-
Call Now
1800-102-2727
Saturated Hydrocarbons– Definition, Types, Physical & Chemical Properties, Uses, Practice Problems & FAQs
Have you ever visited a spa or attended an aromatherapy session? If yes, you know how aesthetically they use candles of various colours, sizes and shapes to enhance the overall luxurious experience of such salon treatments.
Even if you haven’t visited one, you must have seen the utilities of candles in all vividity, in such places. You will be even more amazed to know how these candles have a beautiful link with the concept of “saturated hydrocarbons”.

The only hydrocarbons present in paraffin wax are saturated ones. Four to Twenty atoms of carbon-based saturated hydrocarbons can be found in paraffin wax.
Paraffin wax is most frequently used in candle making. Saturated hydrocarbons are used to create paraffin wax, also known as mineral wax, which can be either white or colourless. By dewaxing light oil stocks, it is extracted from petroleum.
Saturated hydrocarbons sourced from mineral oil-based saturated hydrocarbons (MOSH) or synthetic sources also form an integral base component in lipsticks and lip care products.
Let’s explore more about this famous range of compounds– “Saturated Hydrocarbons”!
Table of Contents
- Saturated Hydrocarbon– Definition
- Saturated Hydrocarbon– Types
- Properties of Saturated Hydrocarbons
- Reactions of Saturated Hydrocarbons
- Saturated Hydrocarbon– Uses
- Difference between Saturated Hydrocarbon & Unsaturated Hydrocarbons
- Practice Problems
- Frequently Asked Questions–FAQs
Saturated Hydrocarbon– Introduction
Any of a family of organic compounds with only carbon (C) and hydrogen (H) atoms are known as hydrocarbons. The hydrogen atoms bind to the carbon atoms in a variety of ways to create the compound's structural framework.
Hydrocarbons can be classified in the following ways:
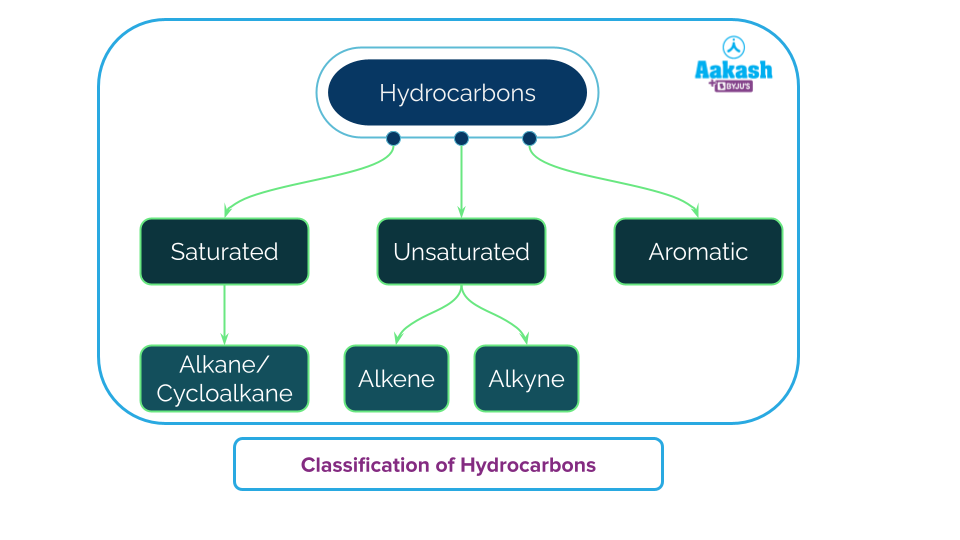
You must have soaked a sponge in water and squeezed it out to remove all water soaked within. When the sponge is soaked in water and seems not to absorb water anymore, we say that it is saturated with water. Saturated hydrocarbons are similar compounds that consist of hydrogen and carbon alone and all the carbon valencies seem to be satiated by hydrogen atoms.
- When all carbon-carbon bonds are single bonds in a hydrocarbon, the hydrocarbon is said to be saturated. A hydrocarbon is an organic substance made up only of hydrogen and carbon.
