-
Call Now
1800-102-2727
Preparation of Polyethene With Mechanism- Introduction of Polyethylene, Preparation of Polyethene, Mechanism of Polymerization of Ethylene, Application of Polyethylene, Practice Problems and FAQs
Our civilization has evolved from stone age, to now digital age. There was one time when plastic- a polymer was considered as boon and occupied every aspect or use of the world. Plastics are of many varieties manufactured to suit the purpose of intended use- from the light weight milk cover to chemical reaction tanks
When you go to the shop to buy a soft drink bottle, do you know what is used to make soft drink container?
Though the flavors of the soft drinks differ, they are packaged in a similar type of bottle made of polyethylene.
Polyethylene or Polythene (PE) is a light, versatile synthetic resin derived from the polymerisation of ethylene. It is the most widely used plastic in the world, with applications ranging from clear food wrap and grocery bags to detergent bottles and automobile fuel tanks.
But,if polyethylene is not recycled, its widespread use complicates waste management. Polyethylene, like other synthetic plastics, is difficult to biodegrade and thus accumulates in landfills.
In this article, we will learn about the process of producing polyethylene.
Table of Content
- Introduction of Polyethylene
- Preparation of Polyethene
- Mechanism of Polymerization of Ethylene
- Application of Polyethylene
- Practice Problems
- Frequently Asked Questions-FAQs
Introduction of Polyethylene
Polyethylene, also known as polythene or polyethene, is one of the world's most widely used plastic materials. Polyethylenes are addition polymers that typically have a linear structure. These synthetic polymers are primarily used in packaging. Polyethylene is frequently used in the manufacture of plastic bags, bottles, plastic films, containers, and geomembranes. It should be noted that over 100 million tonnes of polyethene are produced each year for both industrial and commercial use.
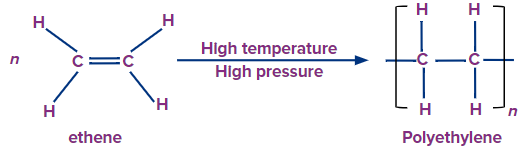
Polyethylene is represented by the general formula (C2H4)n. The majority of polyethylenes are thermoplastic (they can be remolded by heating). Some modified polyethylene plastics, on the other hand, have thermosetting properties. Cross-linked polyethylene is an example of this type of polyethylene (often abbreviated to PEX).
Preparation of Polyethylene
Ethylene or ethene has a molecular formula C2H4. Each carbon shares two hydrogen atoms and a double bond between them. The pi-bond of the double bond is weak and ready to open up and bond with other atoms or groups. This unsaturation of double bond property is useful in the polymerization of ethene.
Polymerisation of polyethylene takes place when large number of ethene molecules combines together. This type of polymerisation reaction is known as addition polymer In addition polymerisation, molecules of the same or different monomers add on to one another, resulting in the formation of a macromolecule/polymer. The repeating unit of the polymer has the same molecular formula as the parent monomers. The most common type of addition polymerisation occurs in compounds with multiple bonds. This type of polymerisation is also known as chain growth polymerisation because it results in an increase in chain length.
Mechanism of polymerization of Ethylene
Polyethylene represents one of the most important polymeric materials that can be produced through free radical polymerisation. The process of free radical polymerisation consists of three steps, which include:
- Chain initiation step
- Chain propagation step
- Chain termination step
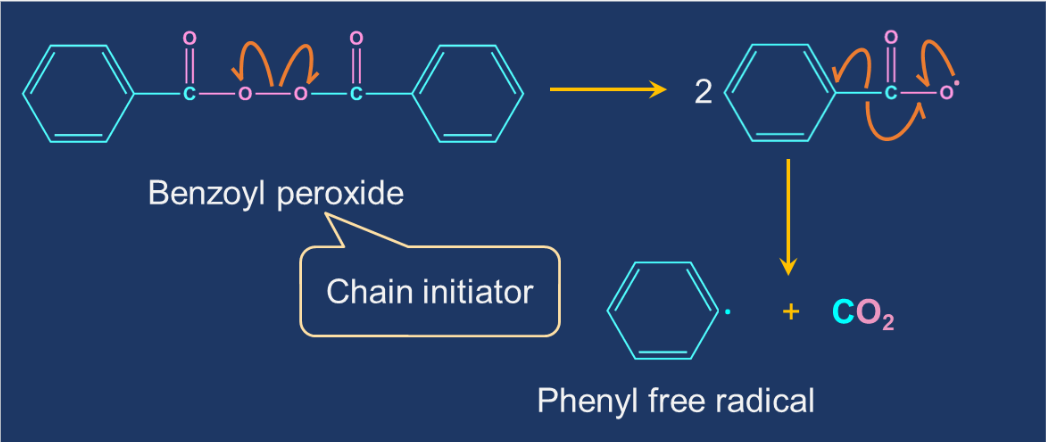
Chain initiation step: The initiator molecule in free-radical polymerisation is a free radical. A polymerisation initiator is a molecule that starts the process of polymerisation. The most common free-radical initiators are tert-butyl peroxide and benzoyl peroxide. These initiators decompose into tert-butoxide and benzoyl peroxide under mild conditions. These reactive intermediates combine with the monomer to create a larger free-radical molecule.
