-
Call Now
1800-102-2727
Thermochemistry - Introduction, Enthalpy and Internal Energy Changes, Measurement, Laws, Enthalpy, Types, Practice Problems and FAQs
Are you concerned about your health? Does your diet contain the proper ratio of macro- and micronutrients, vitamins, fibres, and other nutrients?
The World Health Organization (WHO) restricts the consumption of fats (30%), fruits and vegetables (approximately 400g), simple sugars (10%),and other foods, and eventually recommends eating no more calories (or energy) than you expended. It is clear that every food product releases heat energy during digestion for the body to use. Any chemical that passes through a physical or chemical transition, including those that include food, releases or absorbs heat energy.

Thermochemistry is the branch of chemistry that deals with such heat energy changes accompanying physical or chemical changes.
We will learn about thermochemistry and the rules that govern it in this article.
Table of contents
- Enthalpy and Internal Energy Changes
- Measurements of Energy Changes
- Laws Governing Thermochemistry
- Enthalpy of Reaction
- Types of the Heat of Reactions
- Practice Problems
- Frequently Asked Questions
Enthalpy and Internal Energy Changes
Every system possesses energy from its inherent nature and potential or kinetic energy. The chemical bond holding atoms and molecules together, their arrangement etc contributes to their inherent energy, while the temperature of the substance creates additional energies of vibration motion, electronic transition etc. So, a change in the bonding nature (breaking and making of chemical bonds during chemical fractions) and a change in temperature changes the energy of the products.
Estimating the energy possessed by the chemical species is though difficult, it is unnecessary. We are interested in the change of energy between the initial and final state, which can also be measured accurately.
The changes can be conducted under either constant volume or constant pressure conditions. The heat energy changes measured at constant volume conditions are called Internal energy change(U) and the heat energy changes measured at constant pressure conditions are called Enthalpy(H).
The enthalpies measured under standard conditions of one bar pressure are called standard enthalpy and represented as H°
Enthalpy and internal energy have units of energy as Calories or Jules.
Measurements of Energy Changes
A calorimeter can be used to experimentally measure internal energy and enthalpy. An isolated system with a fixed volume or pressure is the design of a calorimeter. Calculating internal energy, enthalpy, stability, heat capacity, and other thermochemical quantities often involves the use of calorimetry.
The heat effect that follows the transformation may be indicated by an increase or decrease in temperature, as the case may be, of the substances present if the vessel containing the reacting system is so insulated that no heat flows into or out of the system (adiabatic condition). For the right design of equipment for use in chemical processes, accurate temperatures of reaction values are required.
- Measurement of Change in internal energy U
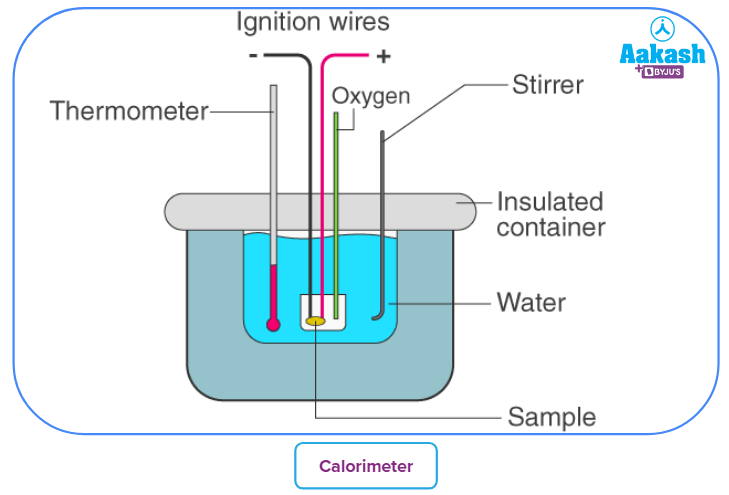
Measurement of heat changes is based on the first law of thermodynamics, ie heat energy lost by the system will be equal to the heat energy gained by the surroundings.
The internal energy changes are measured in a closed fixed-volume container called a BOMB calorimeter.
The energy changes in the system produce equivalent changes in the vessel and water surroundings. Measuring the temperature change, the heat changes are calculated by the formula
Q = mCvT , where m is the (mass of water + water equivalent of the calimeter), Cv is the heat capacity
(heat required to raise the temperature of one gram of a substance by one degree temperature) and
T is the difference in temperature before and after the process.