- Saturated hydrocarbons are hydrocarbons that, as their name implies, are "saturated"—that is, they have no carbon-carbon multiple bonds—and all of their carbon atoms are connected to four other atoms.
- Alkanes, which are acyclic hydrocarbons with solely sp3 hybridised carbon atoms, are typically referred to as "saturated hydrocarbons." An alkane has the generic formula CnH(2n+2).
- From the examples below we can see that every molecule is saturated with a carbon-carbon single bond and the rest of the carbon valencies are satisfied by hydrogen atoms alone.


- Alkanes with a monocyclic ring structure are known as cycloalkanes. Cycloalkanes are regarded as saturated hydrocarbons since all of their carbon-carbon linkages are single bonds.
- A saturated hydrocarbon's general formula is thus Cn H(2n + 2 - 2r), where r is the total number of rings in the molecule.
- Saturated Hydrocarbons consist of only σ-covalent bonds.
Saturated Hydrocarbon– Types
A saturated hydrocarbon can be categorised into the following kinds depending on whether its structure is linear, branching, or ring-shaped:
Alkanes (Linear/Branched):
Alkanes' structures can include either a straight or a branched carbon chain. These saturated hydrocarbons have sp3 hybridised carbon atoms all throughout.
The length of an alkane's carbon chain affects both its melting and boiling points. The melting or boiling point rises with the length of the chain. This is due to the large molecular weights of compounds with lengthy carbon chains.
Successive homologues of alkanes differ from each other in the molecular formula by a -CH2 only.
Examples:
CH3-CH3 (Ethane), CH3-CH2-CH3 (Propane), and CH3-CH2-CH2-CH3 (Butane).
Alkanes with up to four carbon atoms are gases at ordinary temperatures, whereas those with five to seventeen carbon atoms are liquids. At room temperature, alkanes with more than 18 carbon atoms are solids. Alkanes being hydrophobic are insoluble in water.
The total amount of carbon atoms in an alkane determines how many chain isomers are conceivable. For instance, octane (C6H14) contains a total of 6 potential chain isomers, whereas butane only has two.
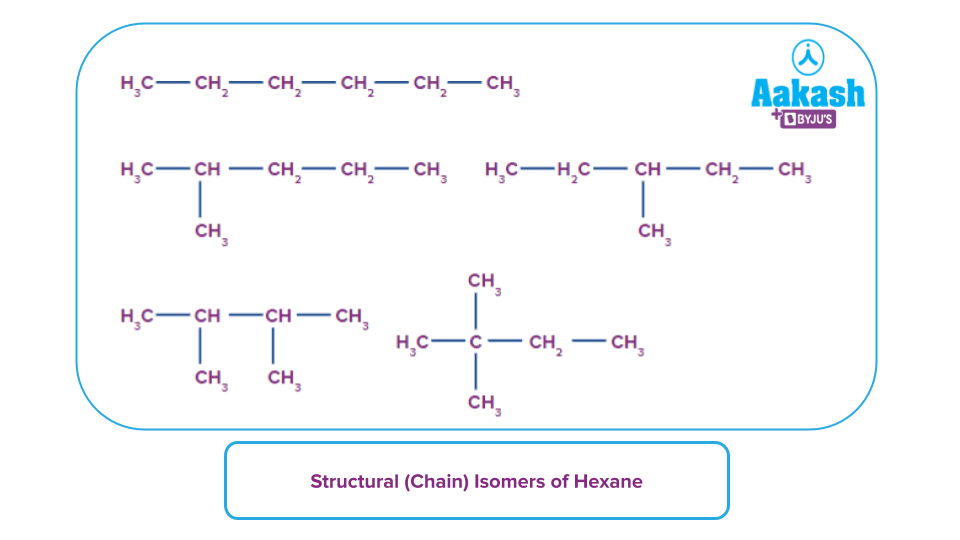
Let's check some examples of straight chain alkanes:
In branched chain hydrocarbons, small chains of carbon atoms are attached to one or more carbon atoms of the parent chain.
