-
Call Now
1800-102-2727
Reactions of Saturated Hydrocarbons: Introduction, Physical Properties, Chemical Reactions, Applications, Practice Problems & FAQs
LPG cylinders have become the essential source of energy for cooking in urban and semi-urban kitchens.
But, do you know what is inside those LPG cylinders - a gas or a liquid?
LPG standing for liquified Petroleum Gas is a cooking gas primarily composed of propane and butane which are saturated gaseous hydrocarbons.

Methane gas, a saturated hydrocarbon, is created during the digestion of commercial and residential garbage by microorganisms. Electricity can be produced by the burning of this methane gas.
Hexane and other alkanes are utilised in adhesives for shoes and leather goods, while butane is used in aerosol spray cans. Gasoline, which is used as a fuel, contains heptane as well as other alkanes including octane and decane.
Different hydrocarbons are all mixed together to form coal, petroleum, and natural gas. They serve as fuel for the production of energy. Furthermore, they are employed in the production of chemicals that are useful in both industry and daily life.
Table of Contents:
- Introduction of Saturated Hydrocarbons
- Physical Properties of Saturated Hydrocarbons
- Chemical Reactions of Saturated Hydrocarbons
- Applications of Saturated Hydrocarbons
- Practice Problems
- Frequently Asked Questions(FAQs)
Introduction of Saturated Hydrocarbons:
An organic compound composed exclusively of hydrogen and carbon is known as a hydrocarbon.
Organic molecules known as saturated hydrocarbons not only contain hydrogen and carbon but, importantly, all the carbon valencies appear to have been satisfied by hydrogen atoms.
Saturated hydrocarbon can be visualised in many ways-
- Where all of its carbon-carbon bonds are single bonds.
- Has no carbon-carbon multiple bonds
- Each of their carbon atoms is attached to four other atoms or
- That only includes sp3 hybridised carbon atoms.
They are called ‘Alkanes” and have a general formula of CnH(2n+2)
Cycloalkanes are also alkanes with a single monocyclic ring. Due to the fact that every carbon-carbon linkage in cycloalkanes is a single bond, they are categorised as saturated hydrocarbons.
The general formula for a saturated cyclic hydrocarbon is Cn H(2n + 2 - 2r), where r is the sum of all the rings in the molecule.
All the bonds in saturated hydrocarbons are -covalent bonds.

The examples below demonstrate how every molecule has a carbon-carbon single bond that completely fills its carbon valencies, leaving only hydrogen atoms to fill the remaining carbon valencies.
Saturated hydrocarbons are classified into three types based on whether their structure is linear, branched, or ring-shaped:
The carbon chains of alkanes can be either straight or branched. All the carbon atoms in these saturated hydrocarbons are sp3 hybridised.
Alkanes' successive homologues only differ from one another in the chemical formula by a -CH2.
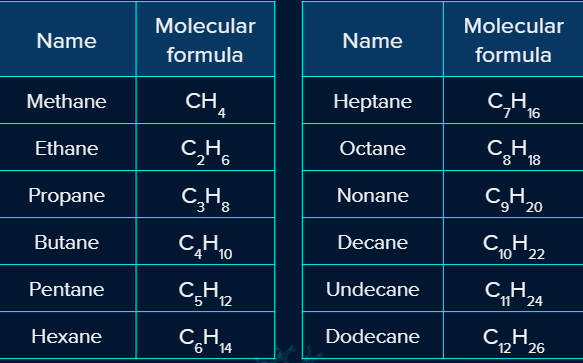
Examples:
CH3-CH3 (Ethane), CH3-CH2-CH3 (Propane), and CH3-CH2-CH2-CH3 (Butane).
Let's look at a few examples of straight chain alkanes:
In hydrocarbons with branched chains, one or more carbon atoms from the parent chain are joined by shorter chains of carbon atoms.
