-
Call Now
1800-102-2727
Orbitals-Definition, Shape of Orbitals, Boundary Surface Diagrams - Definitions, Formula, Practice Problems
Most of you would have come across a model of the solar system. The trajectories are followed by the planets around the sun. Did you know what the path was called?
It was called an orbit, a term coined by Johannes Kepler. The adjective "orbital" had the meaning "relating to an orbit".
However, with different models of the atom being proposed by Dalton, Rutherford and Bohr, it became quite clear that electronic orbits did not resemble planetary orbits at all. After Schrodinger had solved his wave equation (the basis of quantum mechanics) for the hydrogen atom, it was theorized that the electrons are spread out like unmoving clouds.
By solving Schrodinger equations, we get space (region) around the nucleus where the probability of finding an electron is maximum.
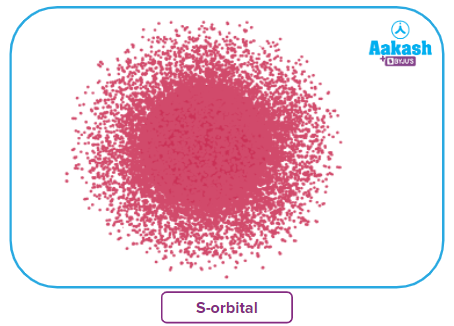
Table of contents
- Orbitals
- What are Nodes?
- Nodes of s orbitals
- Nodes of p orbitals
- Nodes of d orbitals
- Shape of orbitals
- Practice problems
- Frequently asked questions-FAQs
Orbitals