As the molecular size or surface area of the molecule increases, we see that the intermolecular Van Der Waals forces also rise. Alkanes have higher boiling points as their molecular weight rises. In contrast to their structural isomers, straight-chain alkanes are shown to have a higher boiling point. The melting point rises as a result of branching because the surface area of molecules reduces, making them more compact, making it simpler to pack them firmly and making it difficult to break that compact structure. The surface area of the molecule lowers as branching increases, as well as the van der Waals forces of attraction, which may be overcome at a somewhat lower temperature. Therefore, when branching increases, an alkane chain's boiling point decreases.
Cycloalkanes:
It is significant to remember that even polycyclic alkanes—alkanes having several rings in their structures—are defined as cycloalkanes and, as a result, fall under the category of saturated hydrocarbons.
sp3 hybridised carbon atoms are present in the rings of cycloalkanes.
A few examples are Cyclopropane (C3H6), Cyclobutane (C4H8), Cyclopentane (C5H10), Cyclohexane
(C6H12).
Cycloalkanes often have melting and boiling temperatures that are greater than those of alkanes with the same number of carbon atoms.
Ring strain affects the cycloalkanes' reactivities because of their structural characteristics. Cyclopropane has the largest ring strain of all the cycloalkanes due to its 60o carbon-carbon bond angle. Therefore it is the most reactive among all cycloalkanes.
At normal temperature, the first four classes of cycloalkanes are considered to be in a gaseous form. Additionally, these compounds are said to have greater melting points and densities.
Since the carbon-hydrogen bonds' electronegativity is discovered to be too low, these compounds are considered to lack any form of polarity. The liquid form of cycloalkanes is believed to be the best form of solvent for other organic compounds, and this family of saturated hydrocarbons is stated to be insoluble in water.
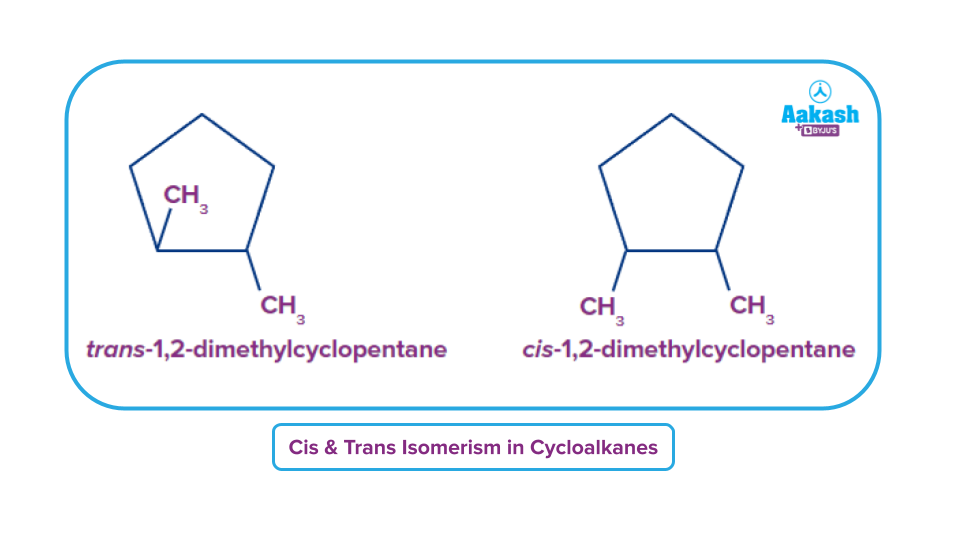
Cis-Trans isomerism in cycloalkanes:
Cycloalkanes show cis-trans isomerism owing to the restricted rotation of the carbon carbon bonds present inside the rings. The lack of unrestricted rotation leads to the creation of cis-trans isomers, also known as geometric isomers. The Latin word "cis" means "on the same side,". Latin is also the source of the word "trans," which means "across from."
Conformations in alkanes & cycloalkanes:
There are infinite arrangements (conformations) which arise due to the free rotation around the carbon-carbon bond, out of which different conformations corresponding to energy minima are called conformers. Hence alkanes like ethane and cycloalkanes show various conformations.
They are defined as conformations that are most stable and have the lowest potential energy as a result of free rotation from 0° - 360°.