It's important to keep in mind that even polycyclic alkanes, or alkanes with many rings, are classified as cycloalkanes and, as a result, belong to the class of saturated hydrocarbons. The rings of cycloalkanes also contain sp3 hybridised carbon atoms.
Examples: Cyclopropane (C3H6), Cyclobutane (C4H8), Cyclopentane (C5H10), Cyclohexane
(C6H12).
Physical Properties of Saturated Hydrocarbons:
1. Boiling point: Boling point generally increases with the molecular weight of the hydrocarbons. Among the similar molecular weight hydrocarbons, linear hydrocarbons have a higher boiling point than branched hydrocarbons due to close packing.
Among the homologues series, hydrocarbons having up to four carbons are gases, fifth to thirteenth are liquids and above are solids and generally referred to as paraffin.
2. Melting point: Alkanes' melting points generally follow the same pattern as their boiling points do. With an increase in molecular mass, alkanes' melting points rise.
Alkanes have different melting points depending on their size, shape, and molecular packing. As a result, it has been found that even-numbered alkanes have a greater melting point trend than odd-numbered alkanes.
3. Solubility: Alkanes are a type of non-polar molecule because of the extremely low electronegativity between carbon and hydrogen as well as the covalent nature of the C-C and C-H bonds.
Alkanes are not miscible in polar solvents such as alcohol, water etc. but are extremely miscible in non-polar solvents such as ether, benzene, carbon tetrachloride, petroleum etc.
4. Density: When the limit value of 0.8 g cm-3 is achieved, all alkanes are lighter than water since the densities of alkanes grow as the molecular masses rise.
Chemical Reactions of Saturated Hydrocarbons:
The types of reactions that alkanes undergo are as follows:
- Controlled Oxidation
- Combustion Reaction
- Halogenation Reaction
- Nitration Reaction
- Sulphonation Reaction
- Aromatization
- Isomerization
- Reaction with steam
- Pyrolysis
Controlled Oxidation:
Alkanes produce a wide range of oxidation products when heated at high pressure with a controlled supply of oxygen or air and the appropriate catalysts.
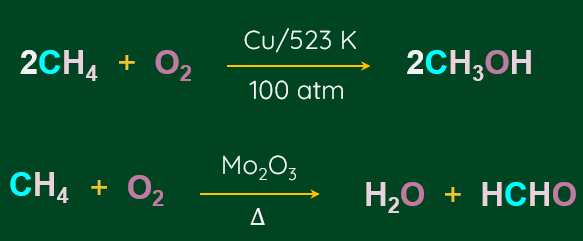
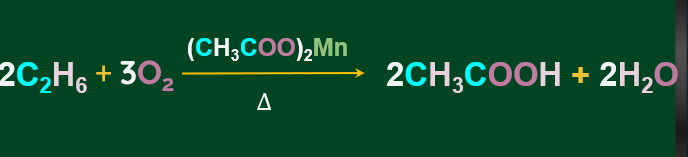
In general, alkanes are resistant to oxidation in the presence of oxidising agents like potassium permanganate (KMnO4), although potassium permanganate (KMnO4) can convert some alkanes with tertiary H-atoms to alcohol.
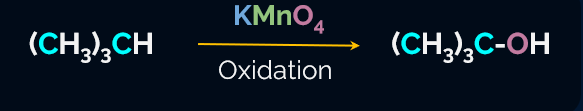
Combustion Reaction:
It involves heating a substance in the presence of enough oxygen and air. The material entirely oxidised to carbon dioxide (CO2) and water (H2O) while also releasing a significant quantity of heat.
The majority of combustion processes are exothermic in nature.