- Boundary surface diagram is a good diagrammatic representation of shapes of atomic orbitals.
- It is the resultant of the solution of Schrödinger wave equation.
- An atomic orbital is a space around the nucleus where the probability of finding an electron is maximum.
- Electrons are considered matter-wave (which have rest mass) that can be represented as a cloud rather than a point mass. In an electron cloud, the electron density is not equally distributed.
- The electron density varies with respect to the distance from the nucleus.
- Each orbital can only hold two electrons at a time. Orbitals also contain phases or signs that represent various aspects of the orbitals. The phases represent mathematical functions; however, they are typically depicted in orbital drawings with distinct coloured or shaded lobes.
What are Nodes?
- A node is a location where the chances of finding electron density are the least.
- The nodal plane is just the plane that goes through the nucleus and you cannot locate an electron on this plane.
Types of nodes:
- Angular nodes
- Radial nodes
Calculation of angular node = l (expressed in terms of Azimuthal quantum number)
Calculation of Radial node = n-l-1 (expressed in terms of principal & Azimuthal quantum number)
number of total nodes = n-l-1+l=n-1
|
Orbital |
Principal quantum number (n) |
Azimuthal quantum number (l) |
Radial node (n-l-1) |
Angular node (l) |
Total node (n-1) |
|
1s |
1 |
0 |
0 |
0 |
0 |
|
2s |
2 |
0 |
1 |
0 |
1 |
|
2p |
2 |
1 |
0 |
1 |
1 |
|
3p |
3 |
1 |
1 |
1 |
2 |
|
3d |
3 |
2 |
0 |
2 |
2 |
|
4s |
4 |
0 |
3 |
0 |
3 |
|
4p |
4 |
1 |
2 |
1 |
3 |
|
4d |
4 |
2 |
1 |
2 |
3 |
|
4f |
4 |
3 |
0 |
3 |
3 |
|
Maximum number of electrons present in a shell = 2n2 Number of orbitals present in nth shell = n2 Angular Momentum of any orbit = Number of orbitals in a subshell = 2l+1 Maximum number of electrons in particular subshell = 2 *(2l+1) Orbital Angular Momentum = |
|||||
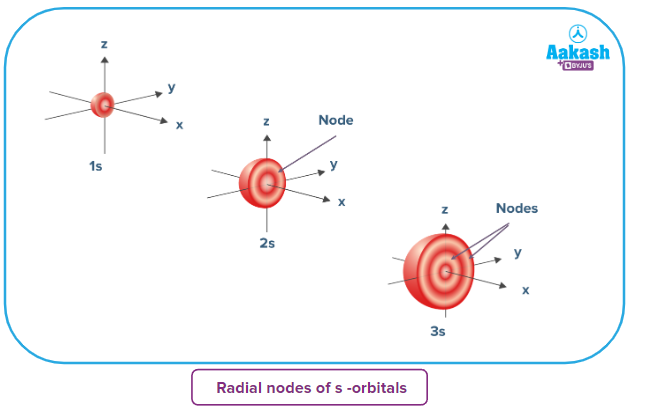
Radial nodes of s-orbitals
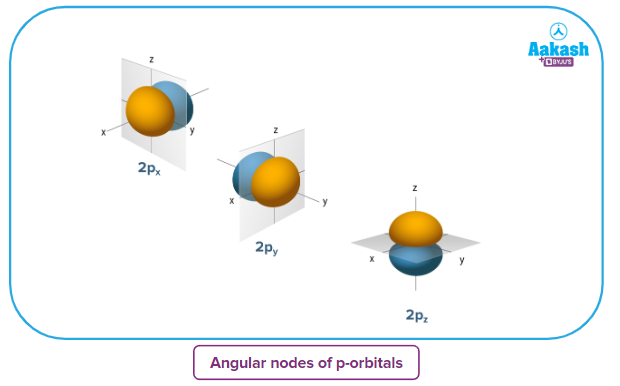
Angular nodes of p-orbitals
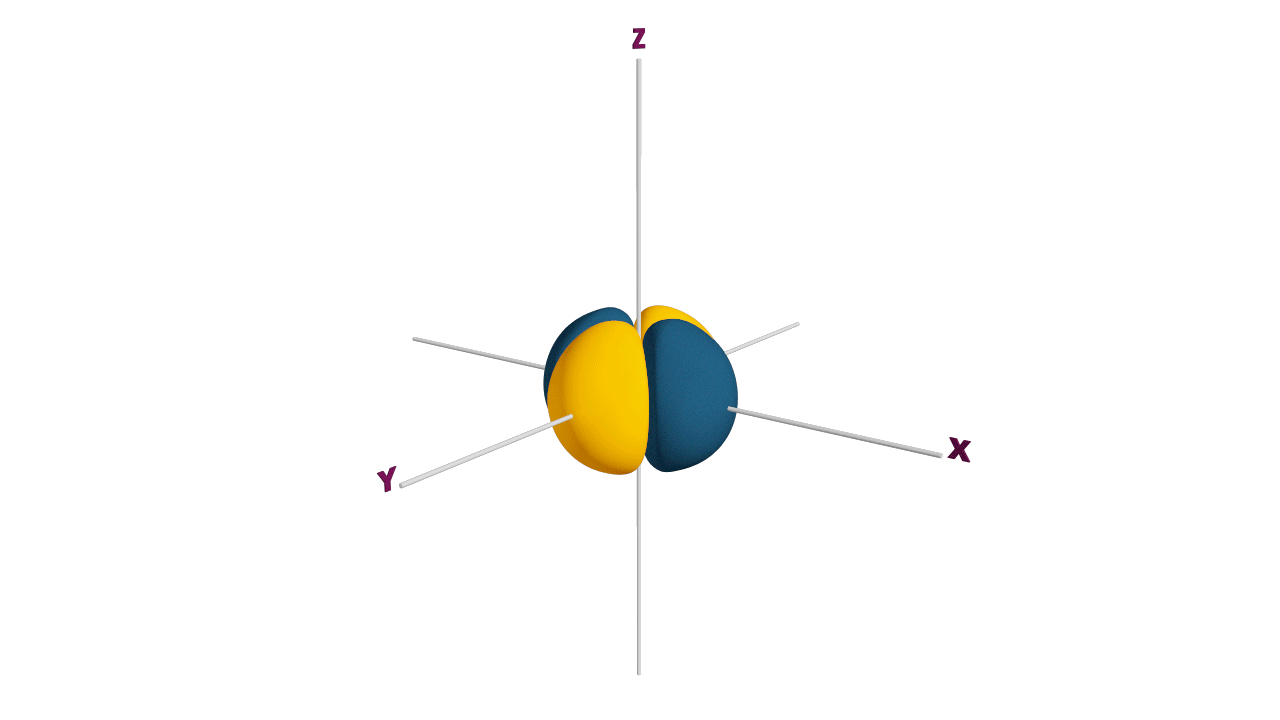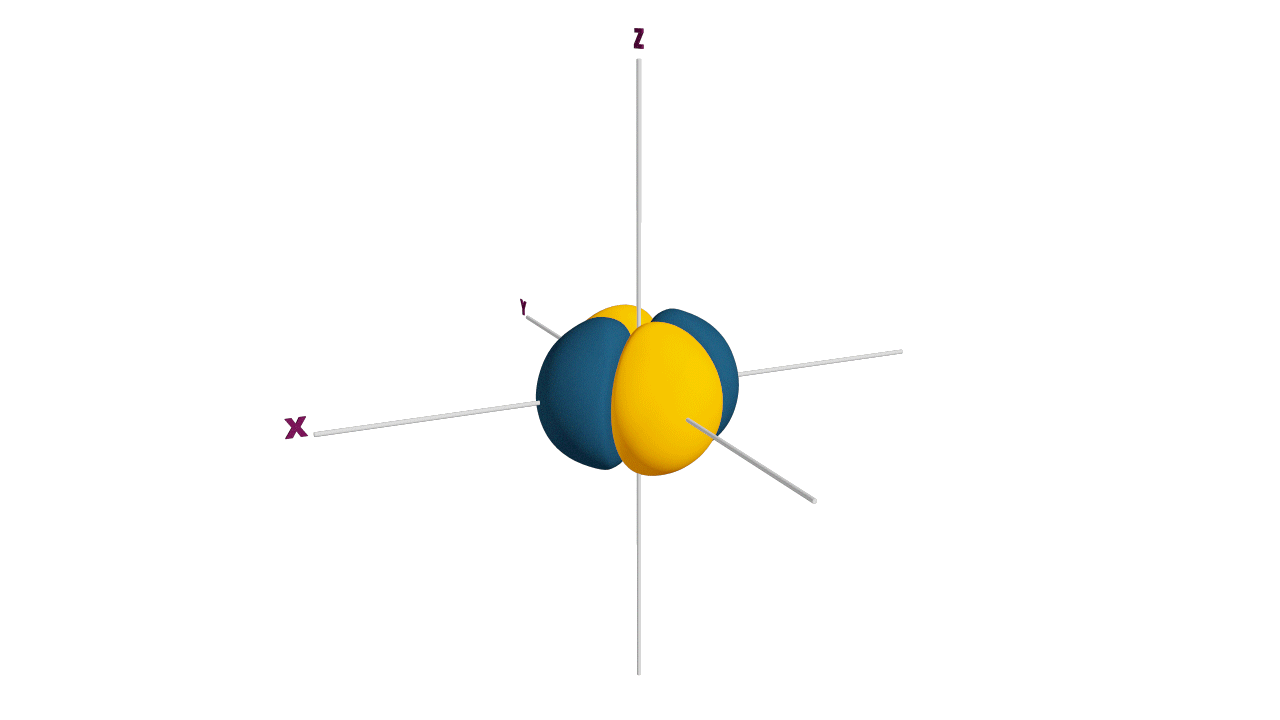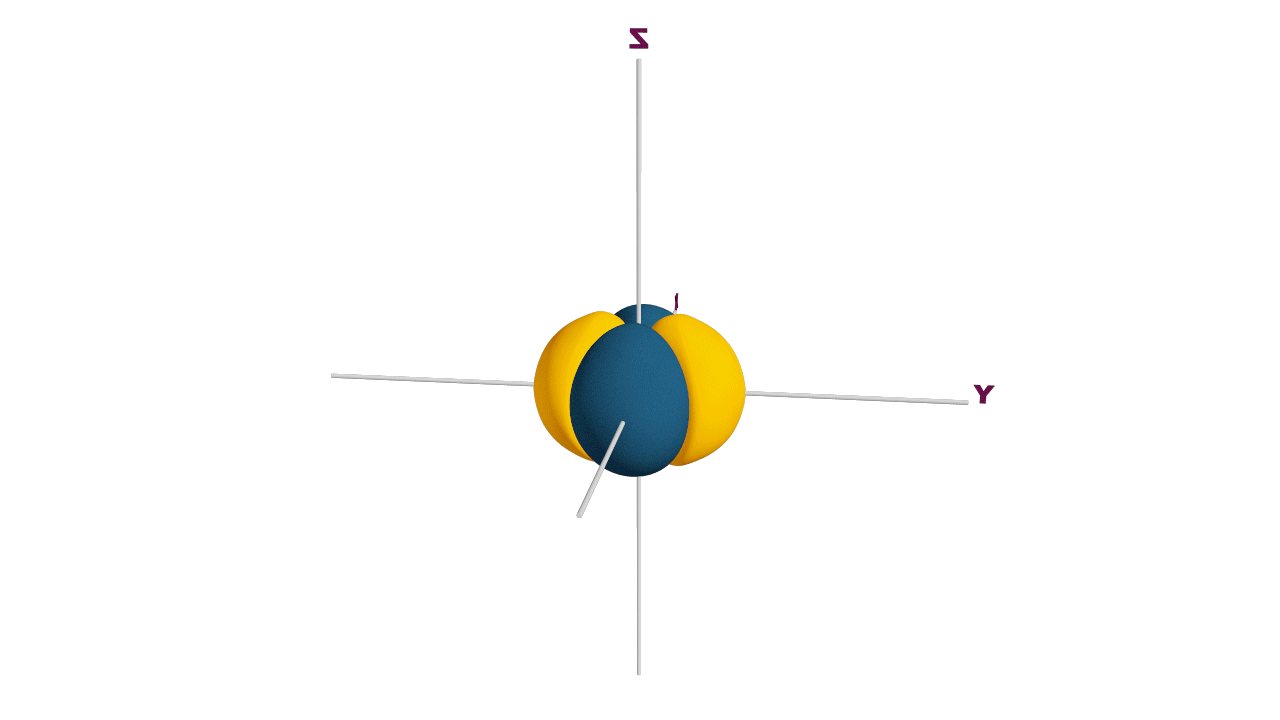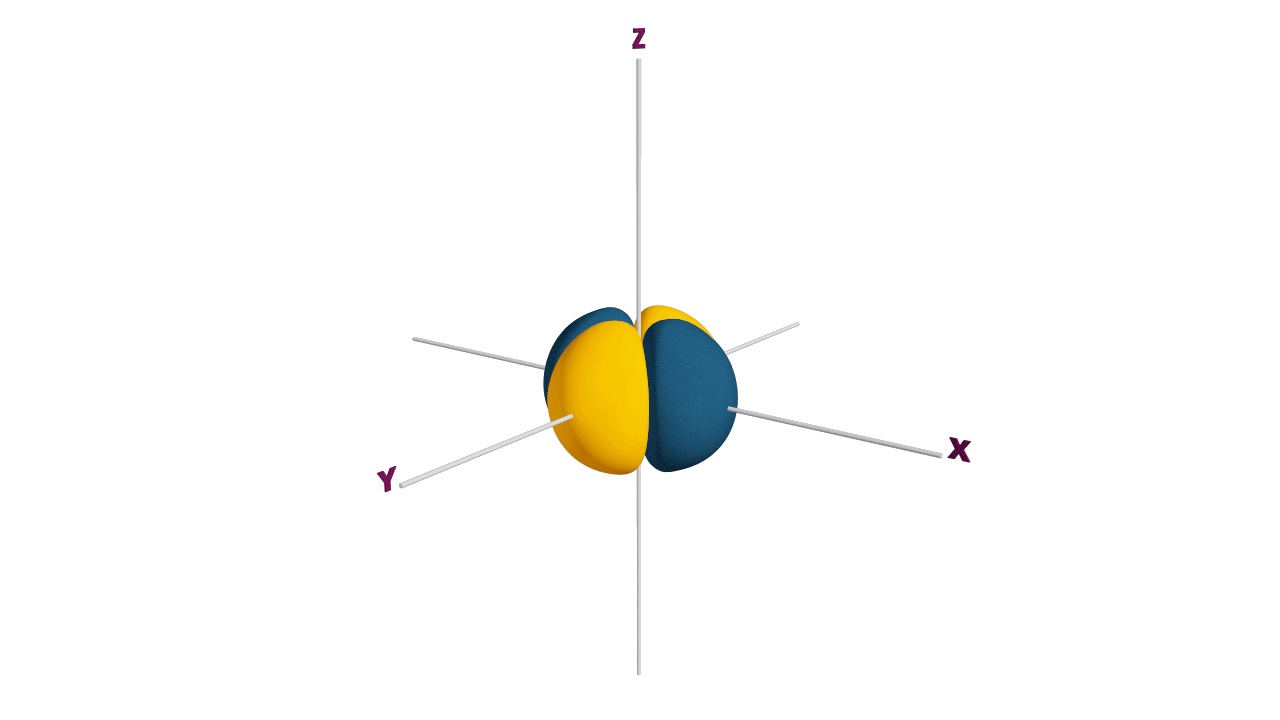
