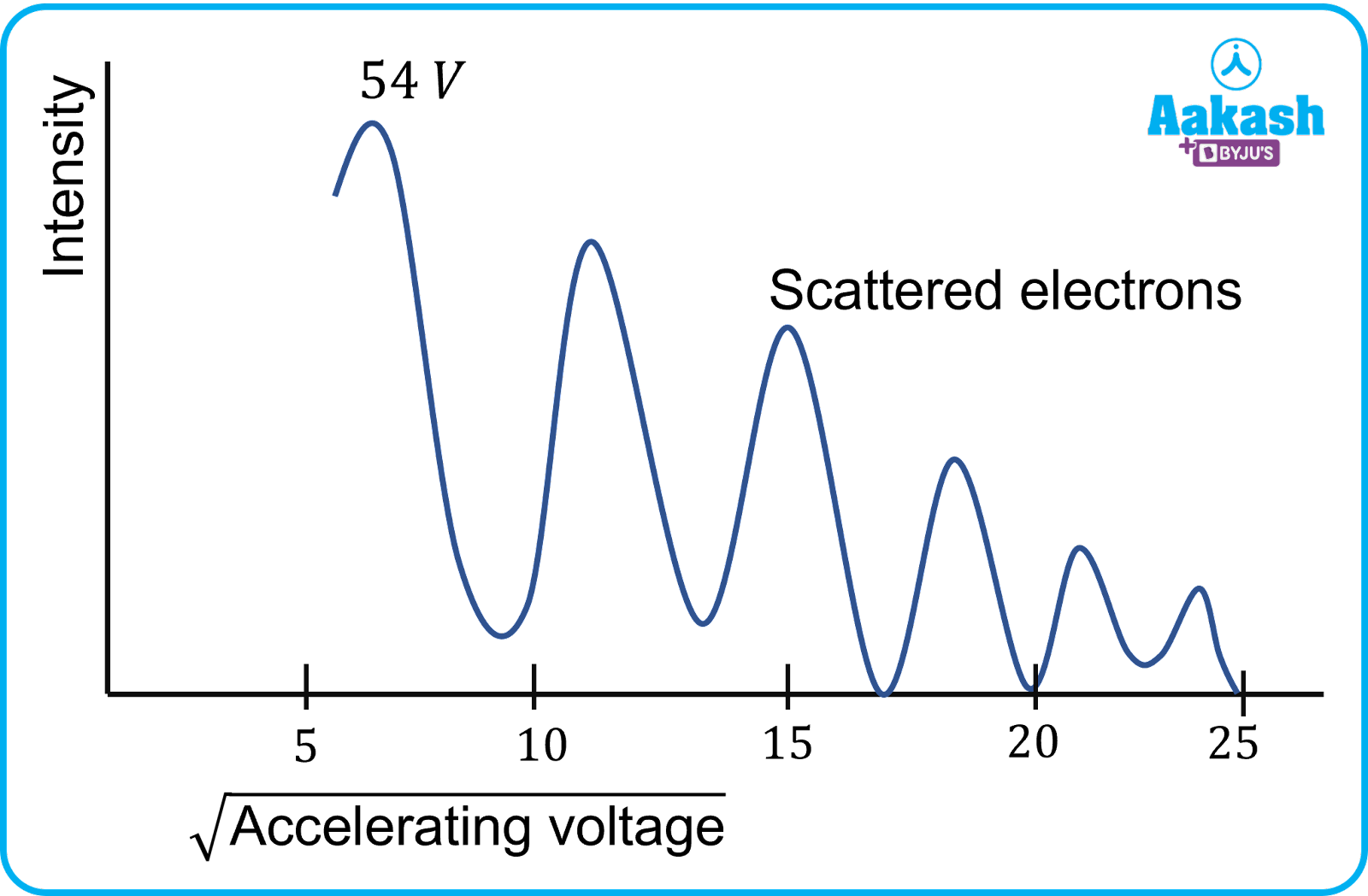
- It was noticed that a strong peak appeared in the intensity (𝐼) of the scattered electron beam for an accelerating voltage of 54 𝑉 at a scattering angle 𝜃=50°.
- This peak was the result of constructive interference of the electrons scattered from the nickel target .
- The wavelength of matter waves was practically found to be 0.165 𝑛𝑚 from the electron diffraction measurements.
- Theoretically, the de Broglie wavelength λ associated with electrons
For 𝑉=54 𝑉
𝜆=hP=1.227V nm 𝜆=0.167 𝑛𝑚
This value is in close agreement with the experimental value of the wavelength obtained using Bragg Law.
- Thus, the Davisson and Germer experiment confirms the wave nature of electrons and de Broglie's hypothesis.
G.P. Thomson’s Experiment
This experiment also provides evidence for the existence of the wave nature of electrons.
The highly accelerated beam of electrons is diffracted through a thin gold foil and the diffraction pattern is collected on the photographic plate . It is observed that the diffraction pattern consists of circular fringes just like the diffraction of light through a circular aperture.
Heisenberg’s Uncertainty Principle
- It is not possible to measure both the position and momentum of a particle at the same time exactly.