Even a very basic alkane like ethane may take on an infinite variety of forms due to the free rotation that exists around a carbon-carbon single bond.
For example: Conformers of Butane, an alkane that has C-C bonds.
Fully eclipsed, gauche, eclipsed, and anti are the four conformational isomers of Butane. When we rotate the butane molecule around the axis of the C2-C3 bond, it normally exhibits distinct conformational isomerism as shown in the diagram below:
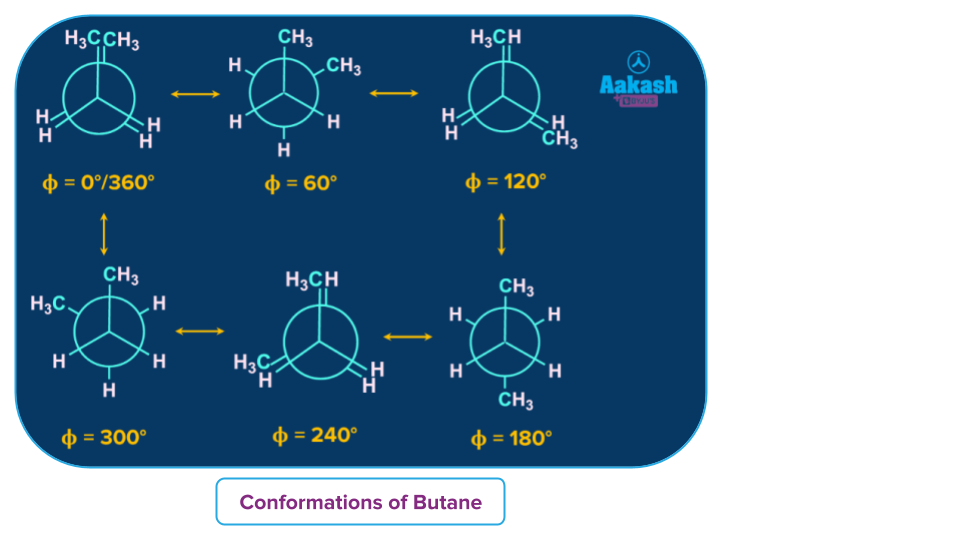
The one with ⲫ value equal to is known as a fully eclipsed form of butane, the conformation with ⲫ value equal to 180° is known as anti conformation, with ⲫ values equal to 120° & 240° are eclipsed or partially eclipsed conformations and with ⲫ values equal to 60° & 300° are gauche conformations of butane.
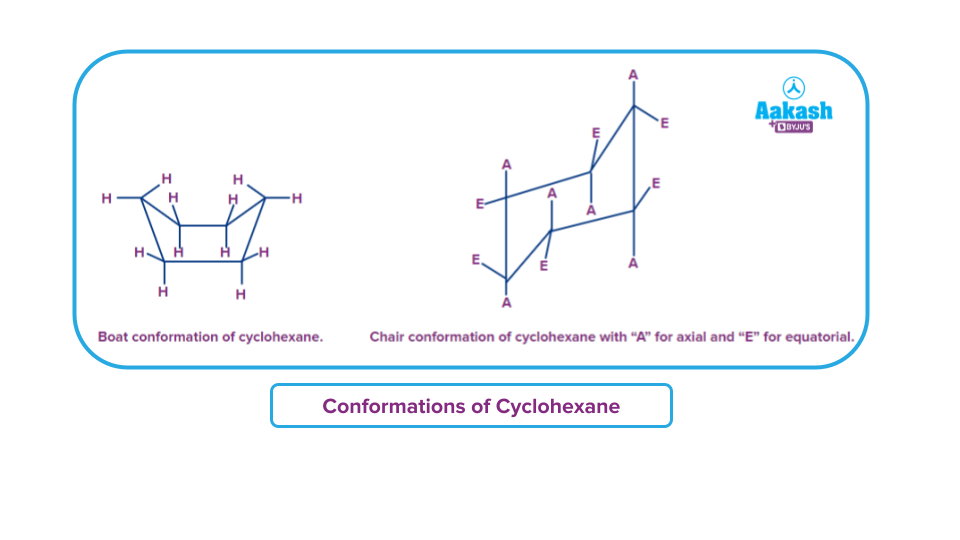
Apart from cyclopropane, cycloalkanes also take on several conformations.