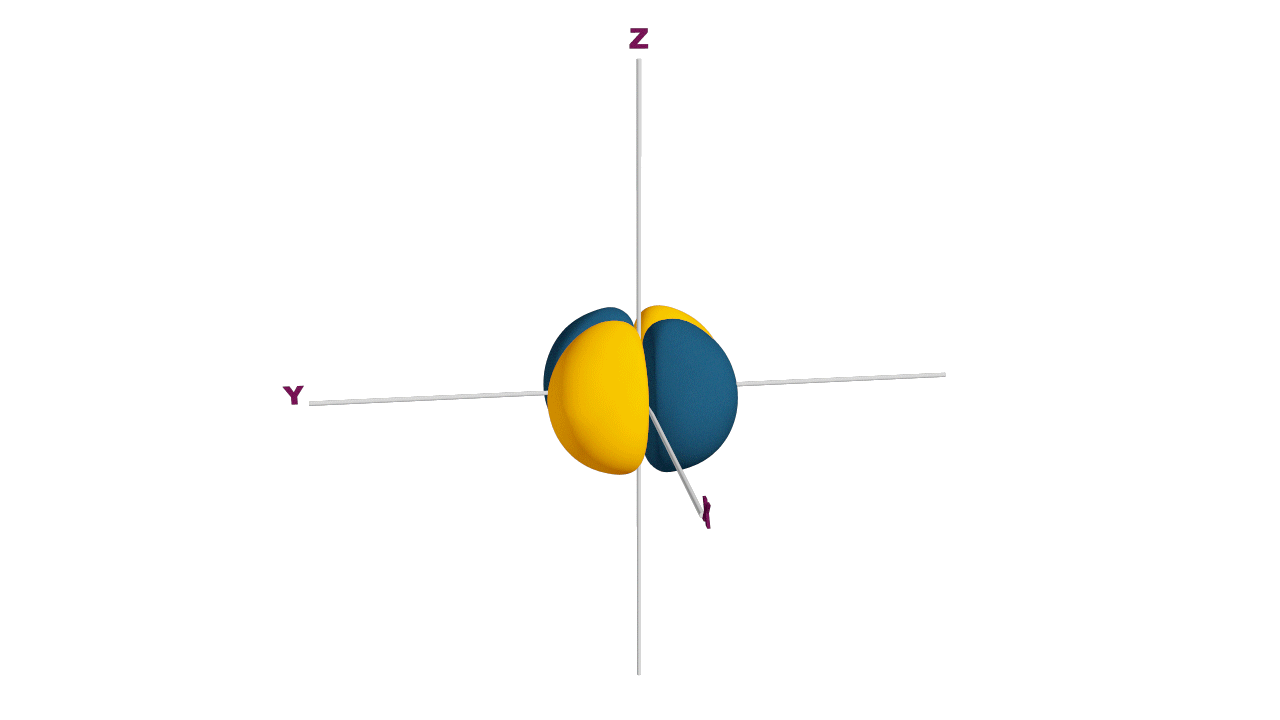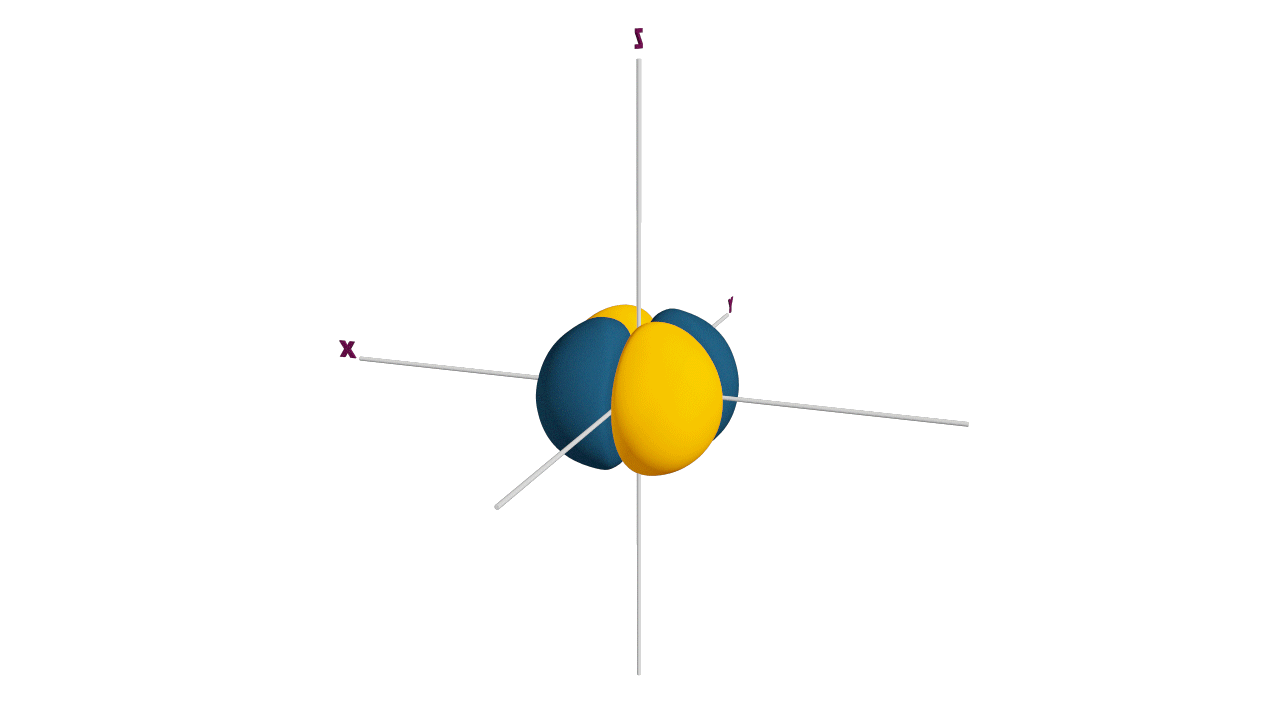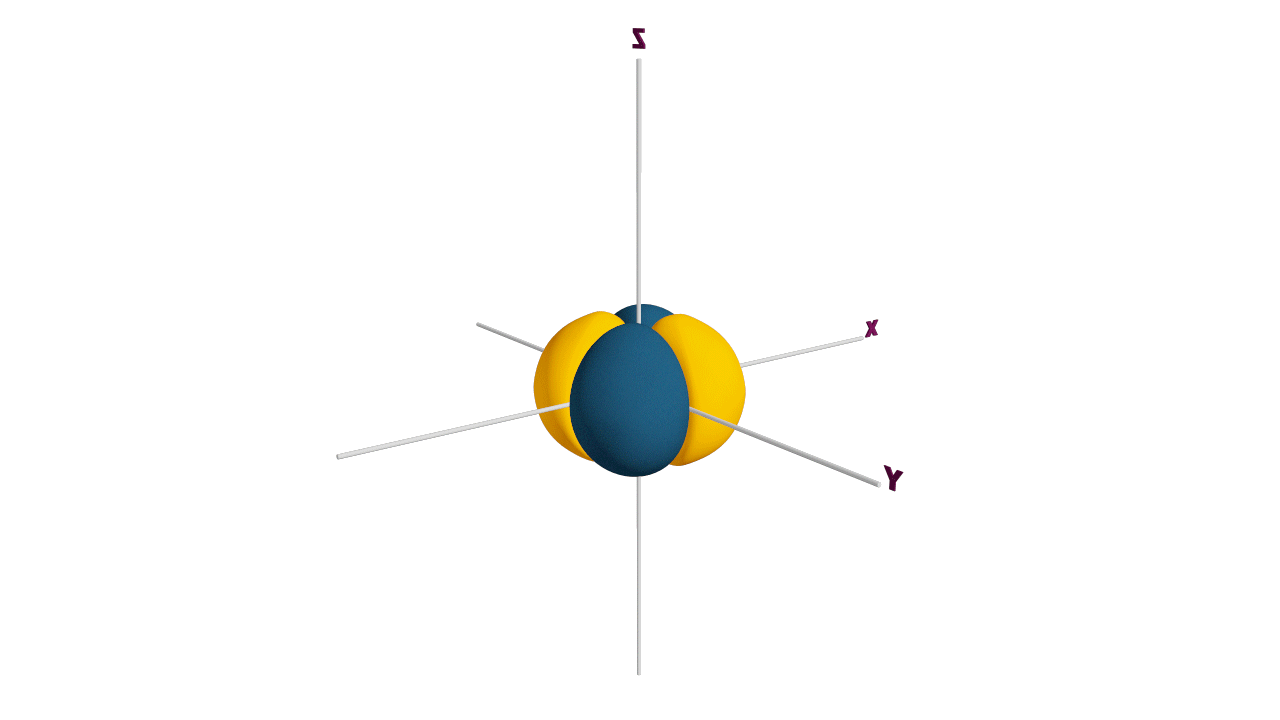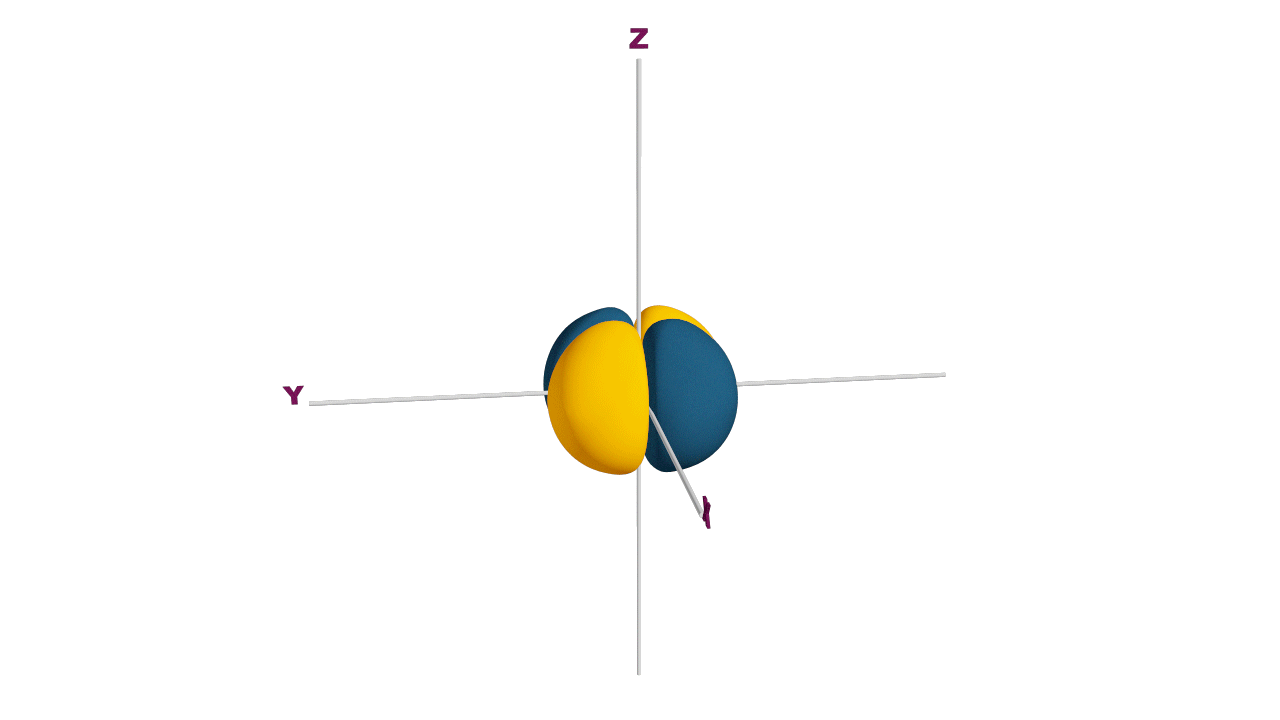
Angular nodes of d-orbitals
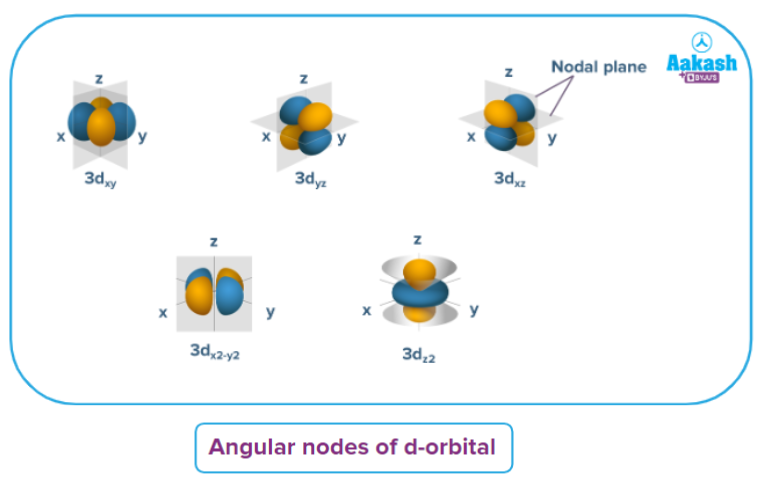
Shapes of orbitals
Shape of the orbitals are plotted with the help of the solutions of the Schrodinger wave equation.
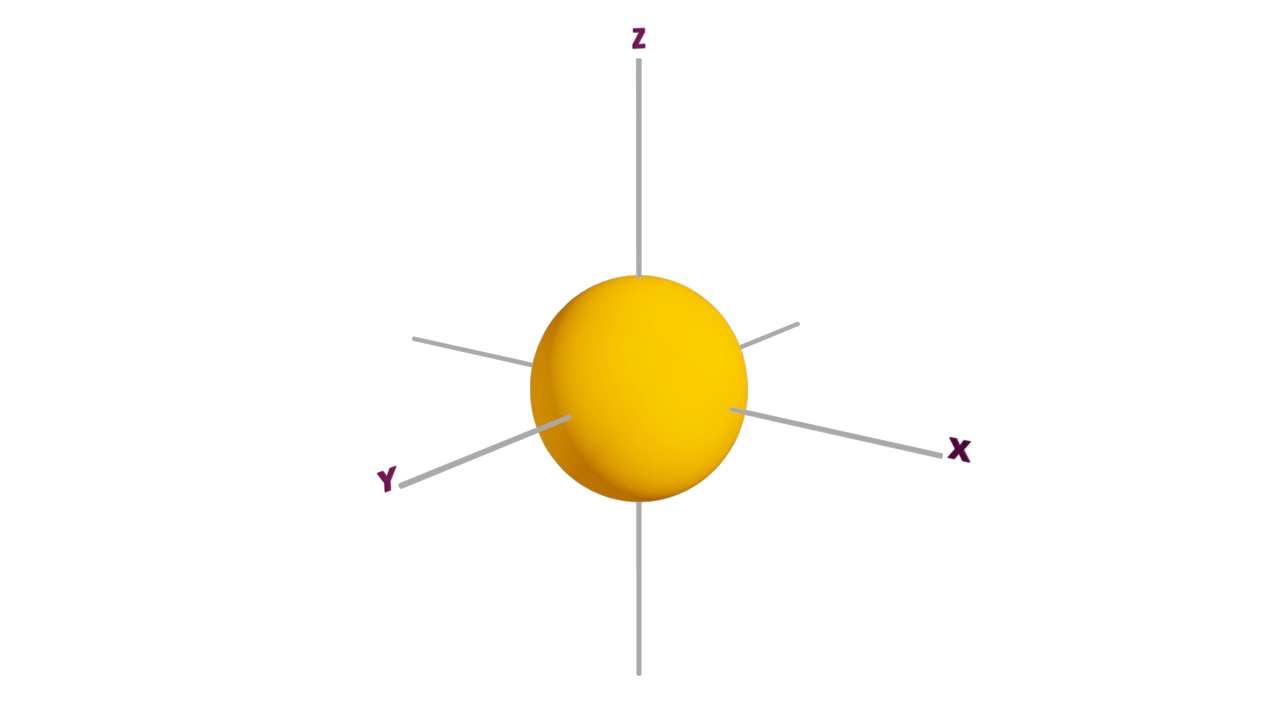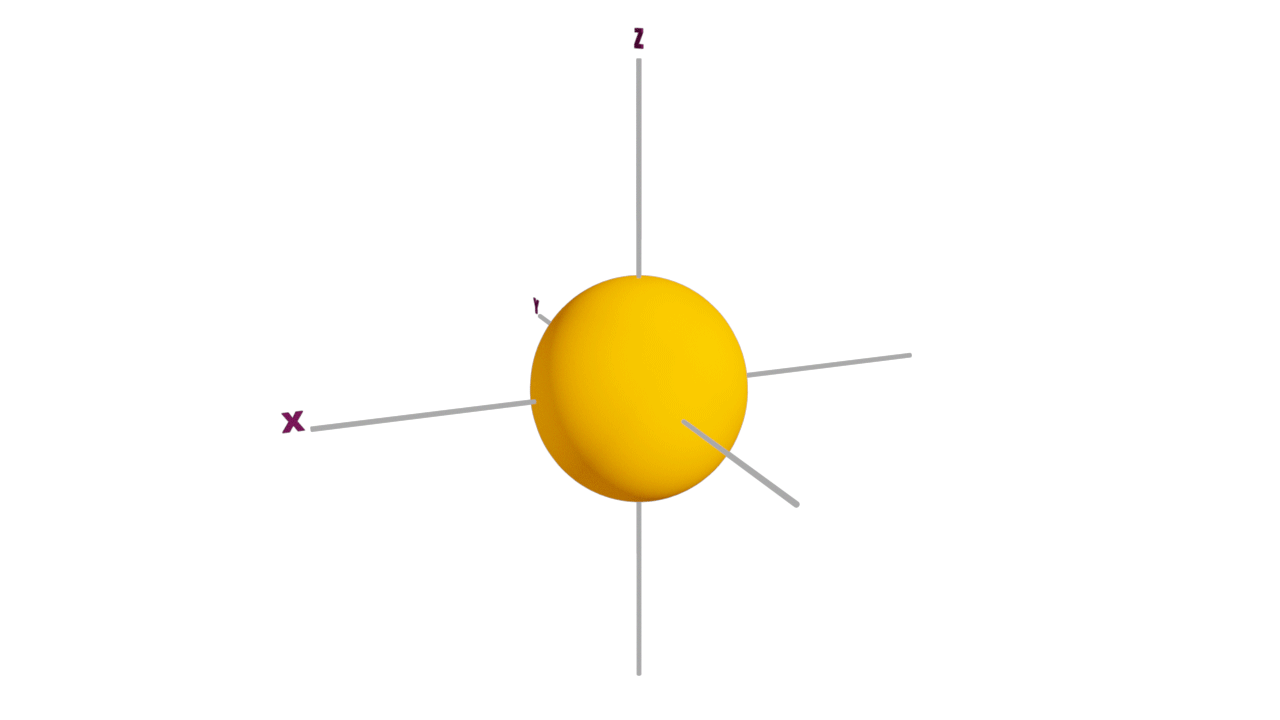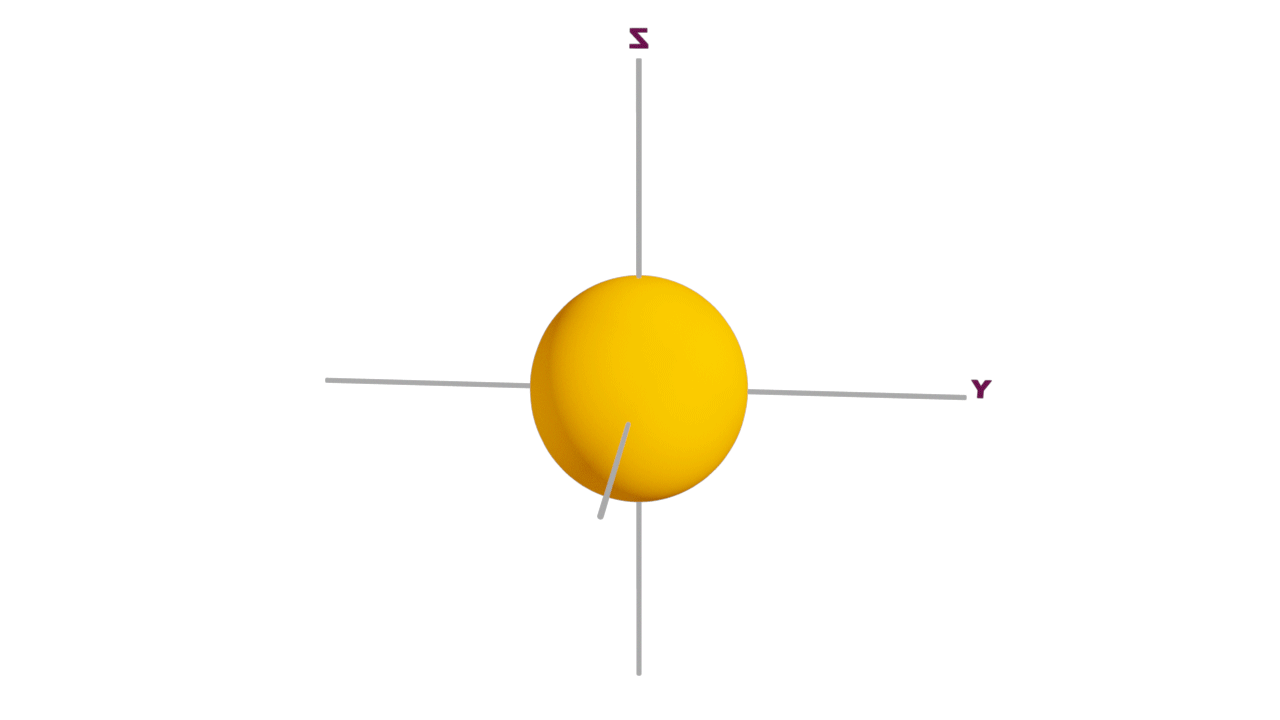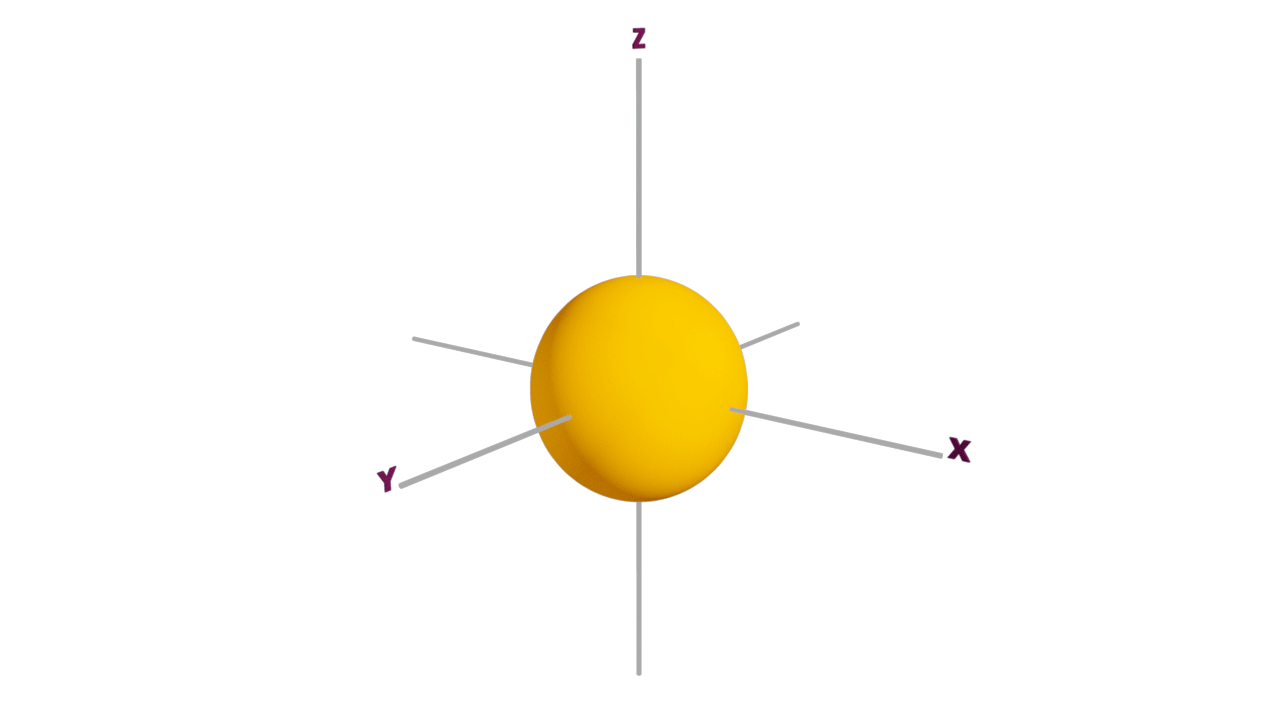
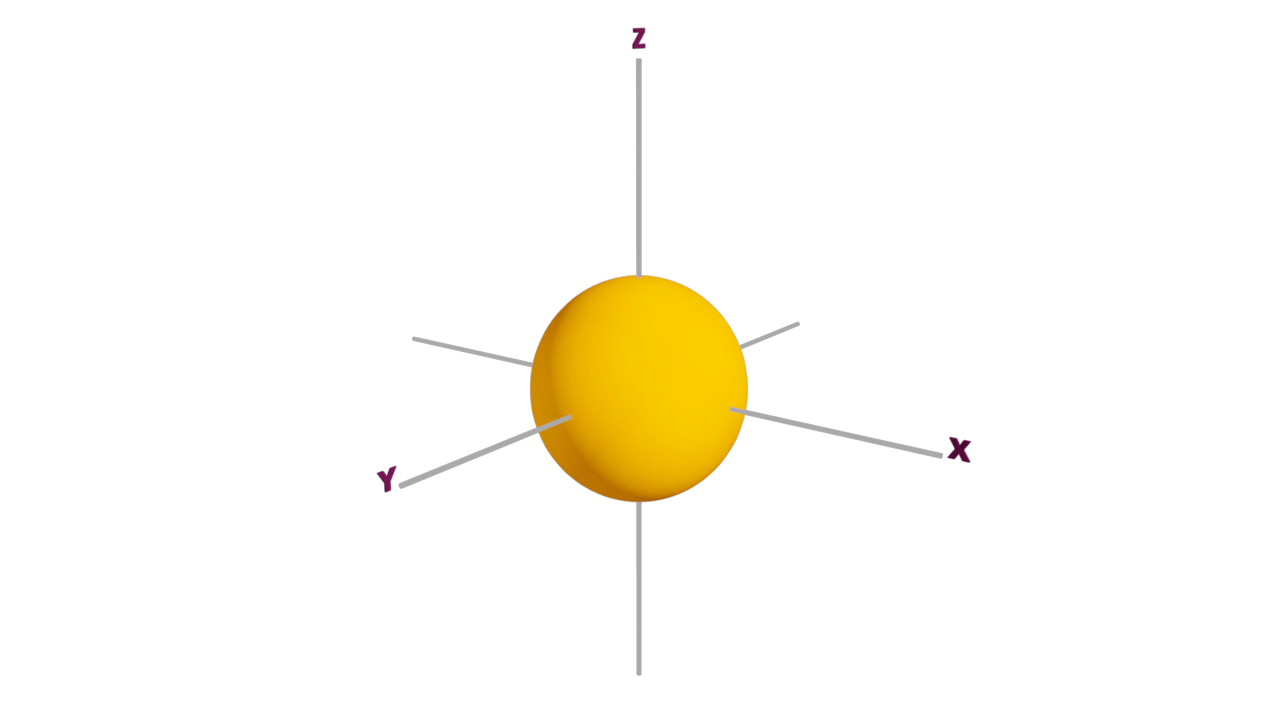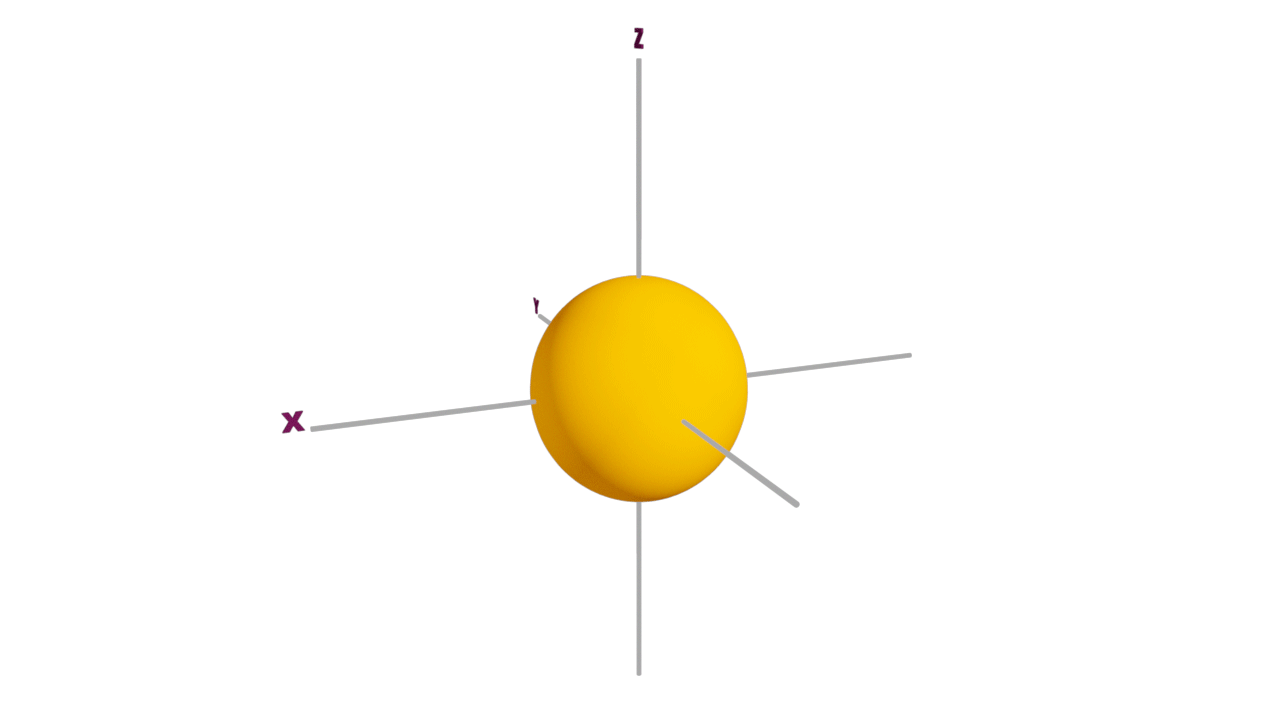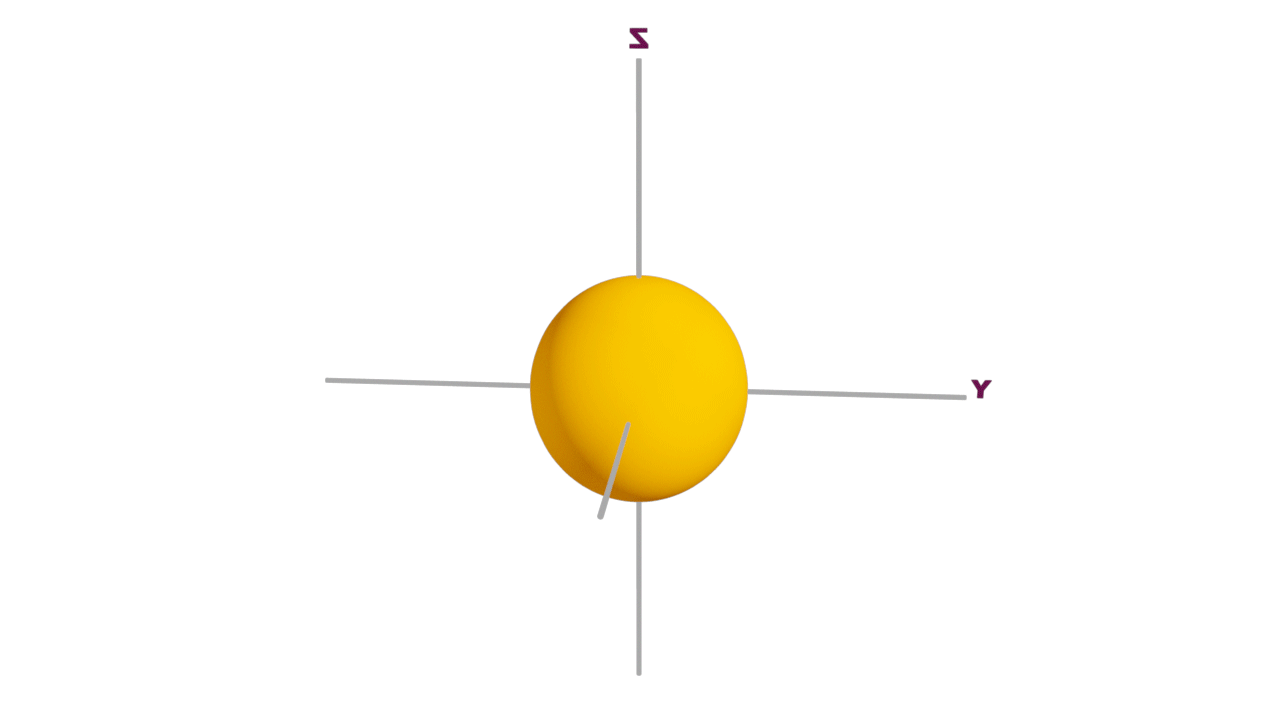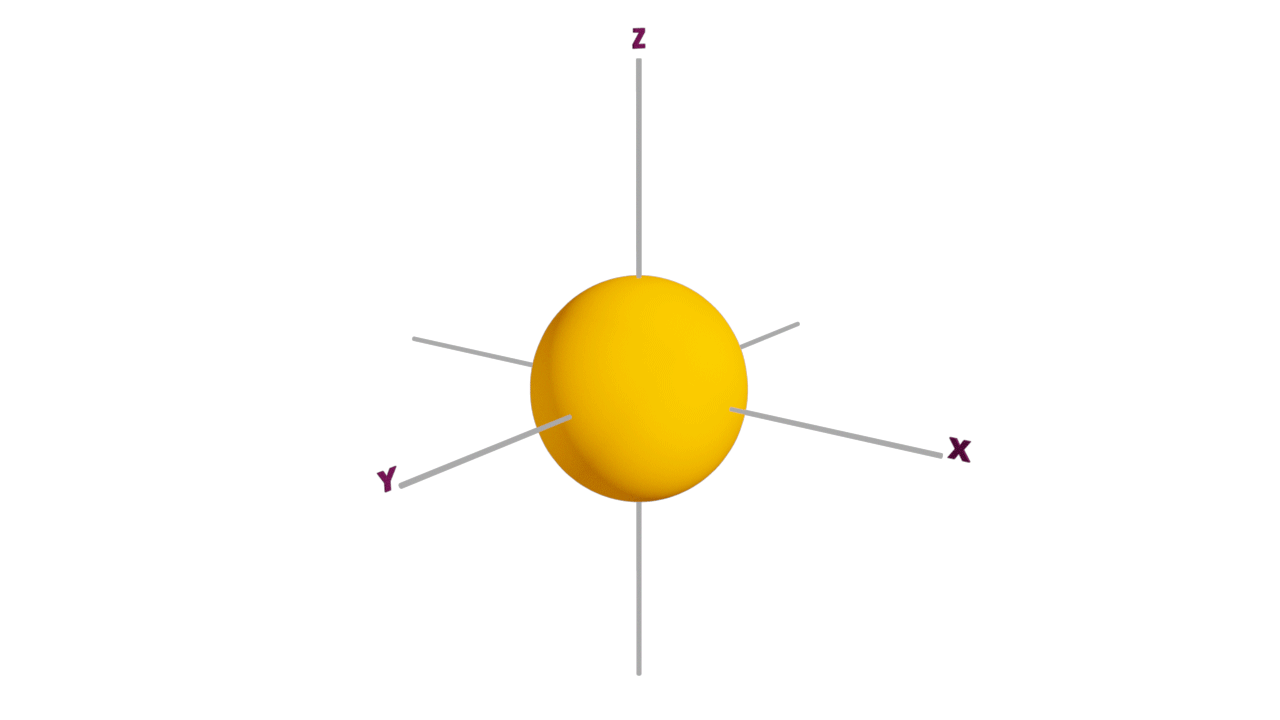
s-orbital:
- The shape of an s-orbital is spherical.
- s orbitals are non-directional and it is closest to the nucleus, having the lowest energy.
- Three things happen to s orbitals as n (principal quantum number) increases:
- They become larger, extending farther from the nucleus.
- They contain more nodes. This is similar to a standing wave that has regions of significant amplitude separated by nodes, and points with zero amplitude.
- For a given atom, the s orbitals also become higher in energy as n increases because of their increased distance from the nucleus.