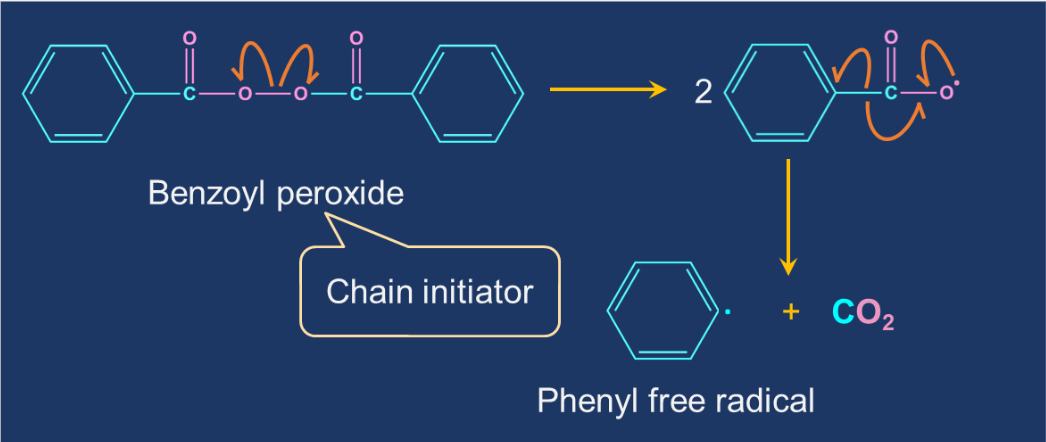
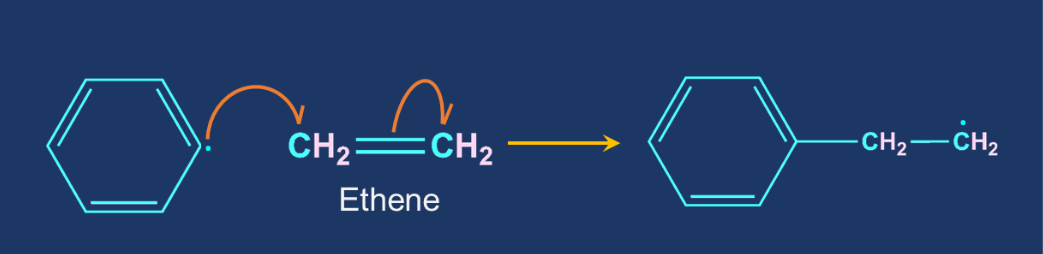
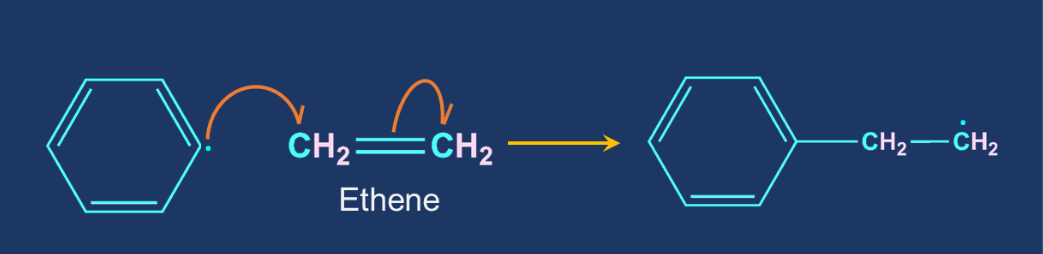
Chain Propagation Step: The free radical formed in the chain initiation step react with the ethene molecule to generate new free radical. The products are formed by the repeated addition of monomers to reactive intermediates. The reaction temperature, catalyst type, and radical concentration can all influence the number of ethylene monomer units and thus the molecular weight of the polyethylene molecule.
