-
Call Now
1800-102-2727
Rate of Radioactive Decay- Introduction, Disintegration Constant, Half-life, Average-life & Radioactive Equilibrium
You all must be aware of one of the most powerful countries in the world in terms of nuclear energy. But are you aware of the accident which occurred in 1986? Let me tell you one fact, the disaster I'm mentioning here is one of the worst disasters in nuclear power generation. The disaster occurred in Chernobyl in 1986 at the Chernobyl nuclear power plant in the soviet union. But what do you think might be the reason for such a disaster? This disaster was an eye-opener in terms of nuclear power generation where the radioactive sample gets decayed in a nuclear reactor to produce energy. So, let’s dive in deeper and understands how radioactive substances decay and how to calculate the rate at which they can decay.
Table of Contents
- Introduction to Radioactive Decay
- Disintegration Constant
- Half-life
- Average life
- Radioactive Equilibrium
- Practice Problems
- Frequently Asked Questions-FAQs
Introduction to Radioactive Decay
The decay of radioactive samples takes place by the disintegration of the atomic nucleus. Radioactive disintegration or radioactive decay is defined as the process in which unstable radioactive elements emit energy in the form of radiation to form comparatively stable nuclei.
For example,
The rate of disintegration of substances depends upon the nature of the disintegrating substance and its total amount. The rate of radioactive disintegration follows the first-order kinetics.
A first-order reaction is a chemical reaction in which the rate of reaction depends upon the first power of concentration of reactants.
For example,
Consider a reaction, A P
Mathematically, Rate = -d[A]dt ∝ [A]1
Rate = -d[A]dt = k[A]1= k[A]
Here, -d[A]dt refers to the rate of reaction and "k" is the rate constant for the first-order reaction.
Disintegration Constant
Radioactive disintegration is similar to a chemical reaction where unstable nuclides of one radioactive sample change to another type of nuclide and follow first-order kinetics.
Consider a nuclear reaction, where all the radioactive nuclides decay following first-order kinetics.
Let us consider the decay of a radionuclide A as:
AProduct
Suppose the number of radionuclides of the radioactive substance A present at the start of the observation (i.e., t=0) be NO and after the time (t), the number of radionuclides those remain undecayed be N. Considering particular instant of time, a small number of radionuclides dN disintegrate in small-time dt then,
Rate of change of A into products (Rate of decay)
Note: Here negative sign indicates a decrease in the number of radionuclides of A with the increase in time.
As we know, the rate of radioactive decay is proportional to the total number of atoms (radionuclides) at that time. Therefore,
Mathematically, Rate =
is called a decay constant or disintegration constant.
Decay constant (): It is defined as the fraction of the total number of the atoms (radionuclides) that disintegrates per sec at any time.
On rearranging the equation (i), we get;
Integrating on both sides from time t=0 to t.
NoNdNN =- 0t dt
Here, NO and N are the number of atoms (radionuclides) of A at t=0 and t.
Equation (ii) is the required integrated form of first-order radioactive disintegration.
Again. equation (ii) can be written as;
As in the equation (v) No represent the number of radionuclides of the radioactive substance A present at the start of the observation (i.e., t=0) and N represent the number of radionuclides that remain undecayed after time t. Therefore the ratio of No N will be unitless and decay constant () is inversely proportional to time. So,
The unit of the decay constant is s-1.
Note: The amount of radioactive sample can be taken in terms of the number of atoms, number of moles etc. in the radioactive disintegration process.
Graphical representation of log No N vs t curve
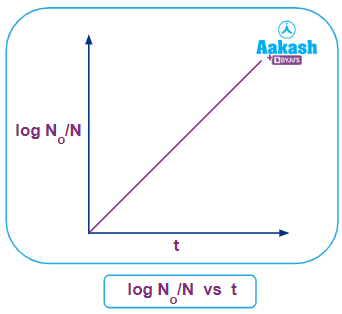
We know the general equation of a straight line is Y=mX+C.....(vi)
Where, 'Y' and 'X' represent the co-ordinate axis, 'm' represents the slope of the straight line and 'C' represents the intercept on Y-axis.
Now, considering the integrated form of radioactive disintegration equation;
Reorganising the above equation we get,
If we compare the equation (vi) and (vii), we can say;
Y-Intercept (C)=0 and Solpe (m)=2.303
Therefore, the above curve between log No N & t is a straight line with a constant slope and if this straight line is extrapolated it will pass through the origin.
Half-life (t1/2)
It is the time duration for the disintegration of half the amount of radioactive substance.
At, t= t1/2 , N = N0 2
Now, considering the integrated form of radioactive disintegration equation, we get;
Putting the value of time and the total amount of sample A at a time (t=t12) in equation (viii), we get;
t1/2 = 2.303 log No No/2
Plot of t1/2 vs N
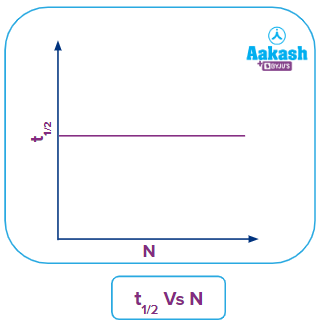
From the equation (ix), we can see that half-life ( t1/2) is independent of the amount of radioactive sample present and depends only on the decay constant. Therefore, the nature of the graph is a straight line parallel to X-axis.