- It can accommodate a maximum of 2 electrons

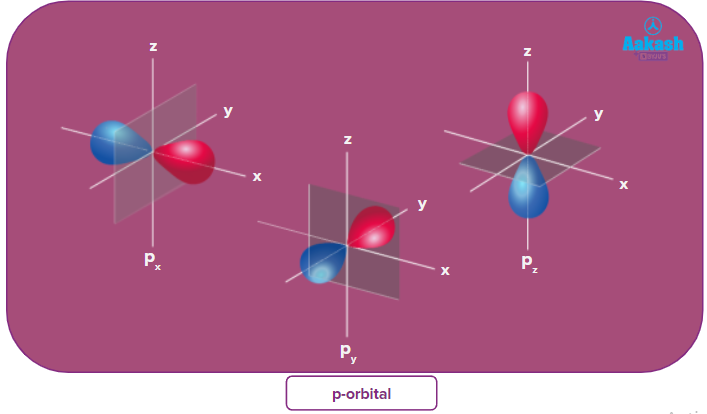
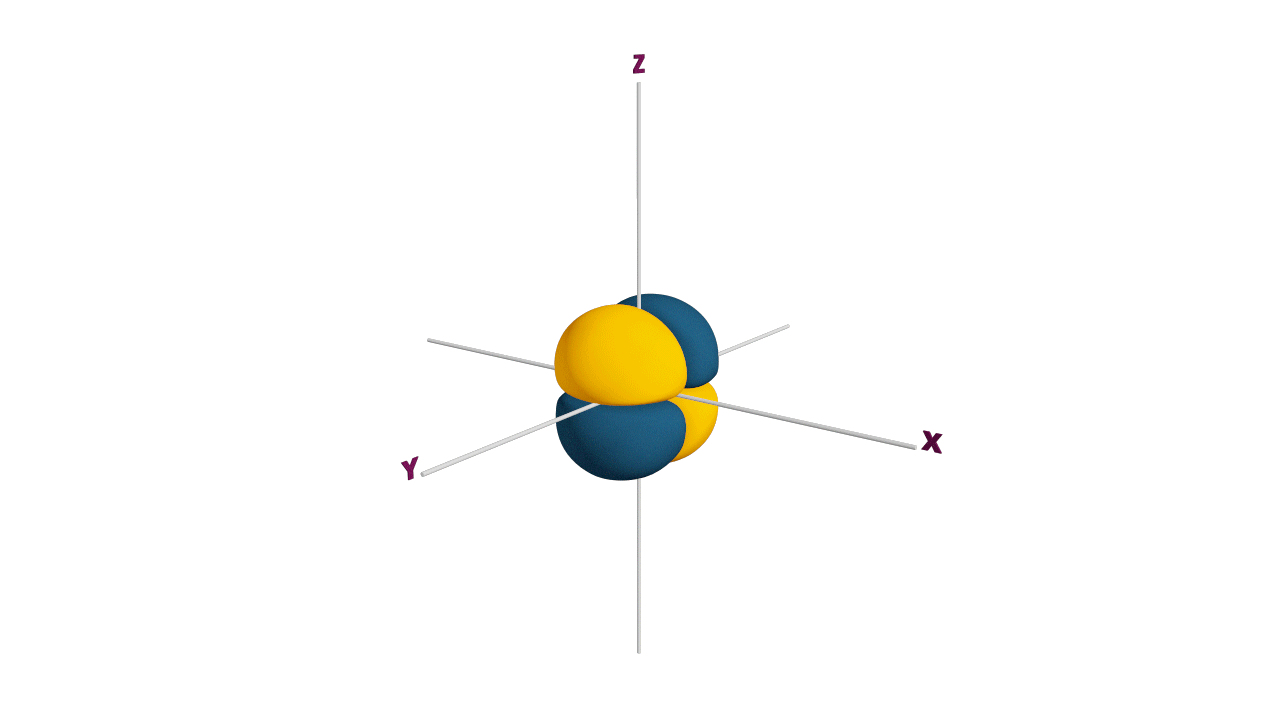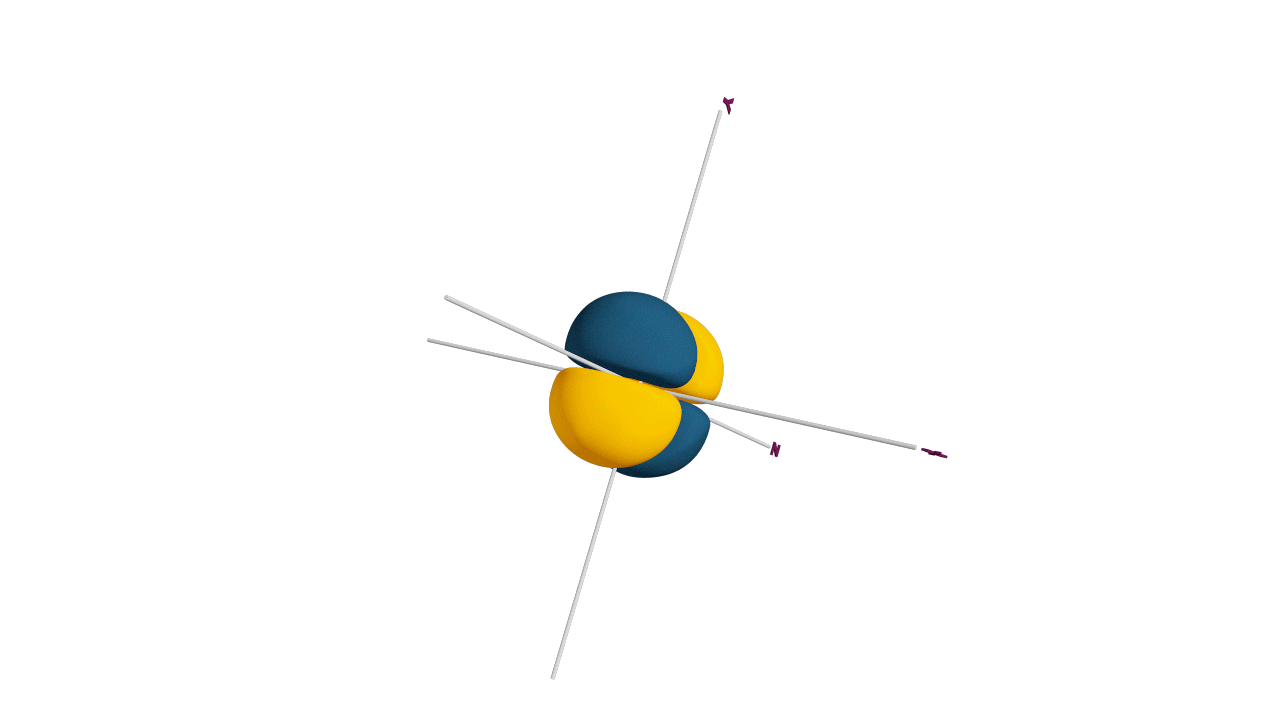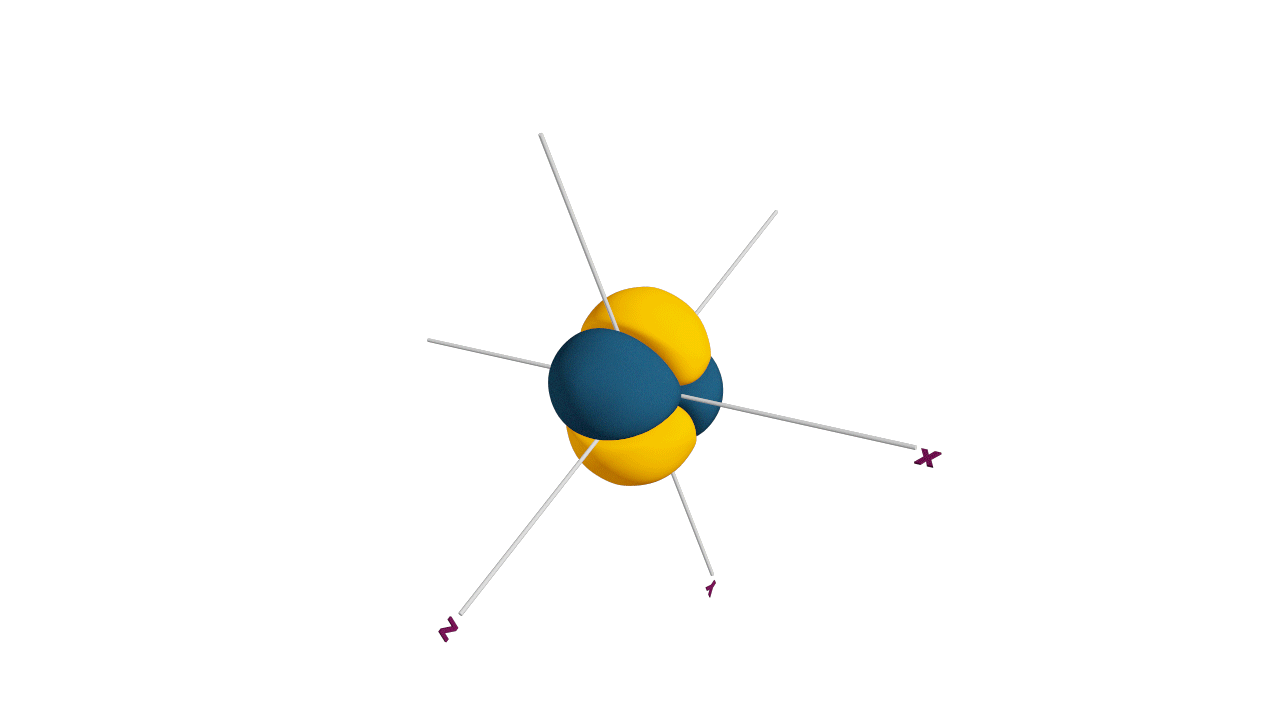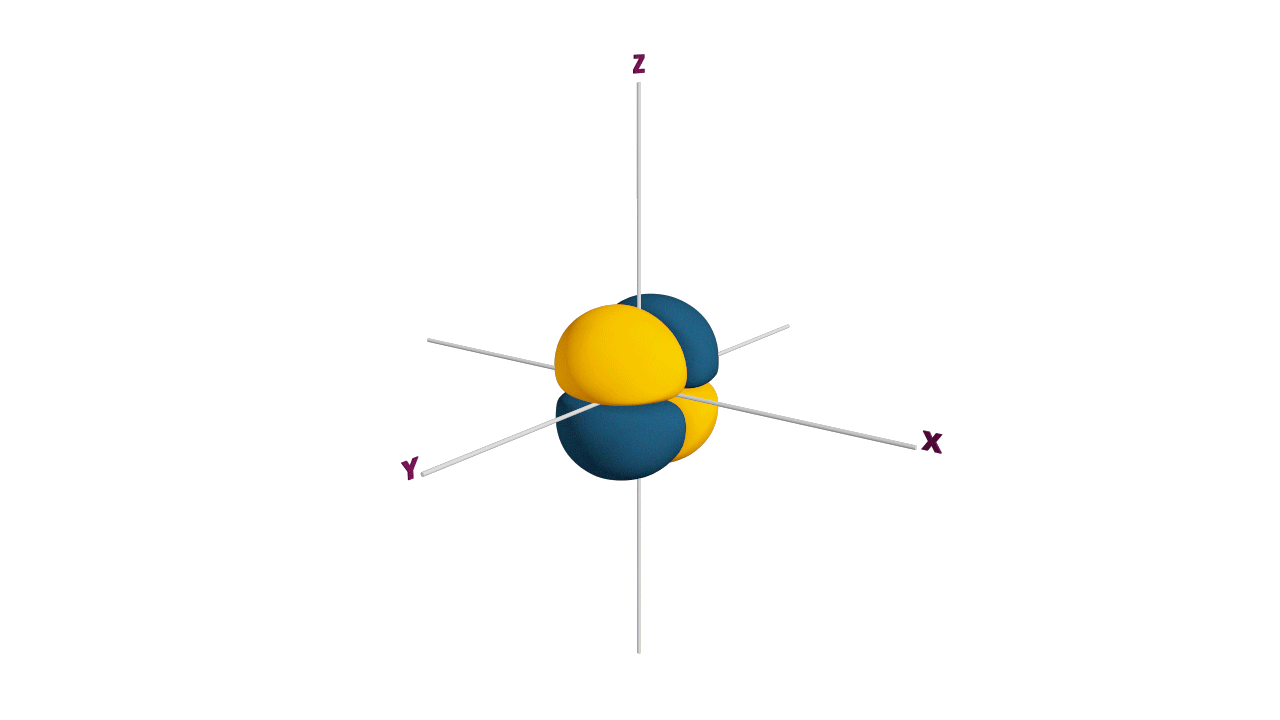
p-orbital:
- The shape of a p-orbital is dum-bell.
- p - orbitals consist of two loops which are separated by a region of zero probability called node
- It can accommodate a maximum of 2 electrons per orbitals and total 6 electrons in one p subshell.
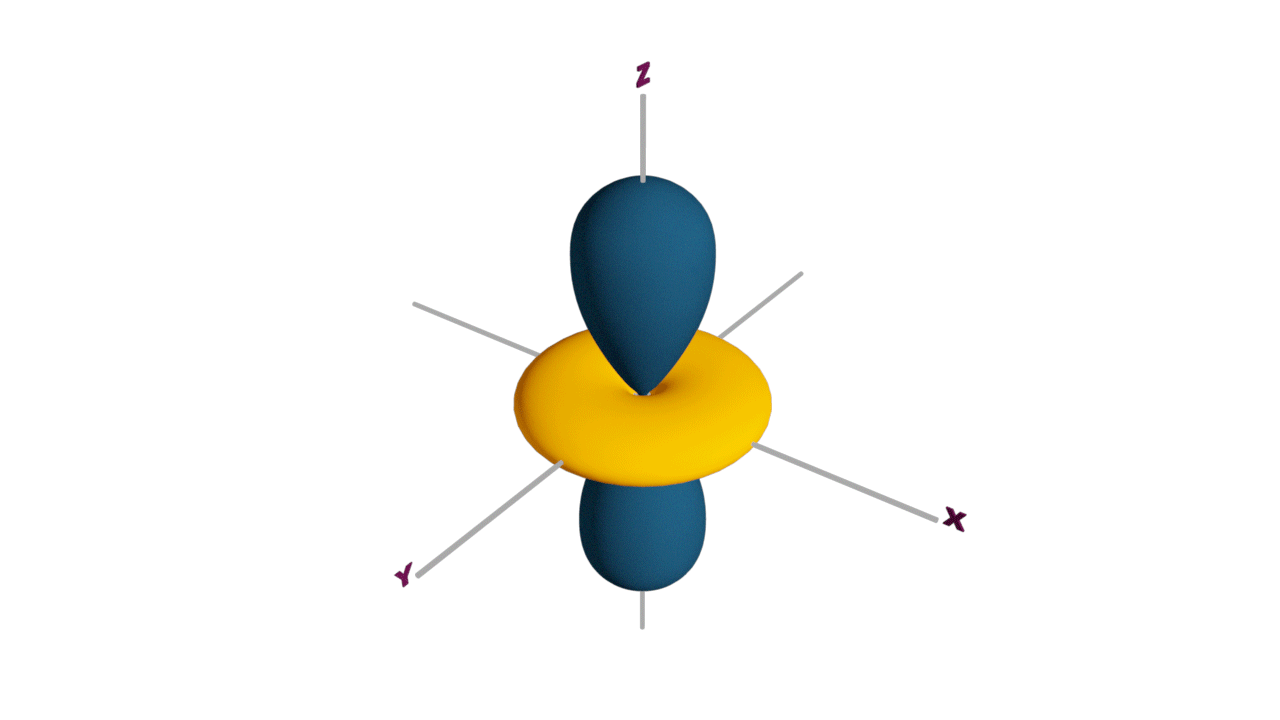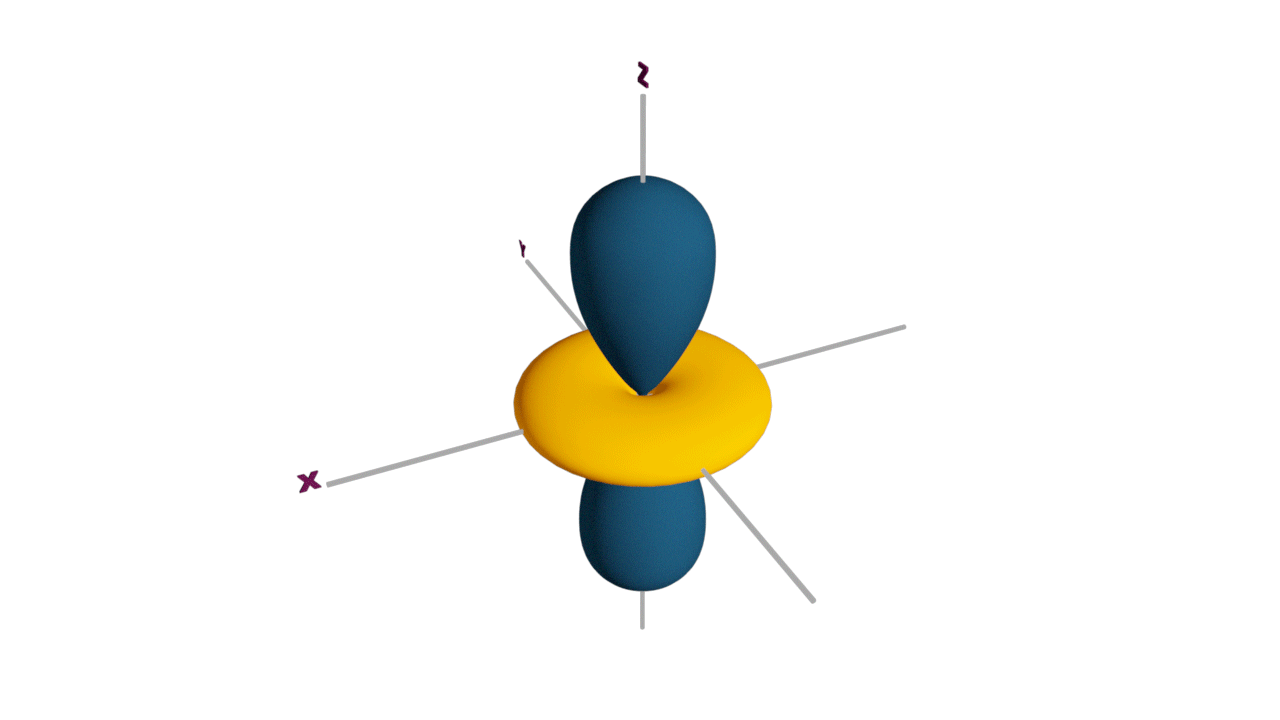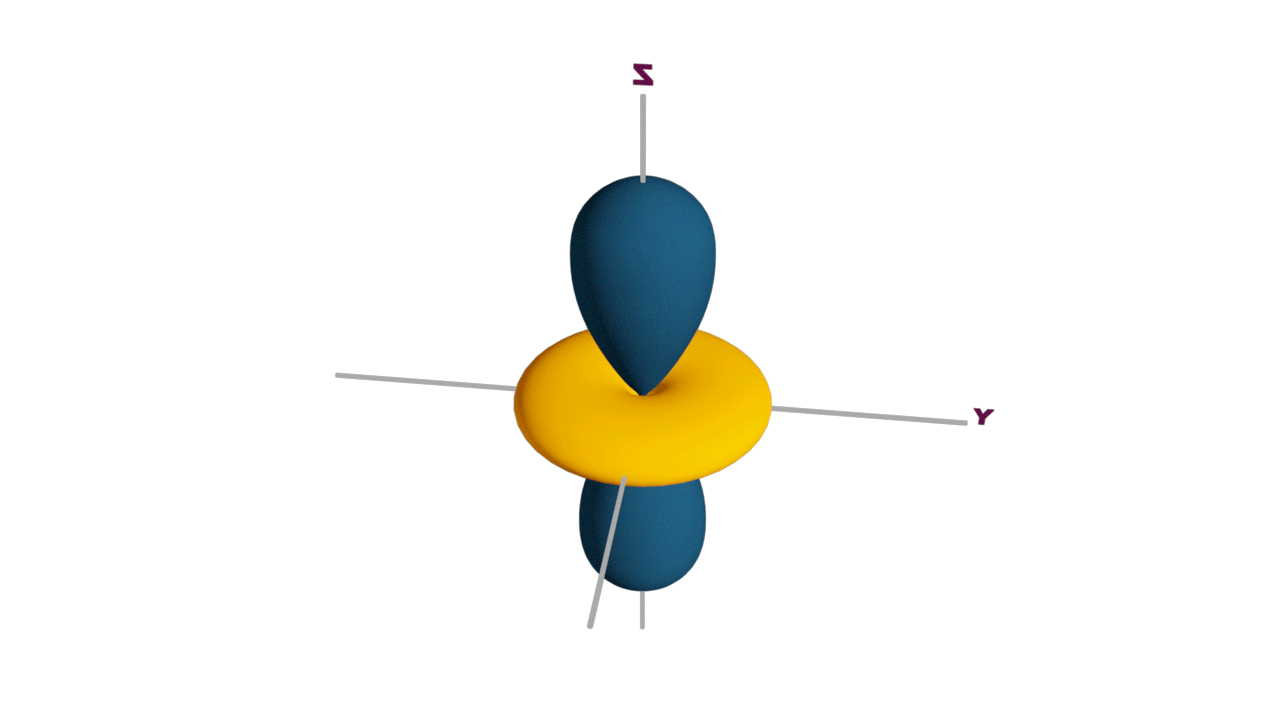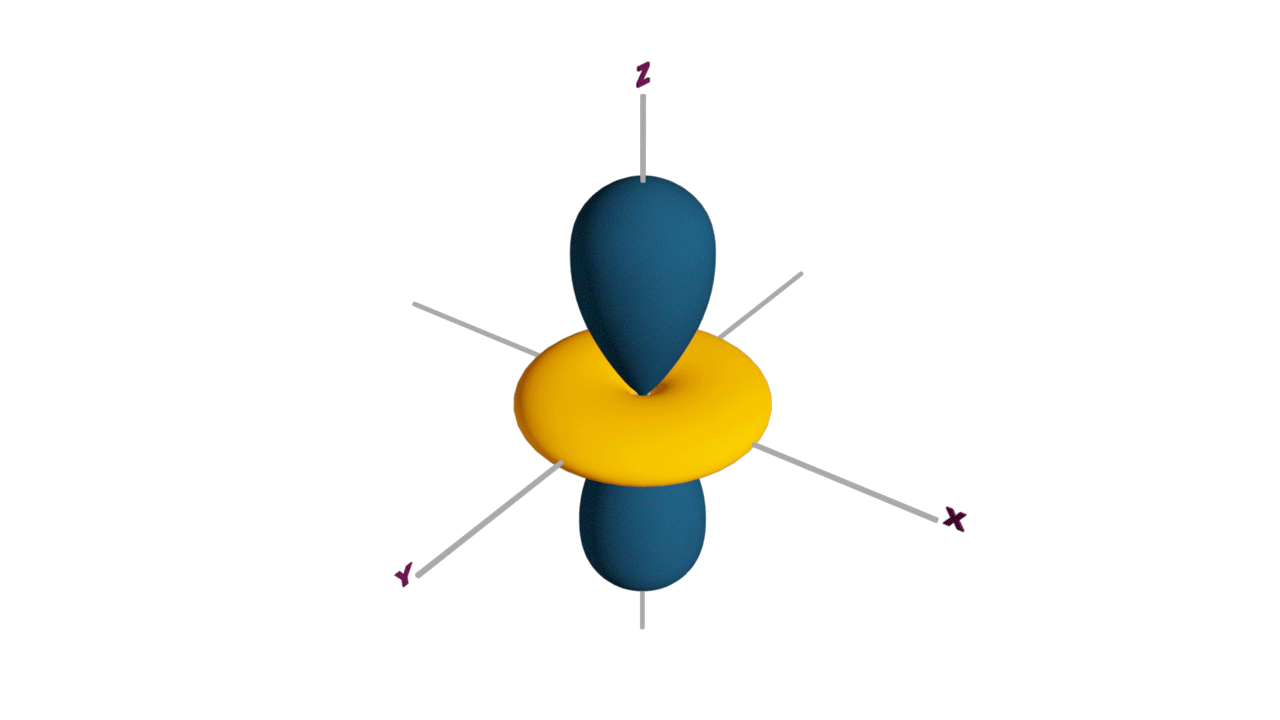
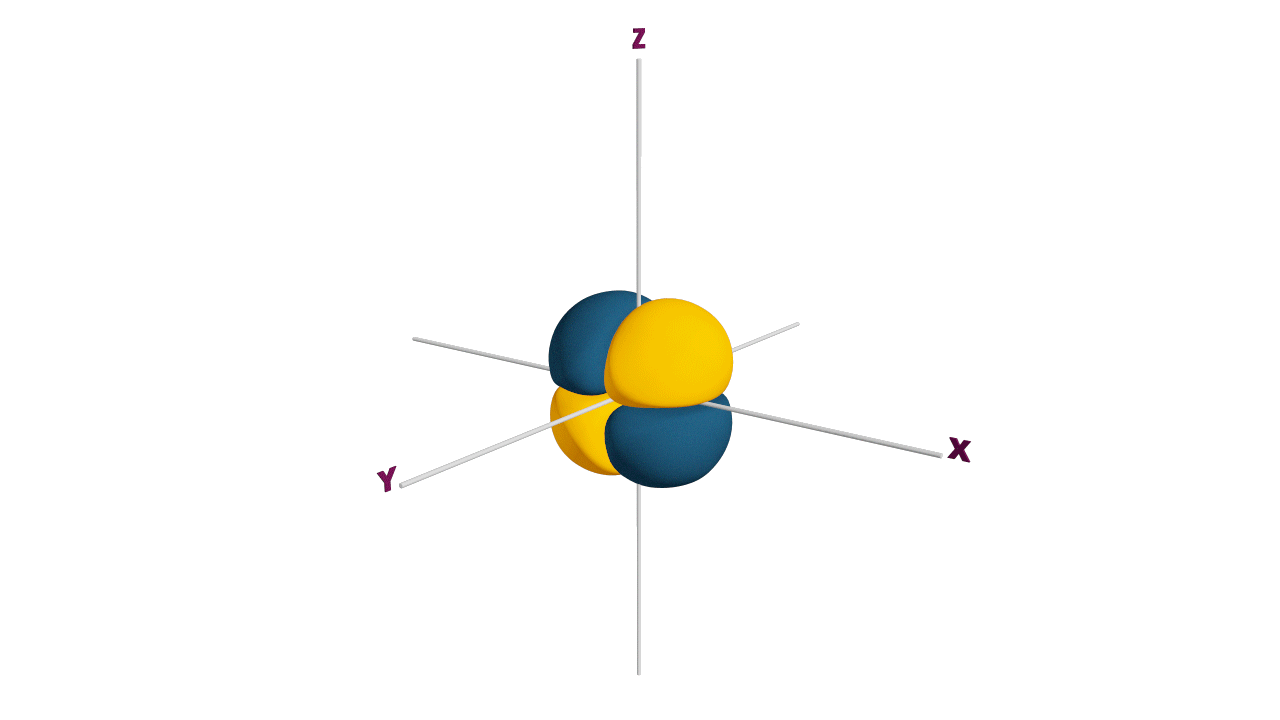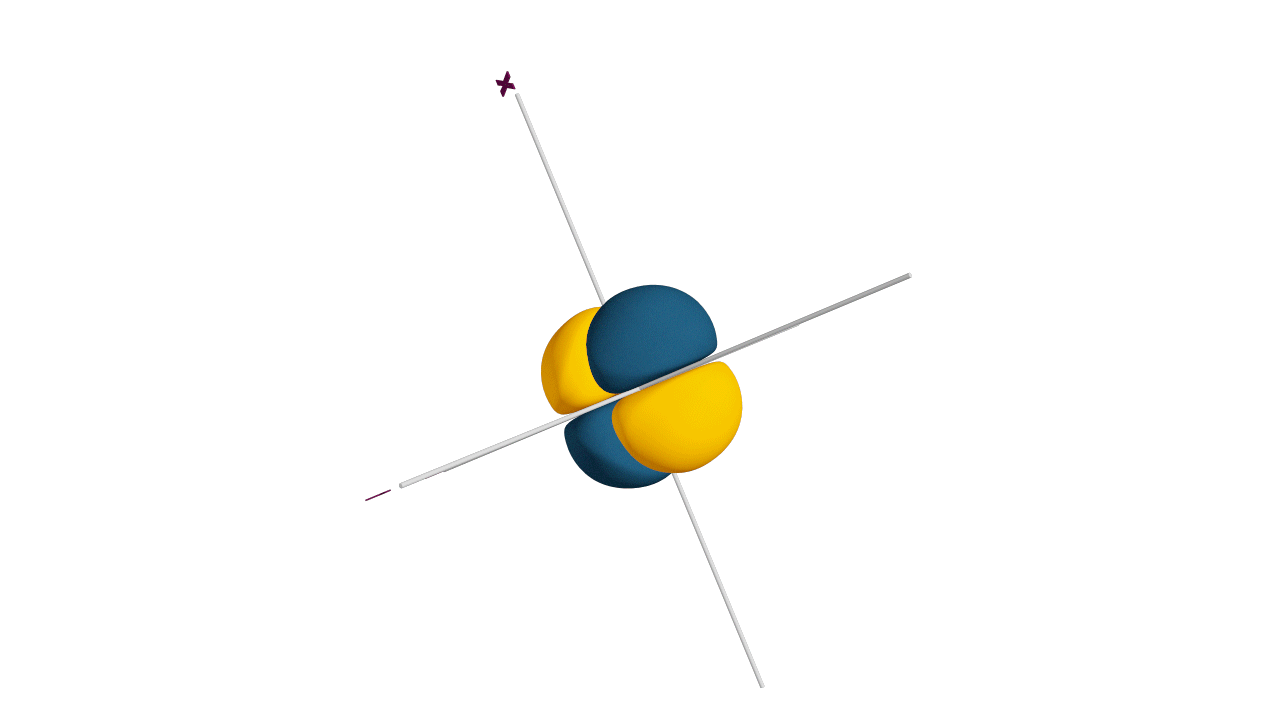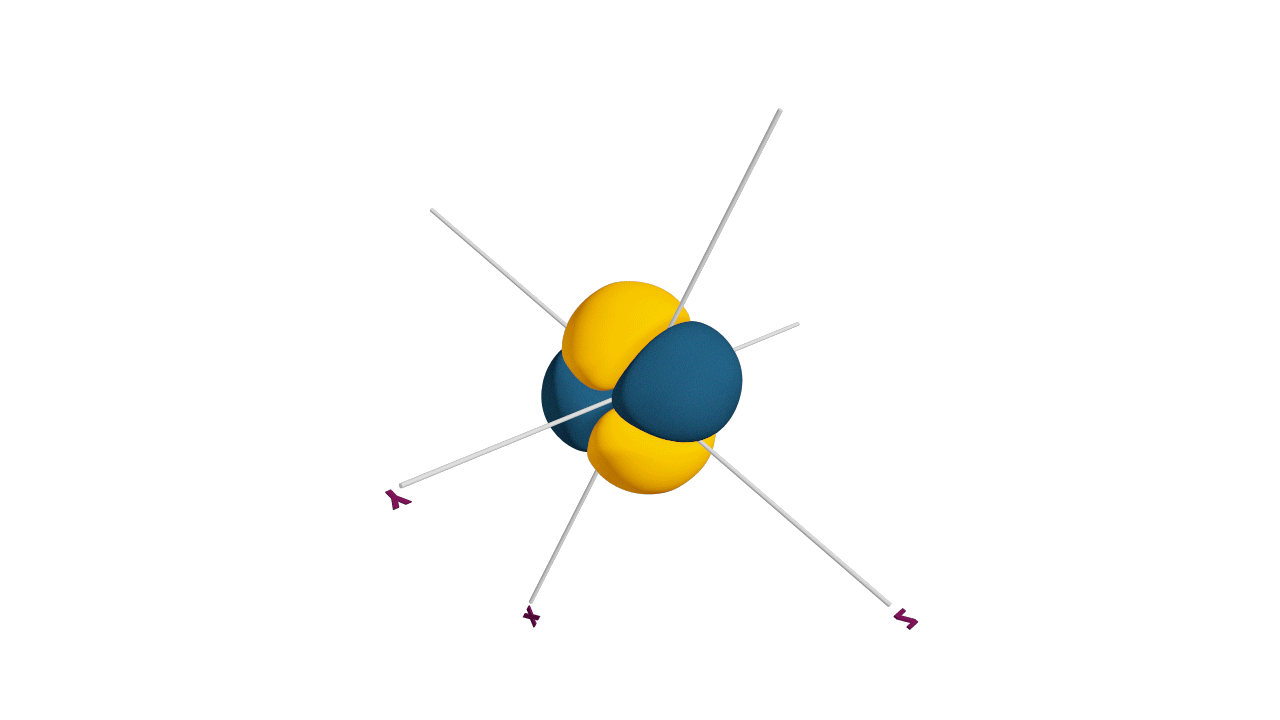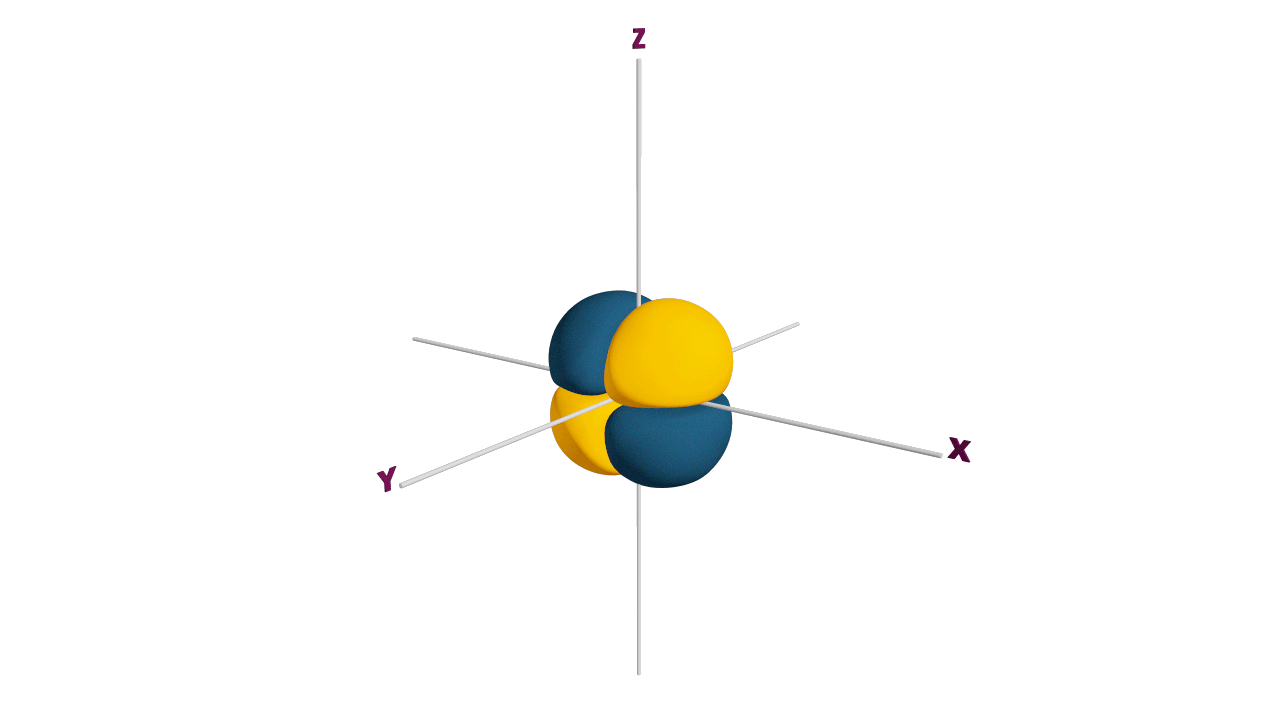
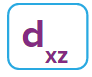
d-orbital:
- The shape of a d-orbital is a double dum-bell.
- There are a set of two different orbital types. One set of orbitals lying on the axis namely (dx2 - y2 and dz2) and the other set lying between axis (dxy, dyz and dzx)