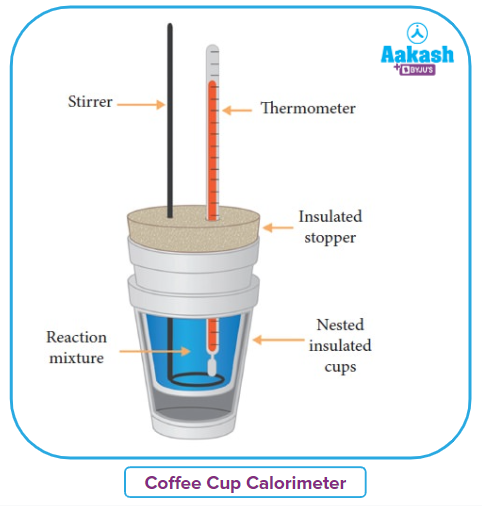
- Measurement of Change in Enthalpy H
Enthalpy is measured in a ‘Coffee Cup’ like insulating material made of polystyrene material, with a cap. A thermometer measures the temperature of the water present in the coffee cup before and after the instrument. Any gas formed will escape through the lid ensuring constant pressure inside the cup.
The heat changes can be calculated from the increase in temperature, which will now correspond to enthalpy.
Laws Governing Thermochemistry
First Law of Thermodynamics
First law of thermodynamics states that, though energy in one form can be converted into another form, the total energy will always be constant. All laws of thermochemistry are based on the first law of thermodynamics.
The heat liberated during a chemical reaction is absorbed by the surroundings. And heat taken during the reaction comes from the surroundings. The heat changes are part of the reaction and the stochiometric reaction with heat changes is called the thermochemical equation.
Enthalpy can be either included in the reaction or written separately outside the equation with a negative sign for heat release and a + sign for heat uptake.
- Exothermic reactions:
Heat-releasing reactions are called an exothermic reactions. Enthalpy(H) can be written as part or exclusive of the reaction.
i) A + B +.... → C + D +.....+ H or as
ii) A + B +.... → C + D +...: H = -ve
Exothermic chemical reactions that include other forms of energy changes also are called exogenic reactions.
- Endothermic reactions
Heat-absorbing reactions are called an endothermic reaction. Enthalpy(H) can be written as part or exclusive of the reaction.
i) A + B +....+ H → C + D +..... or as
ii) A + B +.... → C + D +...: H = +ve
Endothermic chemical reactions that include other forms of energy changes also are called endogenic reactions.
- Relationship between internal and enthalpy changes.
Enthalpy and internal energy of a system are related by the relation-
H=U+PV
Absolute values are not directly measurable and for practical purposes, changes between two states are sufficient.
H=(U+PV)
H=U+PV
H=U+PV + VP
Enthalpy is measured at constant pressure, so P = 0,
H=U+PV = qP
The change in volume is considered only in gaseous systems. In reactions involving solid and liquid phases, changes in volume are negligible and hence for them, H=U
In reactions involving gases as reactant or products or in both,
i) if the number of gaseous moles is the same on the reactant and product side, then, H=U
ii) if the number of gaseous moles is not same on the reactant and product side, then, H≠U
According to ideal gases, PV = nRT: n is the number of moles of the gasper unit volume, R is the gas constant.
PV = n RT
Substituting for PV,
H=U+n RT
1). Lavosion and Laplace Laws:
The law states that the enthalpy of a forward reaction and backward reaction will be the same in magnitude but opposite in direction.
For example, the formation of carbon dioxide from its elements is an exothermic reaction with enthalpy of -393 kJ mol-1. The reaction enthalpy of the decomposition of carbon dioxide to carbon and molecular oxygen will +393 kJ mol-1.
