-
Call Now
1800-102-2727
Second Order Integrated Rate Equation - Order of Reaction, Second Order Reaction Types- Differential and Integrated Form, Identification, Examples, Practice Problems, FAQs
Do you know that every child's chromosomes are made from both of their father's and mother’s chromosomes? Likewise, there are also chemical reactions whose rate of formation of products is dependent on two reactant molecules. These reactions are called second-order reactions.

On this concept page, we will get to know more about the different forms of second-order reactions with emphasis on the second-order integrated rate equation.
Table of Contents
- Order of reaction
- Second Order Reaction Types
- Differential and Integrated Forms
- Identification of Second Order Reactions
- Examples
- Practice Problems
- Frequently Asked Questions (FAQs)
Reaction Order
The Order of reaction indicates the effect of reactants and their concentration on the rate of reaction.
The law of mass action predicts that the rate of a reaction is proportional to the concentration of the reactants. The rate law predicts the rate to be proportional to some power of concentration of each reactant as given below.
The rate equation is given by, Rate of a reaction .;
where [A] and [B].... are the molar concentration of reactants A and B…...
The powers of concentration, namely, x, y…. are some integers called the partial order of reaction with respect to A, B…. respectively.
The order of the reaction will be the addition of these concentration powers. Depending on the sum the reactions can be of the overall orders of zero, first, fractional second order and so on.
Removing the proportionality with a proportionality constant, ‘k’, called the rate constant,
Rate of a reaction [B]y……….(i)
The rate equation (i) is in a differential form.
But in practice, we will be following the change in observable properties like concentration, colour, pressure, etc with the passage of time. Hence we need a relation between concentration and time for practical purposes. Such a relationship is obtained by integrating the differential rate equation. Such a relation is called the integrated rate equation.
The integrated equation gives a quantitative relationship between the concentration of reactant(s) as a function of time.
Second-Order Reaction Types
From the rate law equations given above, it can be understood that second-order reactions are chemical reactions which depend on twice the reactant concentrations. The two concentrations can be of the same molecules or of two different molecules.
Thus, the second-order reactions can be of two types.
- [A]2, where x = 2, y.= 0
- [B]1, where x = 1, y= 1
Where the sum of x and y (which corresponds to the partial order of the chemical reaction in question) equals two.
Second Order Integrated Rate Equation
- Integrate rate equation for one reactant.
Consider the reaction,
2 A → Products,
As per rate law, Rate of reaction = - d[A]dt = k [A]2
Rearranging the differential rate equation, we get,
- d[A][A]2 = k dt or d[A][A]2 = - k dt
Integrate on both sides with the limits from initial reactant concentration of [A0] at start ie t =0 and final reactant concentration [A] after time t= t.
As per the law of integration, the integrated form becomes.
[ 1[A}][A0]{A] = -k [t]0t
- [1[A] - 1[A0]] = -k t or [1[A] - 1[A0]] = k t
This is the integrated rate equation of second order reaction, for a single reactant.
Graphical representation
A plot of the reciprocal of reactant concentration with time give a straight line with a slope of k and intercept of the reciprocal of the initial concentration of reactant.

- Integrate rate equation of multiple reactants
A + B → Products,
The reaction rate of this reaction = - d[A]dt = - d[B]dt
As per rate law, the Reaction rate of this reaction = - d[A]dt = - d[B]dt= k [A]1[B]1
i) Case 1: When [A] = [B],
The Reaction rate of this reaction = - d[A]dt = - d[B]dt= k [A]2
Integrated rate equation is the same as that of a single reactant derived earlier.
ii) Case 2: When [A] ≠ [B],
Reaction rate of this reaction = - d[A]dt = - d[B]dt= k [A]1[B]1
Let, a and b be the initial concentration of reactants A and B.
Let ‘x’ be the amount reacting at time ‘t’. Then the concentration of A and B at time t will be (a-x) and (b-x)
Reaction rate of this reaction = d[x]dt = k [a-x][b-x]
Rearranging the equation, dx(a-x)(b-x) = k dt
Integrating within reacting concentration limits of 0 and x between time 0 and t seconds,
0x dx(a-x)(b-x) = k 0tdt
The integration can be done by after the denominator by a partial fraction, which says,
1(a-x)(b-x) = 1(a-b) [ 1b-x - 1a - x]
So the integration now is
On integration,
Rearranging,
1(a-b)ln [ b(a-x)a(b-x)] = k t
Multiplying bioth sides by (a-b),
ln [ b(a-x)a(b-x)] = k(a-b) t
Graphical representation
A plot of ln [ b(a-x)a(b-x)] with time give a straight line with a slope of k(a-b) and passing through the origin.