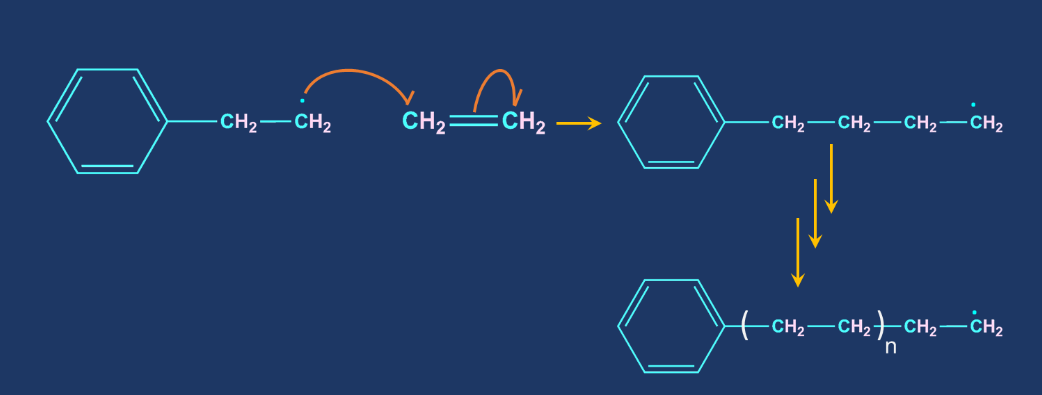
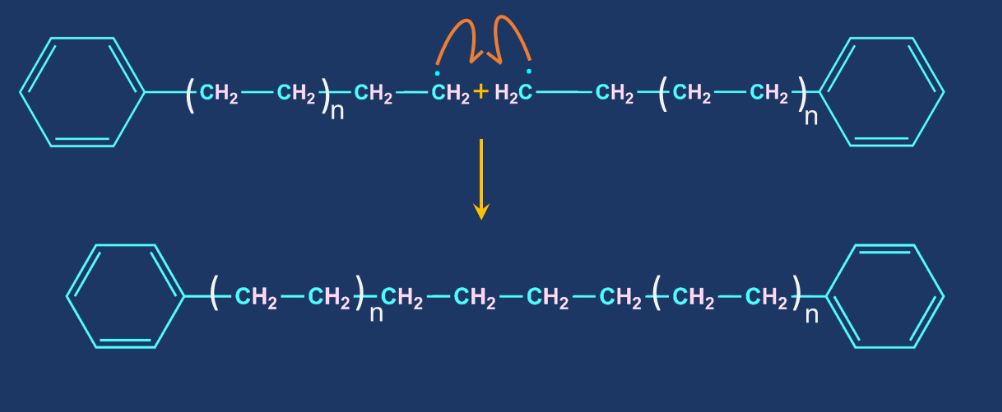
Chain termination step: When one radical combines with another radical, the chain is terminated. It is carried out in the presence of a benzoquinone inhibitor. Radical reaction inhibitors, also known as radical inhibitors, are compounds that have the ability to remove chain-carrying molecules and thus terminate the radical chain reaction.
Applications of Polyethylene
- Polyethylene's most important application is in packaging. This plastic is frequently used to make plastic bags, plastic films, bottles, geomembranes, and containers.
- Polyethylene is also found in crates, trays, milk or fruit juice jugs, and other food packaging products.
- Toys, garbage cans, ice trays, and other household items are made of high-density polyethylene. Because of its versatility, this plastic is suitable for a wide range of applications.
- Ropes, fishing nets, agricultural nets, and industrial fabrics are also made from HDPE. This plastic is frequently used in wirings and cables.
- Due to its high flexibility, low cost and low-density polyethylene (LDPE) is widely used in the production of squeeze bottles, garbage bags, laminations, and food packaging
Practice Problems
Q1. Which among the following mechanism followed in case of polyethylene formation?
- Polymerisation by nucleophilic attack
- Polymerisation by electrophilic substitution method
- Polymerisation by Carbonyl substitution method
- Free radical polymerisation method
Answer: (D)
Solution: Polyethylene represents one of the most important polymeric materials that can be produced through free radical polymerisation. The process of free radical polymerisation consists of three steps, which include:
- Chain initiation step: The initiator molecule in free-radical polymerisation is a free radical. A polymerisation initiator is a molecule that starts the process of polymerisation. The most common free-radical initiators are tert-butyl peroxide and benzoyl peroxide.
- Chain propagation step: The free radical formed in the chain initiation step react with the ethene molecule to generate new free radical. The products are formed by the repeated addition of monomers to reactive intermediates.