- d - subshell can accommodate the maximum number of 10 electrons.

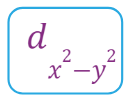


Practice problems
Q1. Number of angular nodes present in 3s orbital
A. 0
B. 1
C. 2
D. 3
Answer: (A)
Solution: angular node = l
For s orbital l=0
Q2. The number of radial nodes of 1s, 2p, and 3d electrons is, respectively,
- 0, 0, 2
- 1, 1, 0
- 2, 2, 2
- 0, 0, 0
Answer: (D)
Solution:
|
orbital |
Principal quantum number (n) |
Azimuthal quantum number (l) |
Radial node (n-l-1) |
|
1s |
1 |
0 |
0 |
|
2p |
2 |
1 |
0 |
|
3d |
3 |
2 |
0 |
Q3. The probability of finding a pz electron is zero in
A. XY-plane
B. YZ-plane
C. XZ-plane
D. Z-axis
Answer: (A)
Solution: electron density present along Z-axis. Along with plane XY zero probability of finding electrons.
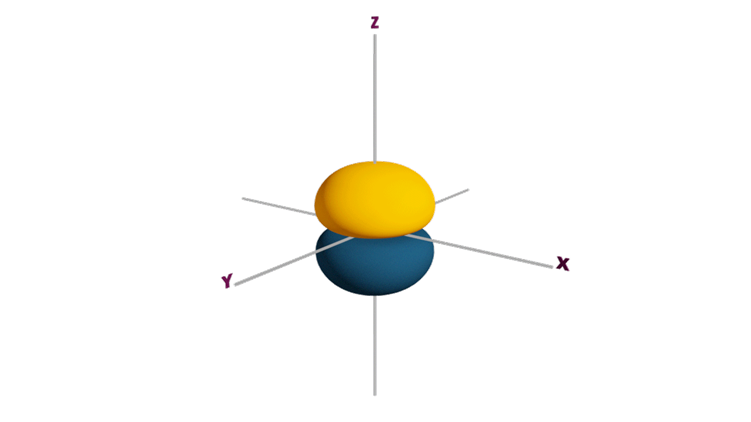

Q4. Which Orbital has maximum symmetry is
A. p - orbital
B. s - orbital
C. dxy -orbital
D. dz2 -orbital
Answer: (B)
Solution: s orbitals are the most symmetrical orbitals because of their spherical shape.

Frequently asked questions - FAQs
Question 1. What do different subshells stand for?