Identification of Second Order Reaction
The order of the reaction as two can be obtained
1. Plotting the reciprocal of reactant concentration against time. A straight line indicates the second order with respect to a single reactant or the same initial concentration of two reactants.
2. A straight line graph passing through the origin on plotting ln [ b(a-x)a(b-x)] against time indicates a second-order reaction of two molecules with different initial concentrations..
3. By calculating the half-life of the reactant.
For a single reactant or the same initial concentration of two reactants, the
half-life = t1/2 = 1k[A0]
Examples Of Second Order reactions:
1. Decomposition of acetaldehyde is second order with respect to acetaldehyde.
2CH3CHO → 2CH4 + 4CO
2. Decomposition of gaseous Hydrogen iodide,
2 HI(g) → H2(g) + I2(g)
3. Hydrolysis of an ester with bases
CH3COOC2H5 + NaOH → CH3COONa +C2H5OH
4. Redox reaction between nitrogen dioxide and carbon monoxide.:
NO2 + CO → NO + CO2
5. Decomposition of ozone to oxygen
2 O3 → 3 O2
Practice Problems
Q1. The given plot represents the variation of the concentration of a reactant R with time. Order of the reaction is:
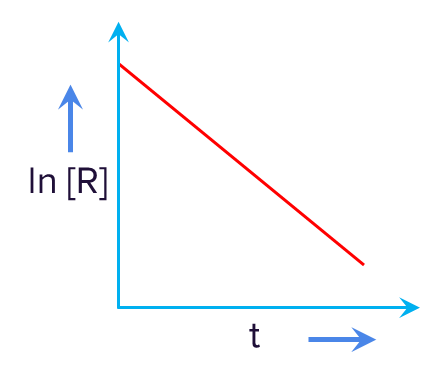
Answer: For the first-order reaction,
Taking ln both sides,
Compare with an equation of a straight line y=mx +c ;
ln[R] on the y-axis and t on the x-axis.
Straight line with a negative slope;
So, the given graph is of the first order.
Q2. Second order reaction rate constant unit is -
a. mol-1s-1
b. s-1
c. Lmol-1s-1
d. molL-1s-1
Answer: Option C
The rate equation of a second order reaction is-
Reaction rate of this reaction =
In unit wise,
Q3. The rate of decomposition of ozone was observed to be X molL-1s-1 at 300K.
What will be the rate of decomposition at the same temperature when the initial concentration of ozone is increased to four times?
a. 4X
b. 8X
c. 16X
d. 32X
Answer: Option C
Rate equation of decomposition of ozone is,
The decomposition rate of ozone = -
If the concentration is 4 times the concentration of ozone,
Decomposition rate of ozone = -
So, the new rate will be 1 times the original rate.
Q4. From the data given below choose the correct stoichiometric reaction.
|
Experiment Number |
Initial concentration of reactant A (molL-1) |
Initial concentration of reactant B (molL-1) |
Initial rate of reaction (moleL-1s-1) |
|
1 |
0.2 |
0.3 |
1x 10-3 |
|
2 |
0.4 |
0.3 |
4x 10-3 |
|
3 |
0.2 |
0.6 |
1x 10-3 |
|
4 |
0.6 |
0.6 |
9x 10-3 |
a. 2A → Products
b. A + B → Products
c. A → Products
d. 2A + 2 B → Products
Answer: Option A
Solution: Comparing experiments one, two and four, the initial rate of reaction doubles and triples with the increase in the concentration of A is two and three times of the first experiments. This indicates the rate of reaction is dependent on reactant A concentration and the power or partial order with respect to A has to be 2.
Comparing experiments one and three concentration of reactant A is constant while that of reactant B is double. Still the rate of reaction remains the same. This means that concentration changes of reactant B do not affect the rate of the reaction or its partial order is zero.
The rate of the reaction can then be written as
Reaction rate of reaction = -
The stochiometric equation for this order will be option A.
Frequently Asked Questions (FAQs)
Q1. A second-order reaction can become a pseudo-first-order reaction when the concentration of two reactants….?
Answer: A second-order reaction occurs when two molecules are involved in the rate-determining step of the reaction. If both molecules are the same or if the concentration of both of them are same, the reaction will only be second order. But if one of the reactants is very high then the rate depends only on the low-concentration reactant and becomes a pseudo-first-order reaction.
Q2. Between the first and second-order reactions, which will be faster?
Answer: The reaction occurs generally by collisions and acquiring the threshold energy of the reaction. In a second-order reaction, there is a condition that the particular reactants have come closer and also at the proper orientations. The chances of such conditions are less compared to a single molecule involving first-order reactions. So, generally, first-order reactions will be faster than second-order reactions.
Q3. What will be the rate constant of a second-order reaction in which 2moles of a substance take 10min to get reduced to one mole?
Answer: Ten minutes is taken to half the initial concentration of 2moles to one mole. This means the half-life of the reaction is 10 minutes.
The half-life of a second-order reaction is,
Substituting for half-life and initial concentration, in the equation,
10 x 60s = 1k 2 or k = 2x10 x60s = 1200seconds.
Q4. What will be the unit of the half-life of a second-order reaction?
a. Zero order
b. First-order
c. Second-order
d. None of them
Answer: Option C
Solution: Half-life of a second order reaction is
K has the unit of mol-1s-1 and concentration unit is mol-1
Substituting the unit in the half-life relation the unit of the half-life of a second-order reaction will be s-1 .