- Chain termination step: When one radical combines with another radical, the chain is terminated. It is carried out in the presence of a benzoquinone inhibitor. Radical reaction inhibitors, also known as radical inhibitors.
Therefore option (D) is correct.
Q2. Select the correct option for initiator used in the preparation of polyethylene by free radical mechanism.
- Benzoyl peroxide
- Benzoquinone
- Hydroxylamine
- Both C and D are correct.
Answer: (A)
Solution:The initiator molecule in free-radical polymerisation is a free radical. A polymerisation initiator is a molecule that starts the process of polymerisation. The most common free-radical initiators are tert-butyl peroxide and benzoyl peroxide. Incase of preparation of polyethylene benzoyl peroxide is used as an initiator. This initiator decompose into phenyl free radical under mild conditions which further generates a free radical when allowed to react with ethene molecule. Therefore, option (A) is correct.
Q3. Select the correct option for the use of inhibitors in the terminating step of the free radical mechanism.
- Benzoyl Peroxide
- Tert-butyl peroxide
- Benzoquinone
- Both A and B are correct
Answer: (C)
Solution: When one radical combines with another radical, the chain is terminated. It is carried out in the presence of a benzoquinone inhibitor. Radical reaction inhibitors, also known as radical inhibitors, are compounds that have the ability to remove chain-carrying molecules and thus terminate the radical chain reaction. Therefore, option (C) is correct.
Q4. Which among the given options represent the use of polyethylene?
- polyethylene (LDPE) is widely used in the production of squeeze bottles, garbage bags, laminations etc
- Ropes, fishing nets, agricultural nets, and industrial fabrics are also made from HDPE.
- Polyethylene is also found in crates, trays, milk or fruit juice jugs, and other food packaging products.
- All of the above
Answer: (D)
Solution: Few important uses of polyethylene includes:
- Polyethylene (LDPE) is widely used in the production of squeeze bottles, garbage bags, laminations etc
- Ropes, fishing nets, agricultural nets, and industrial fabrics are also made from HDPE.
- Polyethylene is also found in crates, trays, milk or fruit juice jugs, and other food packaging products.
Therefore option (D) is correct.
Frequently Asked Questions-FAQs
Q1. What are the different types of polythene?
Answer: Polyethylene can be divided into several types based on its density and the degree of branching in its structure. The following are some important polyethylene types:
- Ultra-high molecular weight polyethylene (UHMWPE)
- Ultra-low-molecular-weight polyethylene (ULMWPE)
- High-density cross-linked polyethylene (HDXLPE)
- High-density polyethylene (HDPE)
- High-molecular-weight polyethylene (HMWPE)
- Cross-linked polyethylene (XLPE)
- Medium-density polyethylene (MDPE)
- Low-density polyethylene (LDPE)
- Linear low-density polyethylene (LLDPE)
- Chlorinated polyethylene (CPE)
Q2. What is the impact of polyethylene on the environment?
Answer: Plastic bags have a significant environmental impact because they take many years to decompose. Furthermore, toxic substances are released into the soil when plastic bags decompose in sunlight, and when plastic bags are burned, a toxic substance is released into the air, causing ambient air pollution.
Q3. What is the difference between Low density polyethylene and high density polyethylene?
Answer:
|
High density polyethylene (HDP) |
Low density polyethylene (LDP) |
|
|
Polymerisation reaction |
|
|
|
Catalyst used |
The reaction is catalysed by the Ziegler-Natta catalyst {(C2H5)3Al and TiCl4} |
The reaction is catalysed by benzoyl peroxide. |
|
Temperature and pressure for reaction |
The reaction occurs at temperatures ranging from 333 K to 343 K and pressure ranging from 6 atm to 7 atm. |
The reaction occurs at temperatures ranging from 350 K to 600 K and pressure ranging from 1000 atm to 2000 atm. |
|
Properties |
They are unreactive, tougher and harder than LDP, and poor electrical conductors. |
They are unreactive, flexible, and poor electrical conductors. |
Q4. List some important characteristics properties of polyethylene.
Answer: Some important characteristics properties of polyethylene includes:
- Polyethylene has a lower mechanical strength than other plastics. These polymers' rigidity and hardness are also relatively low.
- Polyethylene is well-known for its ductility. This plastic is also known to have a high impact strength.
- Polyethylenes are typically waxy in texture.
- Commercial grades of high-density polyethylene (HDPE) and medium-density polyethylene (MDPE) have melting points ranging from 120 to 180 degrees Celsius.
- Polyethylene is well-known for being an excellent electrical insulator due to its high electrical treeing resistance.