- There is always some uncertainty (∆𝑥) in the specification of position and some uncertainty (∆𝑝 ) in the specification of momentum
Δx Δp ≥ h4π
- Means if the position is measured very accurately then the momentum will have a lot of uncertainty and vice versa.
Practice problems
Q. The position of a chloride ion on a material can be determined to a maximum error of 1 m. If the mass of the chloride ion is 5.86 10-26 kg, what will be the minimum error in its velocity measurement?
(h=6.62 10-34 Js)
A.
Given,
x=1 m=10-6 m
From Heisenberg’s Uncertainty Principle,
Δx Δp ≥ h4π
Δx mΔv ≥ h4π
Δv ≥ h4πmΔx=6.62 10-34 4π5.86 10-2610-6=910-4 m/s
Δv ≥910-4 m/s
Therefore, the minimum error in its velocity measurement is 910-4 m/s.
Q. Electrons also show diffraction effects since apart from particle properties it has wave properties too.. Davisson-Germer demonstrated this by diffracting electrons from crystals. The electron waves reflected from the planes of atoms in a crystal interfere constructively. The law governing the diffraction from a crystal is obtained by using this criteria.
Electrons are diffracted from a crystal when accelerated by potential V. If d=1 Å and i = 30o then V should be about h=6.6410-34 Js , m=9.110-31 kg, e=1.610-19 C)
A.
Given, d=1 Å and i = 30o h=6.6410-34 Js , m=9.110-31 kg, e=1.610-19 C
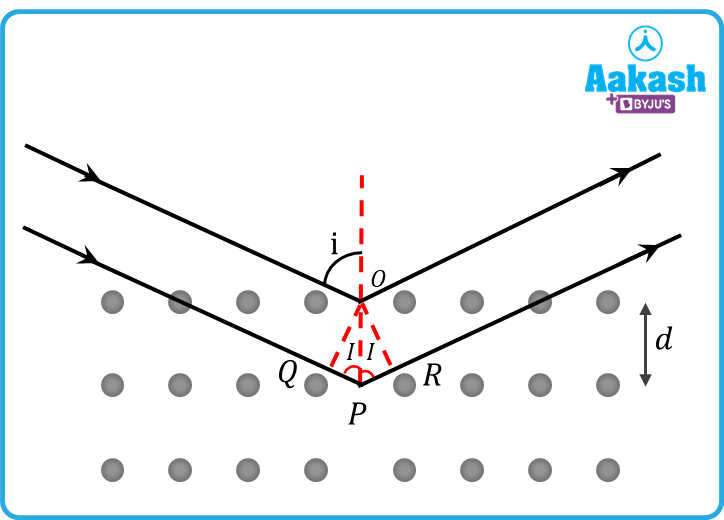
For constructive interference, path difference must be,
x = n
From the figure, we
Path difference, x = QP + PR
= OP i + OP i= 2d i (Since OP=d)
2d i= n
=2d in=2110-1030on=3nÅ
For n = 1 = 3 Å
The de-Broglie wavelength associated with an electron acceleration through a potential difference V is,
=12.27V Å
V=12.2722=150.55350 V
Q. Davisson-Germer experiment is performed first with X-rays of wavelength and then with electrons of the same wavelength . Determine the ratio of energy of X-rays to that of the electrons. (m = mass of electron)
A.
Now, the energy of the X-ray and the electron are as follows,
EX= h=hc
Ee=12mv2=p22m=h22m2 =hp
∴ EXEe=hc2m2h2=2mch
Q. A wet ball weighing 10.1 g has water of 0.1 g on it. The ball is moving with a constant velocity with an uncertainty of momentum of 10-6 kg m/s. What will be the ratio of uncertainty in the measurement of the position of the ball, water and electron in the water molecule ? (mass of the electron, 9.110-28 kg)
A.
From Heisenberg’s Uncertainty Principle,
Δx Δp ≥ h4π
Δx mΔv ≥ h4π
Δx ≥ h4πmΔv
Velocity being constant, uncertainty in the measurement of the momentum is associated with the mass of the matter.
∴ x1m
xball:xwater:xelectron=1mball:1mwater:1melectron
=1(10.1-0.1):10.1:19.110-28=1:100:1.11028
FAQs
Q. What is the importance of the wave nature of electrons?
A. It is used to describe the density of energy states of electrons in solids. The wave nature of matter (or electrons) are just the probability waves that give the probability of finding the electrons at some position.
Q. What does the wave-particle duality of matter imply?
A. It implies that there is a natural tradeoff between the position and finding the momentum of an object. It is not possible to measure or predict the exact position and exact momentum at the same time. This principle is called the Heisenberg uncertainty principle.
Q. If the matter constitutes the wave nature, why don’t we observe it in the normal macroscopic objects like a ball?
A. The wavelength associated with the wave nature of macroscopic objects like a ball is so small that for all practical purposes they are treated as particles. The wavelengths associated with these objects are smaller than the size of a nucleus. Therefore, it is almost impossible to observe their wave nature.
Q. Which theory was supported by the Davisson Germer Experiment?
A. The Davisson Germer Experiment proved the de-Broglie hypothesis that particles have dual nature of a particle and a wave.