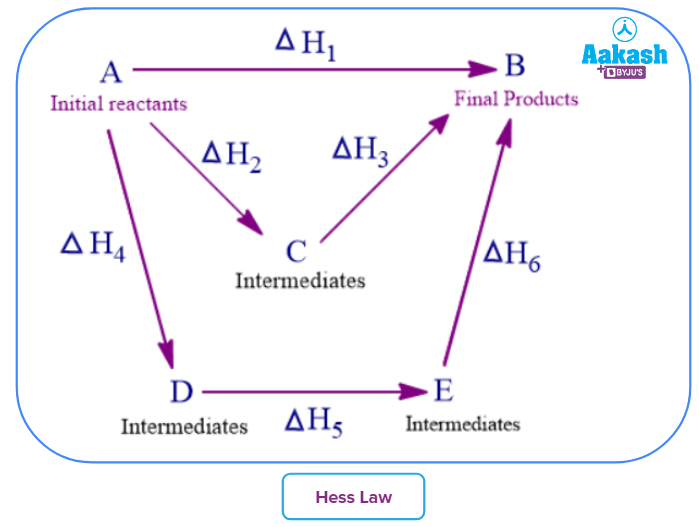
2). Hess Law of Constant Heat Summation
Enthalpy is a state function and hence not depends on the steps involved in the intermediates involved. The enthalpy changes between the same reactants and the same products will remain the same irrespective of the ways in which the final products form from the same reactants.
For example in example, where reactants A and B can form the product AB in three different ways. The enthalpy of the formation of AB will be the same in all three ways.
Enthalpy of Reaction
Enthalpy change is the standard enthalpy of formation, which has been determined for a vast number of substances. In any general chemical reaction, the reactants undergo chemical changes and combine to give products. It can be represented by the following equation:
![]()
For any such reaction, the change in enthalpy is represented as ΔrH and is termed as the reaction enthalpy. The reaction enthalpy is the difference between the total enthalpy of reactants and products..
Mathematically,
ΔtH = Sum of enthalpies of the product – Sum of the enthalpies of the reactants.
![]()
Here, the constants ai and bi denotes the stoichiometric coefficients of the products and the reactants respectively for the balanced chemical reaction under consideration.
Types of the Heat of Reaction
1. The Heat or Enthalpy of Formation
The quantity of heat released during the formation of one mole of a compound when the constituent
elements react is the heat of the reaction.
The standard heat of formation of a compound is the quantity of heat released when the constituent elements in their natural physical state at 25° C and at one atmospheric pressure react to form one mole of the compound.
The standard heat of formation of all elements in their natural physical states at 25° C is assumed to be zero.
2. The Heat or Enthalpy of Combustion
The quantity of heat released when one mole of the compound completely burns in an excess of oxygen is known as the heat of combustion.
The quantity of heat released when one mole of a substance is burned in excess oxygen at 25° C and one atmospheric pressure is known as the standard heat of combustion.

3. The Heat or Enthalpy of Neutralization
The quantity of heat released when 1 gram equivalent of strong acid and 1 gram equivalent of the strong base neutralise each other at 25° C is called heat of neutralization
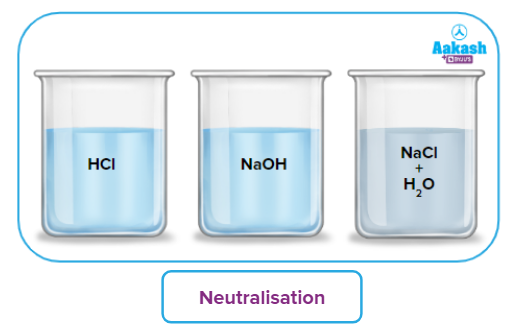
Neutralization of any strong acid and any strong base releases -13.7 kcal eq-1 .
Notes:
1. Ionization energy changes the heat of neutralization
Weak acid or a base is an endothermic process requiring energy of ionization or dissociation which depends on the nature of the acid or the base. As this ionization energy comes from the heat of neutralization, the measured neutralization energy of neutralization of weak acids or weak bases will be less than 13.7 kcal eq-1 to the extent of the heat of ionization.
2. Enthalpy of hydration changes the heat of neutralization.
Ions of the acid and bases get hydrated by either exothermic or endothermic processes. The enthalpy of hydration hence can be either -ve or +ve and depends on the nature of the ions. Smaller and higher charged ions have higher hydration enthalpy adding to 13.7 kcal eq-1.
Aqueous Hydrogen fluoride (HF) though a weak acid, has a high hydration enthalpy than compensating the heat of dissociation to increase the heat of neutralisation of HF larger than -13.7 kcal eq-1 .
4. The Heat or Enthalpy of Solution
The quantity of heat changes accompanying the dissolving of 1 mole of the compound in a solvent of infinite dilution is the heat or enthalpy of solution. Infinite dilution is the amount of solvent whose concentration changes do not affect the measured property.
5. The Heat or Enthalpy of Hydration
The quantity released when one mole of an anhydrous or a partially hydrated form of the salt is dissolved in a solvent at infinite dilution. During the process, the salt is dissociated into ions and the ions are surrounded by the solvent molecules to form hydrated ions.