For combustion, the standard reaction is
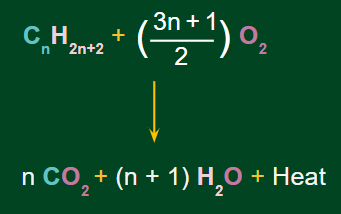
The example of a combustion reaction:
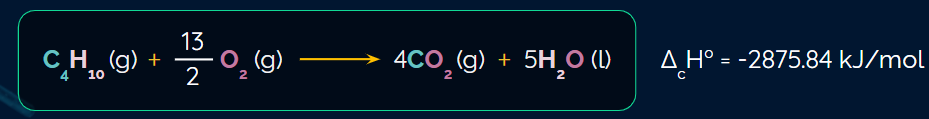
When alkanes burn without enough oxygen, carbon monoxide (CO) instead of carbon dioxide (CO2) is produced.
The quantity of heat produced when a substance is exposed to combustion is known as the heat of combustion.
The following are some factors that impact the heat of combustion:
1. Because of the differences in alkane stability, the heat of combustion is inversely proportional to alkane branching (in isomers). The greater the thermodynamic stability of an alkane, the lower the heat of combustion.
Examples:
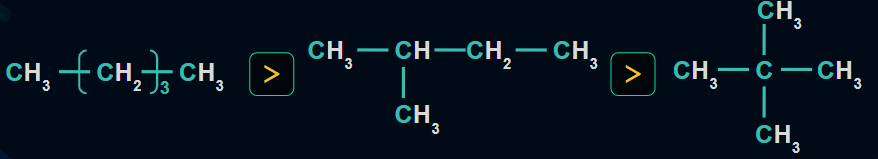
2. The number of carbon atoms is directly proportional to the heat of combustion.
Examples:
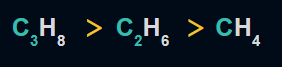
3. The heat of combustion per -CH2 group is inversely related to the size of the ring. When the ring size is increased from 3 to 6, the ring's stability rises, while the heat of combustion (its value) reduces per -CH2 group.
Examples:

Halogenation reaction:
When one atom or a group of atoms in a chemical are replaced by another atom, a substitution process takes place (or group of atoms). Substitution reactions are single displacement reactions.
Saturated hydrocarbons only have a single carbon-carbon bond, making them very inert. Saturated hydrocarbons undertake substitution processes but are often unreactive and do not react with many chemicals.
A hydrocarbon's one or more hydrogen atoms are substituted by other atoms in the substitution reaction of an alkane. Halogenation is the name used to explain the process of substitution by halogen. In the presence of UV light, this reaction occurs.
Rate of reaction of alkanes with Halogens:

While iodination is extremely slow and reversible and occurs in the presence of oxidising substancesHIO3/HNO3, fluorination of an alkane is explosive, violent, and uncontrollable. Alkanes, however, easily undergo bromination and chlorination.
Alkyl halides (RX) and hydrogen halides are created when saturated hydrocarbons such as alkanes or cycloalkanes are halogenated.

Where, X2= Cl2 or Br2
The reaction typically occurs by a free-radical pathway, and if left uncontrolled all of the alkane's hydrogen atoms will be replaced by halide ions.
Nitration Reaction:
Nitration of alkanes is the mechanism where the hydrogen atom of an alkane is replaced with a nitro group (NO2). There are two ways to do it:
(i) Liquid phase nitration: This technique involves heating higher alkanes under pressure at 413 K with fuming nitric acid.
(ii) Vapour phase nitration: When alkanes and nitric acid vapours are heated together in a gaseous composition at 673K - 773 K, lower members of the alkane family can be nitrated.
Sulphonation Reaction:
Sulphonation of alkanes is the process by which a sulphonic acid group (SO3H) replaces the hydrogen atom in an alkane.
The process involves heating higher alkanes with fuming sulfuric acid.
Aromatization:
Each of the n-terminal hexane's carbon atoms undergoes the removal of one hydrogen atom in this reaction, resulting in the formation of cyclohexane. This reaction is called cyclization.
Next, three hydrogen molecules are removed from the cyclic ring atoms to create benzene. The reaction is known as aromatization. Reforming is another name for the n-alkane aromatization process.
Aromatization of alkane can be achieved by utilising Cr2O3/V2O5/Mo2O3 at 10-20 atm pressure.