The most energetically beneficial form of cyclohexane is in a "chair conformation," whereas the other is in a "boat conformation." Axial hydrogen atoms are those that are above or below the ring. Equatorial refers to those that roughly lay in the ring's plane. The six equatorial hydrogens change to axial when the chair forms "flips," and the six axial hydrogens change to equatorial.
Properties of Saturated Hydrocarbons
Without any lone pair electrons, carbon always forms 4 bonds. (Carbon monoxide is one exception to this). Carbon atoms that are solely bonded have bond angles of 109.5o. Carbon atoms with tetrahedral shape make alkanes. With the bonded atoms at the vertices of a tetrahedron, any compound with four single bonds of carbon will have a tetrahedral form around any of the single bound carbon atoms.
- Non-polar hydrocarbons exist. Water cannot mix with them.
- Both their melting and boiling points are relatively low. At room temperature, the first four alkanes (methane through butane) are all gases.
- In general, hydrocarbons are less dense than water.
- A homologous series is a family of substances that only differs in the amount of -CH2 groups placed in the carbon chain. These individuals are referred to as homologues.
- A homologous series' members have very comparable chemical reactivity.
- A molecule's physical characteristics gradually change as additional carbons are added and its size grows.
- The melting and boiling temperatures rise with the number of carbon atoms (increase in molecular weight).
- With the exception of very tiny alkanes, each additional carbon in the chain raises the boiling point by 20 to 30 degrees.
Reactions of Saturated Hydrocarbons
Combustion:
Saturated hydrocarbons on reaction with an excess of oxygen yields water and carbon dioxide.
When alkanes are burnt with inadequate oxygen then CO is formed instead of CO2.
Large amounts of heat are released during combustion and hence saturated hydrocarbons are used as fuels. Heat of combustion is directly proportional to the number of carbon atoms in the saturated hydrocarbon and inversely proportional to the branching present in an alkane chain.
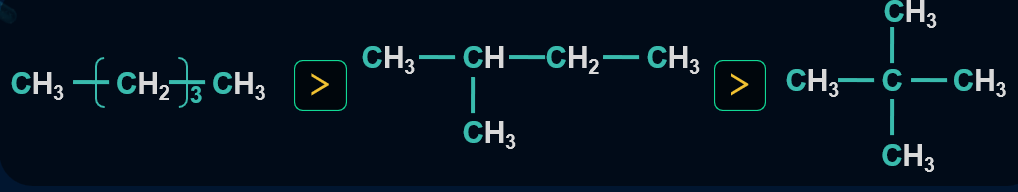
The heat of Combustion per -CH2 group is inversely proportional to the size of the ring. On increasing the size of the ring from 3 to 6 stability of the ring increases and hence heat of combustion (its value) decreases per -CH2 group.

Halogenation:
Saturated hydrocarbons undergo substitution reactions. This type of reaction involves the replacement of one atom with another atom or group of atoms. In halogenation, hydrogens present in the saturated hydrocarbons are substituted with a halogen (-Cl or -Br etc).
Halogenation of saturated hydrocarbons like alkanes or cycloalkanes produces alkyl halides (RX) and hydrogen halide.

Where, X2= Cl2 or Br2
Tertiary hydrogens (3o) are substituted much more rapidly than that of secondary (2o) hydrogens, which react faster than primary (1o).
The reaction usually takes place via a free-radical mechanism and if uncontrolled, all hydrogens present in the alkane get substituted by a halide ion.
Controlled oxidation of alkanes:
Alkanes on heating in a regulated supply of O2 or air at high pressure and in the presence of suitable catalysts, give a variety of oxidation products.