Answer: The subshells which are present in the elements of the periodic table are namely s, p, d, and f, respectively which stand for sharp, primary, diffuse, and fundamental. These notations were given to groups of lines originally noted in the spectra of the alkali metals.
Question 2. What is the difference between a shell/orbit and an orbital?
Answer:
|
Orbital |
Shell/orbit |
|
It represents the 3-dimensional motion of an electron around the nucleus. |
Represents planar motion of electron |
|
Region of space around nucleus where a finding of the electron is a maximum |
Well defined circular path around nucleus |
|
An orbital can hold maximum 2 electrons |
Maximum number of electron held in an orbit will be 2n2 |
|
Non-directional in nature |
Directional in nature |
Question 3. How many subshells are present in the elements of the periodic table?
Answer: Our periodic table consists of 118 elements and to define their electronic configuration we only use s, p, d, and f subshells generally. For hypothetical elements, we can use g (9 orbitals), h (11 orbitals), etc subshells.
Question 4. Which d orbital does not contain any nodal plane?
Answer: dz2,
Two lobes oriented in the z-axis with the ring of high electron density concentrated in the xy plane. It contains no nodal plane but rather has 2 nodal cones.
Question 5. What are subshells?
Answer: A group of orbitals building up the orbit/shell of an atom are called subshells. E.g- s subshell, p subshell, d subshell, f subshell
Question 6. What is the Schrodinger wave equation?
Answer: Schrodinger wave equation is a mathematical expression describing the energy and position of the electron in space and time, taking into account the matter wave nature of the electron inside an atom.
It is based on three considerations. They are;
- Classical plane wave equation,
- Broglie’s Hypothesis of matter-wave, and
- Conservation of Energy
Schrodinger equation gives us a detailed account of the form of the wave functions or probability waves that control the motion of some smaller particles. The equation also describes how these waves are influenced by external factors. Moreover, the equation makes use of the energy conservation concept that offers details about the behavior of an electron that is attached to the nucleus.