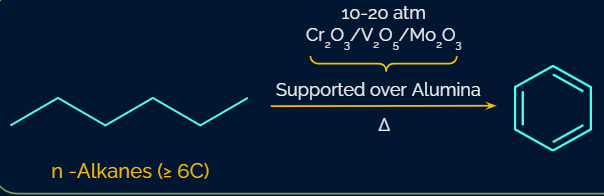
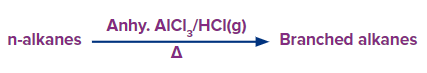
Isomerization:
The process of isomerization involves changing one isomer into another isomer. Straight chain alkanes are transformed to branched-chain alkanes by heating with Aluminium trichloride and Hydrochloric acid (AlCl3+ HCl) at about 473 K and 35 atm pressure.
Reaction with Steam:
At 1270 K methane combines with steam to generate carbon monoxide and dihydrogen in the presence of a nickel catalyst. This approach is utilised in the industrial synthesis of H2. The mixture produced here is known as Syn gas which is used for chemical synthesis.

Pyrolysis:
Pyrolysis, which differs from combustion, is the process of breaking down a compound into smaller pieces by applying heat but in the absence of air. Because there is no air present, no compound oxidation takes place. Alkane pyrolysis is commonly referred to as cracking.
The higher alkanes are often heated to high temperatures under pressures of 6-7 atm, either with or without the aid of a catalyst. The following is an example of the corresponding chemical reaction:
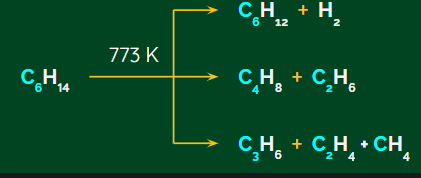
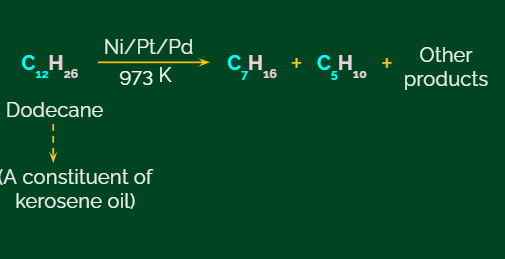
An example of using this chemical process of catalysed pyrolysis to produce oil gas from kerosene is given below.
Applications of Saturated Hydrocarbons:
- For a long time, hydrocarbons have played a vital role in our daily life.
- CNG, which we utilise as a transportation fuel, is mostly composed of alkanes methane and ethane. Similarly, LPG or cooking gas is primarily composed of propane and butane.
- In its simplest form, methane gas is fuel for cars, heaters, and ovens. Highly refined methane is utilised as liquid rocket fuel.
- Most often, saturated hydrocarbons are used as a source of combustible fuel. The main ingredients in gasoline, naphtha, jet fuel, and specialised industrial solvent combinations are alkanes and cycloalkanes.
- Several cryogenic refrigeration systems employ ethane as a refrigerant. Ethylene gas is additionally produced using it.
- Propane is used as a fuel for hot air balloons and as a propellant in a variety of aerosol sprays. Additionally, it is employed in the creation of propylene gas.
- Mobile stoves, camp warmers, and lighters all run on butane as fuel. Additionally, it serves as the propellant for deodorants. In order to avoid engine damage, octane is typically blended with gasoline.
- Automobile fuels, diesel, petroleum gas, and other heavy oils are the principal uses for cycloalkanes.
Practice Problems:
Q1. Aromatization of n-heptane gives
(A) Toluene
(B) Benzene
(C) Cyclo benzene
(D) None of the above
Answer: (A)
Solution: In the presence of oxides of vanadium (V)Molybdenum (Mo) and Chromium (Cr), n-heptane undergoes dehydrogenation and cyclization to create toluene when heated to 773 K and 10-20 atm of pressure. It doesn't create a ring with seven members.