6. The Heat or Enthalpy of Transition
The quantity of heat changes accompanying the conversion of one mole of an isotope into another form of an isotope of the same element is called the heat of transition.
7. Lattice Energy
Lattice energy is the quantity of heat released when 1 mole of ions in the gaseous phase forms a solid lattice.
8. The Heat or Enthalpy of Hydrogenation
This is the quantity of heat given out when 1 mole of an unsaturated organic compound is converted into a saturated compound with hydrogen.
9. The Bond Dissociation Energy
The heat energy required to break one mole of the chemical bond in a molecule is the bond dissociation energy.
Practice Problems
Q1. Enthalpy of combustion is:
- Always Positive
- Always Negative
- Can be positive or negative
- None of the above
Answer: (B)
Solution: Enthalpy of combustion is the amount of heat released when one mole of a compound is combusted (completely) in excess of air (O2). Combustion is an exothermic process and hence the enthalpies will be always -ve.
Q2. Calculate the standard enthalpy of the formation of carbon disulfide, given that the standard enthalpy of combustion of carbon (s), sulphur (s), and carbon disulfide (l) are 390, 290, and 1100 kJ mol-1 respectively.
A. 160 kJ mol-1
B. 30 kJ mol-1
C. 130 kJ mol-1
D. 60 kJ mol-1
Answer: C
Solution:
Given:
…………(1)
…………..(2)
………….(3)
Applying this operation on the equations, eq (1) +2eq (2) - eq (3), we get;
C
So, standard enthalpy of formation of carbon disulfide (l) is 130 kJ mol-1.
Q3. The heat of combustion of CH4, C2H4 and C2H6 are –890, –1411 and –1560 kJ mol-1, respectively. Which of the following has the lowest calorific fuel value in kJ g-1?
A. CH4
B. C2H4
C. C2H6
D. All of the above.
Answer: (B)
Solution:
Step 1: Calculating the calorific value of CH4
Molar mass of CH4 = (12 + 4) = 16 g mol-1
= 55.625 kJ g-1
Step 2: Calculating the calorific value of C2H4
Molar mass of C2H4 = ((12x2)+ (4x1)) = 28 g mol-1
= 50.39 kJ g-1
Step 3: Calculating the calorific value of C2H6
Molar mass of C2H6 = ((12x2) + (6x1)) = 30 g mol-1
= 52 kJ g-1
So, C2H4 has the lowest calorific fuel value.
Q4. Calculate the enthalpy of the formation of acetic acid using the given information.
- Combustion of Acetic acid
- Combustion of Grahite
- Combustion of Hydrogen
Solution:
The formation reaction of acetic acid is:
Given:
………(1)
…………. (2)
……….. (3)
Applying this operation on the equations, 2eq (3) +2eq (2) - eq (1), we get;
Frequently Asked Questions-FAQs
Q1. What is the difference between standard reaction enthalpy and standard enthalpy of combustion?
Answer: Standard enthalpy of combustion is defined as the enthalpy change when one mole of a substance is completely burnt in the presence of oxygen with all the reactants and products in their standard state under standard conditions (298 K and 1 bar pressure) while the standard enthalpy of reaction (rH°) is the enthalpy change that occurs in a system when the matter is transformed by a given chemical reaction when all reactants and products are in their standard states under standard conditions (298K and 1 bar pressure).
Q2. Is it possible for the enthalpy of combustion to be positive?
Answer: The answer to this question is no. Enthalpy of combustion is the heat energy released when one mole of a substance burns entirely in oxygen and the combustion reactions are always accompanied by the evolution of heat, therefore, the value of cH° is always negative.
Q3.Can you tell us some real-life examples of enthalpy in action?
Answer: Enthalpy in operation can be seen in refrigerator compressors and chemical hand warmers. The vaporisation of refrigerants in the compressor and the reaction to iron oxidation in a hand warmer both induce a change in heat content under constant pressure which is an enthalpy change.
Q4. What is the enthalpy of combustion used for?
Answer: Combustion reactions are exothermic. But the enthalpy depends on the composition. Higher the carbon and hydrogen content higher the enthalpy of combustion. Compounds with a high enthalpy of combustion are used as fuel. The enthalpy of combustion is directly proportional to the calorific value. Hence the enthalpy of combustion is used to compare the fuel efficiency.