Average Life
It is the ratio of the total life period of all the atoms (radionuclides) and the total number of the atoms (radionuclides).
Mathematically,
Average life
Average life
Thus, the average life of a radioactive substance is reciprocal of the decay constant.
Average life
Putting the value of the decay constant () from equation (ix) in the equation (x), we get;
Average life
Radioactive Equilibrium
It is obtained when the rate of formation of an element and its further disintegration are the same.
Let us consider that a radioactive element C disintegrates to give D which is also radioactive and disintegrates into E as shown below,
CDE
The element D is said to be in radioactive equilibrium with C if its rate of formation from C is equal to the rate of decay of D into E.
Let 1 and 2 be the decay constant of C and D respectively and N1 and N2 be the number of atoms of radioactive elements C and D present at the equilibrium.
Then using the equation (i) we get,
Rate of formation of D = Rate of decay of C = 1N1
Rate of decay of D = 2N2
At radioactive equilibrium,
Rate of formation of D = Rate of decay of D
1N1=2N2
Putting the value of average life in equation (xii) from equation (xi), we get;
When the value of the decay constant of parent atom (C) is less than the value of decay constant of daughter element (but both are not too small), then the transient equilibrium is reached when;
Practice Problems
Q. If the half-life of a radioactive element X is 8 hours. Select the correct option for the approximate value of the decay constant of the given radioactive sample.
- 1.44102 min-1
- 1.4410-2min-1
- 1.4410-3min-1
- 1.44103min-1
Answer: C
Solution: According to the given data,
Half-life of a radioactive sample = 8 hours=480 min
Let, Decay constant = min-1
As we know;
Half-life
Putting the value of t1/2 in the above equation, we get;
Q. The half-life of a radioactive sample is 2 hours. If 180 g of radioactive sample was taken initially. The mass of radioactive sample that remained undecayed after 10 hours is______ g.
Answer: 5.55 g
Solution:
According to the given data,
Half-life of the radioactive sample = 2 hours=120 min
Initial mass of the radioactive sample (No) = 180 g
Time taken for the sample to decay = 10 hours=600 min
Let, the mass of radioactive sample left after 10 hours (N)=x g
Let, the decay constant be min-1.
As we know;
Half-life
Putting the value of t1/2 in the above equation, we get;
As we know;
Putting the value of in the above equation and solving the equation, we get;
Taking antilog both the side and solving it, we get;
X5.55 g
Q. If 10 g of radioactive element X undergoes radioactive disintegration and forms Y. How much time (hours) will be required to disintegrate 30% of a radioactive sample X. Given the half-life of the radioactive element X is 5 hours.
Answer: 155 min
Solution:
According to the given data,
Half-life of the radioactive sample = 5 hours=300 min
Initial mass of the radioactive sample (No) = 10 g
Let, time taken for the sample to decay = t min
Mass of the radioactive sample left after 5 hours (N) = 7 g
Let, decay constant = min-1
As we know that,
Half-life
Putting the value of t1/2 in above equation, we get;
As we know;
Putting the value of in the above equation and solving the equation, we get;
t155 min
Q. If the disintegration constant of an unknown radioactive sample is 1.5410-8 year-1. Calculate the half-life of the radioactive sample in (years).
Answer: 4.5107 years
Solution: According to the given data,
Disintegration constant of an unknown sample () = 1.5410-8year-1
Let, the half-life of the unknown sample be t1/2 year.
As we know that,
Half-life
Putting the value of disintegration constant of an unknown sample () in the above equation, we get;
Half-life
Frequently Asked Questions-FAQs
Q. What is the time of completion of a sample undergoing radioactive decay?
Answer: When the reaction undergoes radioactive decay, it will take infinite time for a radioactive sample to decay completely.
To prove it let’s assume;
At the time of completion time, reactants are consumed completely such that, N= 0, substituting this value in the equation, N =NO e-t
Thus, radioactive disintegration takes infinite time for completion and never completes.
Q. What is the effect of temperature on the decay constant of the radioactive samples?
Answer: According to the research it was found that the degradation rate was temperature independent. Since then, various studies have proven that external variables like temperature, air pressure, or the surrounding material have no effect on alpha and beta decay. Electron capture decays, on the other hand, are known to be vulnerable to their surroundings and have thus been classified as a separate category.
Q. What is the significance of the half-life of a radioactive sample?
Answer: Half-lives are essential because they can help you determine whether a sample of radioactive material is safe to handle. The radioactivity of a sample is deemed safe when it goes below detection limits.
Q. How can we differentiate the half-life of a radioactive substance and the average life of a radioactive sample?
Answer: The half-life of a radioactive substance is the time during which half the amount of a given sample of radioactive substance disintegrates. Whereas, the average life of a radioactive sample denotes the average estimated life span of the radioactive sample.