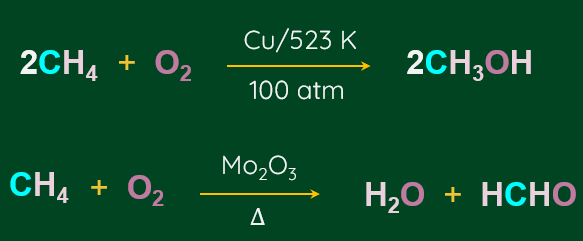
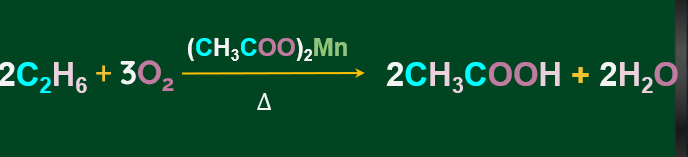
Generally, alkanes resist oxidation in presence of oxidising agents like KMnO4, but alkanes having tertiary H-atoms can be oxidised to alcohol by KMnO4.
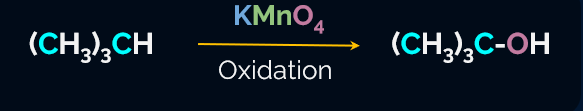
Aromatisation of n-alkanes:
Aromatization is the process of turning an aliphatic chemical into an aromatic compound. Under conditions of high pressure, high temperature, and the presence of a catalyst, alkanes with six to ten carbon atoms are transformed into benzene and its homologues.
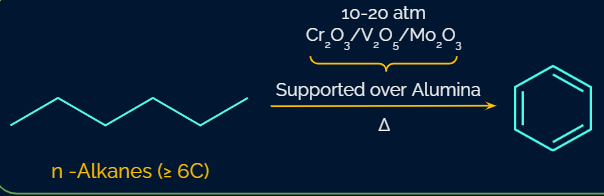
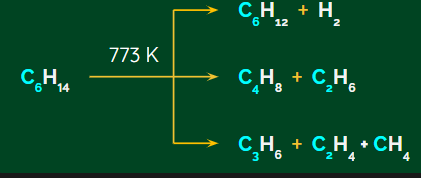
Pyrolysis:
Pyrolysis is the process of heating a substance in the absence of air until it decomposes to give lower alkane, alkene and hydrogen.
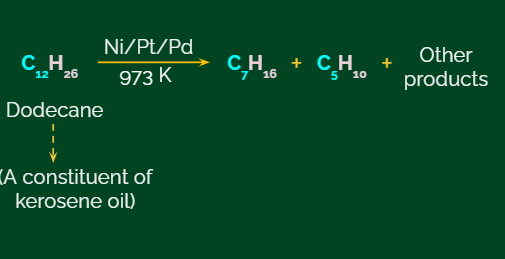
Given below is an example of utilising this chemical reaction of catalysed pyrolysis for the preparation of oil gas from kerosene.
Reaction of alkanes with steam:
Alkanes on reaction with steam at very high temperatures produce a mixture of carbon monoxide and hydrogen gas. It is a method used for the industrial production of hydrogen.

Saturated Hydrocarbon– Uses
Solvents, cooking oils, and fuels all contain alkanes. Here are a few more applications for saturated hydrocarbons.
- Several vehicles, water heaters, and ovens run on methane as fuel. Liquid methane may be used as rocket fuel when purified.
- Ethane is a refrigerant used in a number of cryogenic refrigeration systems. Additionally, it aids in the creation of ethylene.
- Propane is a saturated hydrocarbon that is utilised as a propellant in several aerosol sprays. Hot air balloons may also run on this substance as fuel.
- Because it helps avoid engine damage, octane is a crucial part of gasoline.
- Additionally, cycloalkanes are used in heavy oils, diesel, gasoline, and other transportation fuels.
- The utilisation of cycloalkanes is also involved in the production of nylon and rubber.
- Cycloalkanes are employed as an organic solvent in the manufacture of medicines for use in medical applications.
- These are employed in both the food industry and the production of hair products.
- In the medical profession, cycloalkane known as cyclopropane is employed as an anaesthetic drug.
- Cancer patients are treated with carboplatin, which is made from cyclobutane.
- Certain kinds of cycloalkanes are employed in the pigmentation process as well as in the production of perfumes.
- The tissues of plants and animals contain some of these saturated hydrocarbons as steroids.
