-
Call Now
1800-102-2727
Atomic mass, size, composition of nucleus, practice problems, FAQs
The structure of atoms was always a subject of intrigue for scientists. Many, including JJ Thomson and Neils Bohr had suggested various models to explain the motion of electrons inside an atom; Rutherford, along with his disciples Geiger and Marsden, conducted the famous gold foil experiment, in which they observed that most of the alpha particles(helium nuclei) went undeflected; meaning that majority of the space in an atom is empty; the entire positive charge appears to be concentrated at the core of an atom called its nucleus. Understanding the structure, size and volume of nucleus is vital; it finds various applications in nuclear phenomena like nuclear fission and nuclear fusion. In this article, we will explore more on the size, composition and density of a nucleus.
Table of contents
- What is nucleus?
- Composition of the nucleus
- Size of the nucleus
- Nuclear density
- Isotopes, isobars and isotones
- Practice problems
- FAQs
What is nucleus?
The entire positive charge of an atom is concentrated at the center called its nucleus. It consists of protons(+vely charged) and neutrons(no charge). The nuclear force which exists between them is charge independent; i.e. .
Composition of the nucleus
Nucleus is made up of positively charged protons and neutrons; the electrons revolve around the nucleus.The mass of the proton (mp) and neutron (mn) are as follows;
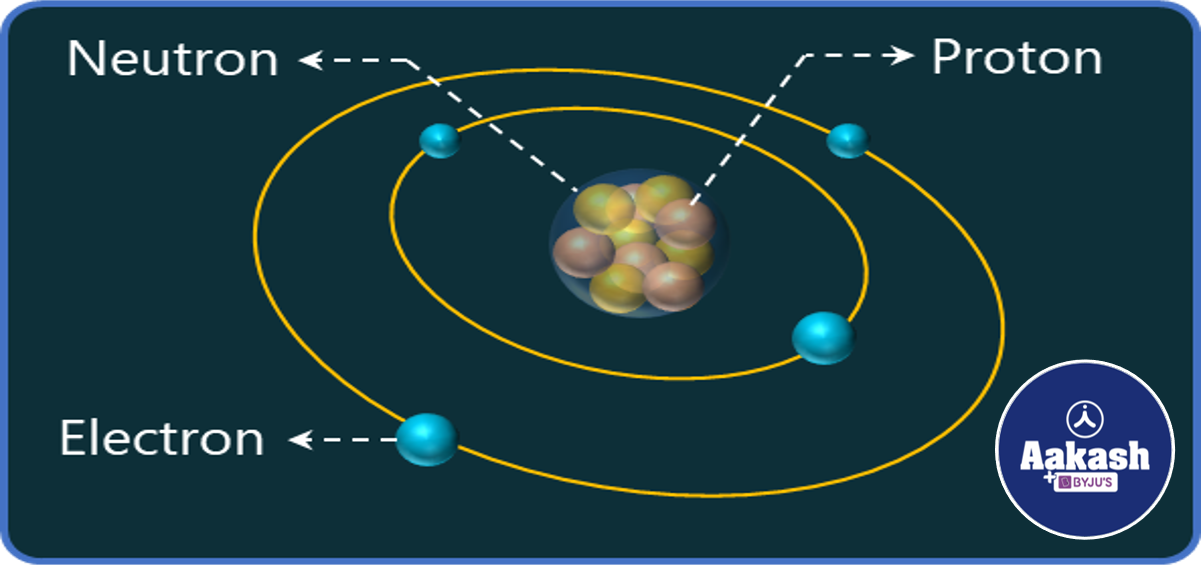
Fig showing an atom with the nucleus at the center
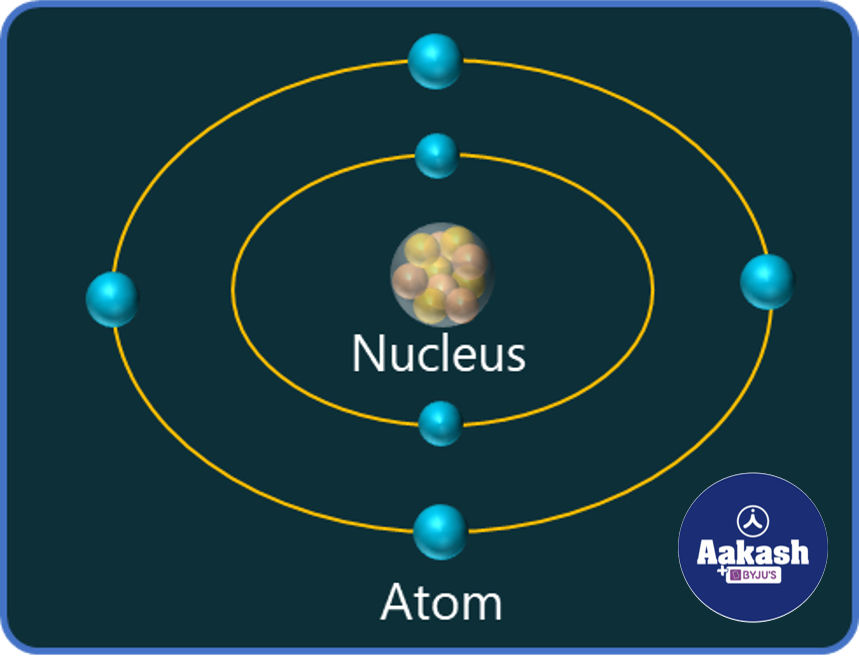
Nucleus in an atom
Atomic Number
The atomic number is defined as the number of protons; which in turn is equal to the number of electrons. It is represented by the letter Z.
Z=No. of protons=No. of electrons
Mass number
The total number of neutrons and protons which are present in the nucleus is called the nucleons or mass number.(A)
A=Number of nucleons=Number of neutrons+protons
Any element X, can be written in the form
The total charge in the nucleus is equal to Ze, where e= is the electronic charge; in other words it is the charge carried by one electron.
Size of the nucleus
After much experimentation, it was found that the volume of the nucleus is directly proportional to the number of nucleons i.e. the mass number. If R represents the radius of the nucleus, having mass number A , then