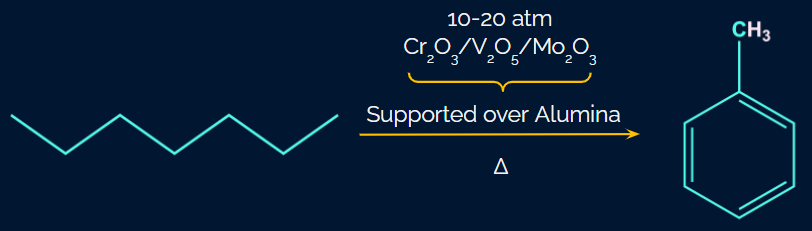
Q2. Saturated hydrocarbons burn with
(A) Blue flame
(B) Yellow flame
(C) Red flame
(D) None of these
Answer: (A)
Solution: Unsaturated carbon compounds do not burn entirely and produce a flame containing unburnt or partially burned carbon particles. Such a flame is polluting and has a yellow colour. It is referred to as sooty flame.
While saturated carbon compounds often burn completely and produce a clear blue flame. This is referred to as a non-sooty flame.
Q3. Gaseous hydrocarbon is produced from a liquid hydrocarbon by
(A) Cracking
(B) Hydrolysis
(C) Oxidation
(D) Distillation
Answer: (A)
Solution: The hydrocarbon with a greater molar mass generates a mixture of hydrocarbons with a lower molar mass during cracking or pyrolysis. Thus, we can conclude that a liquid hydrocarbon can be transformed into a mixture of gaseous hydrocarbons via cracking.
Q4. Branched-chain alkanes are also known as
(A) Paraffins
(B) Isoparaffins
(C) Naphthenes
(D) Neoparaffins
Answer: (B)
Solution: Isoparaffins are branched-chain alkanes. A general name like "paraffin" might make it difficult to distinguish between pure substances and mixes of isomers, or substances with the same chemical formula, as pentane and isopentane.
Frequently Asked Questions(FAQs):
Q1. Why do alkanes have the name "paraffin"?
Answer: Alkanes have only single covalent bonds. These bonds are quite strong and difficult to break, and breaking one demands a significant amount of energy. Because of this, alkanes are relatively stable and thus inert compounds.
As a result, alkanes are referred to as paraffin since they have a lower affinity for common reagents. In other words, they are inert and inactive.
Q2. Why do unsaturated hydrocarbons react more readily than saturated hydrocarbons?
Answer: The unsaturated hydrocarbons, due to the double and triple bonding of the carbon atoms, are weaker than the single bonded saturated hydrocarbons. Due to the presence of weaker pi bonds, the unsaturated hydrocarbons are more reactive and react faster when used in a reaction. As a result, when a reaction occurs, these unsaturated hydrocarbons break down more readily than single-bonded carbon compounds.
Q3. When compared to cyclopentane and cyclohexane, why do cyclopropane and cyclobutane have a higher strain?
Answer: The carbons in cycloalkanes are sp3 hybridised, which means they do not possess the predicted ideal bond angle of 109.5o; this results in a rise in potential energy due to the need for the carbons to be at an ideal 109.5o. Ring strain can be illustrated in the cyclopropane where the bond angle between the carbons is 60o.
The tetrahedral carbon model demonstrates the cause of ring strain. Cyclopropane (60o) and cyclobutane (90o) have C-C-C bond angles that deviate significantly from the optimum bond angle of 109.5o. Cyclopropane and cyclobutane have a high ring strain as a result of this bond angle. However, since the bond angle between the carbons is much closer to 109.5o in compounds like cyclohexane and cyclopentane, they would have a significantly smaller ring strain.
Q4. What causes cycloalkanes to be nonpolar?
Answer: Due to the minimal electronegativity gap between carbon and hydrogen, cycloalkanes are also non-polar and lack intermolecular hydrogen bonding. They are typically less dense than water and hydrophobic, which means they do not dissolve in water.


