-
Call Now
1800-102-2727
Strength of Oleum - Definition, Practice Problems & FAQs
Which chemical is termed the king of chemicals?
Sulphuric acid (H2SO4) is treated like a king of chemicals because you can find its use in various segments/areas like manufacturing of fertilizers, pigments, and dyes, different drugs, explosives, soaps and detergents, inorganic salts, petroleum refining, lead-acid storage batteries, metallurgical processes, etc. Even consumption of sulphuric acid also reflects the industrial strength of any country.
In the contact process (commercial method of manufacturing sulphuric acid) oleum (H2S2O7) is produced and further mixing with water to produce sulphuric acid of the desired concentration.

Table of contents
- Strength of oleum
- Application of oleum
- Practice problems
- Frequently asked questions-FAQs
Strength of oleum
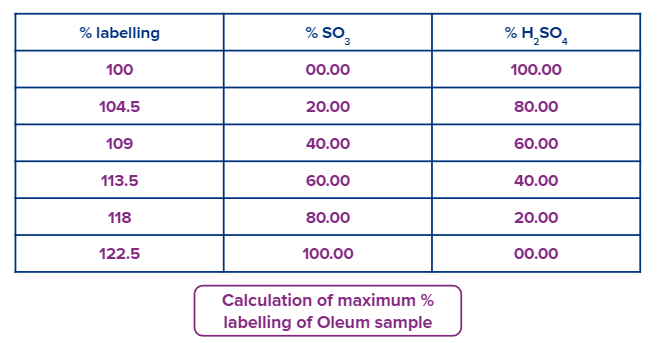
Oleum is a mixture of sulphuric acid (H2SO4) and sulpher trioxide (SO3). To dissolve free SO3 water is mixed with oleum, in this process free SO3 combines with water to form H2SO4.
100 g of sample of oleum is mixed with x g of water, the total weight of obtained sulphuric acid is known as % labelling of oleum.
If a sample of oleum is labelled as 111 % H2SO4, means (111-100 = 11) grams of water added in 100 gram oleum sample to produce pure 111 g pure H2SO4.
If a sample of oleum is labelled as 109 % H2SO4, means 9 grams of water added in 100 gram oleum samples to produce pure 109 g pure H2SO4.
Molar mass of
Molar mass of
Molar mass of
From the above reaction we can see, 18 g H2O combines with 80 g SO3 to produce 98 g of H2SO4.
So, for x% H2SO4 (labelled oleum sample, (x-100) g H2O combines with 8018(x-100) g SO3 to produce x g of H2SO4.
Applications of oleum:
- Oleum is less corrosive than sulphuric acid, it is used for the transportation of sulphuric acids.
- Oleum has been used in the manufacturing of many explosives.
- Oleum is used in organic chemistry for secondary nitration.
Practice problems:
Q 1. Find the percentage of free SO3 in an oleum sample labelled as 121 %
a. 93.33 %
b. 46.67 %
c. 88.89 %
d. None of these
Answer: (A)
Solution: x% H2SO4 (labelled oleum sample, (x-100) g H2O combines with 8018(x-100) g SO3 to produce x g of H2SO4.
Q 2. Find the percentage of H2SO4 the present in an oleum sample labelled as 118 %
a. 40 %
b. 20 %
c. 60 %
d. 80 %
Answer: (A)
Solution:x% H2SO4 (labelled oleum sample, (x-100) g H2O combines with 8018(x-100) g SO3 to produce x g of H2SO4.
So,
Q 3. 0.75 g of oleum mixed with water. The prepared solution is completely neutralized by 25 mL of 0.5 N NaOH. Calculate the free percentage of SO3 in sample
a. 73.65 %
b. 45.87 %
c. 81.47 %
d. 86.98 %
Answer: (C)
Solution: we know, the oleum contains both SO3 and H2SO4. So, the base reacts with both the initial amount of H2SO4 and newly formed H2SO4 by
So, according to the law of equivalence;
Number of equivalent = Mole n-factor
Number of equivalent = Normality Volume (L)
Let, the weight of free SO3 in sample=x g
weight of free H2SO4 in sample=(0.75-x) g
free percentage of
Q 4. Name of the mixture H2SO4.SO3 is
a. Pyrosulphuric acid
b. Oleum
c. Fuming sulphuric acid
d. All of these
Answer: (D)
Solution: Pyrosulphuric acid - H2S2O7. oleum is called fuming sulphuric acid. When SO3 contact with air it quickly reacts with moisture and forms white fumes.
Frequently asked questions-FAQs
Q. What are pyro acids?
Answer: Pyroacids are derived acids, if one H2O molecule is removed from 2 molecules of acid then the pyro prefix is added in the name of acid.
2H2SO4 ------ H2S2O7(removal of 1 H2O molecule)
Sulphuric acid pyrosulphuric acid
2H3PO4 ------ H4P2O7(removal of 1 H2O molecule)
Phosphoric acid pyrophosphoric acid
Q. What are meta acids?
Answer: Meta acids are derived acids, if one H2O molecule is removed from 1 molecule of acid then the meta prefix is added in the name of acid.
H3PO4 ------HPO3(removal of 1 H2O molecule)
Phosphoric acid Metaphosphoric acid
H3BO3 ------HBO2(removal of 1 H2O molecule)
Boric acid Metaboric acid
Q. What is contact’s process?
Answer: it is a commercial method of manufacturing sulphuric acid. Oleum is produced by the process and by dilution required concentration of sulphuric acid is obtained. 96 to 98 % pure sulphuric acid can be obtained from
The general steps involved in manufacturing sulphuric acid by contact’s process:
1. Production of sulfur dioxide by burning sulfur or by the roasting of sulfide ores.
2. Conversion of sulfur dioxide (SO2) to sulfur trioxide (SO3) by a suitable catalyst. (V2O5 is used as catalysts in the contact process) .
3. Sulfur trioxide (SO3) is absorbed in sulphuric acid to give oleum (H2S2O7)
Q. what is the strength of the solution?
Answer: it's a method to express the concentration of the solution, strength of solution means grams of solute present in 1000 mL of solution.
If an oleum sample labelled as
Let,
means the maximum percentage of free SO3 is 100 for an oleum sample labelled as 122.5 %