; is an empirical constant called nuclear unit radius.
Size of the nucleus
Size of the atom
Note: 1 fermi
Nuclear density
So now you have an idea about the nucleus, its size, and what actually constitutes a nucleus. Now let us talk about nuclear density, which gives us an idea about how closely protons and neutrons are packed.
Density of nucleus =
Let m indicate the mass of one nucleon and A is the mass number, then mass of the nucleus=mA, where m indicates the mass of a nucleon, A is the mass number. If R indicates the radius of the nucleus, then
Volume of nucleus= =
∴Density of the nucleus,
I.e. the nuclear density is independent of the mass number A.
Isotopes, isobars and isotones
Isotopes
Atoms of the same element which have same atomic number but different atomic mass are called isotopes
eg.
Isobars
Atoms of different elements having same mass number( A ) but different atomic number ( Z ) are called isobars
eg. and
Isotones
The atoms of different elements having equal number of neutrons A-Z are isotones
eg. and both have 4 neutrons.
Practice problems
Q1. Calculate the approx. nuclear radius of Fe125, if that of Al27 is 6.4 fermi?
Solution) We know that
=
Q2. The nucleus of an atom has a mass number of 24. Then it has;
(a)11 protons and 13 electrons.
(b)11 protons and 13 neutrons.
(c)11 electrons, 13 protons, 11 neutrons.
(d)13 electrons, 11 protons, 13 neutrons.
Solution) b
Mass number=Number of protons+Number of neutrons=11+13=24.
Q3. The radius of in fermi is ()
(a)1.2
(b)4.8
(c)7.7
(d)9.6
Solution) b
Q4. In nucleus, there are
(a)138 neutrons and 88 protons
(b)138 protons and 88 neutrons
(c)226 neutrons and 88 electrons
(d)226 neutrons and 138 electrons
Solution)a
No. of neutrons=226-88=138
No. of protons =88
FAQs
Q1. Define mass number.
Ans) Mass number is defined as the number of nucleons(protons+neutrons) that are present in the nucleus.
Q2. Define atomic number
Ans) Atomic number (Z) is the number of protons in an atomic nucleus which is equal to the number of electrons.
Q3. Write the expression for nuclear density
Ans) Nuclear density, where is a constant and m is the mass of one nucleon.
Q4. Does nuclear density depend upon the mass number?
Ans)
From the equation, it is clear that nuclear density is independent of mass number.