Difference between Saturated Hydrocarbon & Unsaturated Hydrocarbons
At least one double or triple bond exists between two neighbouring carbon atoms in unsaturated hydrocarbons. The following table lists the main distinctions between saturated and unsaturated hydrocarbons.
|
Saturated Hydrocarbon |
Unsaturated Hydrocarbons |
|
In these compounds, all carbon atoms are sp3 hybridised. |
They include hybridised carbons made of sp2 or sp. |
|
If compared to the comparable unsaturated hydrocarbons, contain more hydrogen atoms. |
If compared to the comparable saturated hydrocarbons, contain fewer hydrogens. |
|
Alkanes and cycloalkanes are two examples. |
Alkenes, alkynes, and aromatic hydrocarbons are examples. |
|
Their reactivity to chemicals is generally minimal. |
Compared to their saturated counterparts, they are more responsive and reactive. |
|
Typically, they burn with a blue flame. |
These often have a sooty flame when burning. |
Practice Problems
Q.1. What is the hybridisation of C-atoms in cycloalkanes?
A. sp2
B. sp3
C. sp
D. dsp2
Answer: (B)
Solution: Cycloalkanes are composed of C-C single covalent bonds. Hence they are sigma bonds. So the hybridisation of carbon atoms is sp3. So, the correct answer is option (B).
Q.2. Cycloalkanes are:
A. Non-polar
B. Hydrophilic in nature
C. Serve as an inorganic solvent
D. Highly Stable
Answer: (A)
Solution: Cycloalkanes are non-polar owing to the low electronegativity difference between carbon and hydrogen. Hence they are rather hydrophobic and also better serve as an organic solvent. So only option A is correct.
Q.3. Saturated hydrocarbons consist of:
A. Ionic Bonds
B. Covalent Bonds
C. Covalent Bonds
D. Triple Bonds
Answer: (B)
Solution: Saturated hydrocarbons are composed of carbon-carbon and carbon-hydrogen single covalent bonds. As there is sharing of electrons between them. Hence only sigma bonds are formed. So option B is the correct answer.
Q.4. Saturated hydrocarbons generally burns with a ______ flame.
A. Blue
B. Green
C. Red
D. Black
Answer: (A)
Solution: Because saturated hydrocarbons have a lower carbon concentration and higher hydrogen content as compared to unsaturated ones, they undergo complete combustion and hence produce a blue flame. Incomplete combustion occurs with unsaturated hydrocarbons because of their high carbon concentration and low hydrogen level. Unsaturated hydrocarbons burn with a sooty flame.
So option A is the correct answer.
Frequently Asked Questions–FAQs
Q1. Mention a few natural sources of saturated hydrocarbons along with an example of the daily used product.
Answer: Natural sources of hydrocarbons are coal, petroleum and natural gas and oils derived from fossil fuels. Lighter fluid is actually composed of n-butane and iso-butane. Beeswax produced by bees is composed of high molecular-mass based saturated hydrocarbons. It forms an integral component of high-end lip care products.
Q2. How can we convert unsaturated hydrocarbons to saturated hydrocarbons?
Answer: By various catalytic processes of hydrogenation (reduction) of unsaturated hydrocarbons, multiple bonds of unsaturated hydrocarbons can be reduced to single covalent bonds to form saturated hydrocarbons.
Q3. Are saturated hydrocarbons and saturated fat the same?
Answer: Saturated fats are basically fatty acids of long chain saturated hydrocarbons. Hence they are not exactly the same but have a long saturated hydrocarbon chain in common. Saturated fats are long chain fatty acids (having carboxylic acid functional group) along with long chain carbon-carbon and carbon hydrogen framework (like in saturated hydrocarbons).
Q4. How are saturated fats harmful to health?
Answer: It is challenging for the digestive enzymes in our body to function and break down saturated hydrocarbons, because they contain lengthy, full valence bonds, making them relatively unreactive. They continue to exist in their original form as a consequence, which causes the adipose tissues to build up fat gradually and leads to weight gain. Additionally, this causes plaque to build up in the arteries and veins, which eventually causes them to get blocked and result in cardiac ailments.


