-
Call Now
1800-102-2727
Nucleophilic Substitution Reaction- SN2: What is SN2 Reaction ? Mechanism, Energy Profile Diagram, Rate of the SN2, Reaction, Stereochemistry, Effect of Solvent
Let us consider a small story of the cats and the comfortable chair to understand this topic better. Here, the brown cat (nucleophile) wants to sit on the chair where the grey cat (leaving group) is already sitting.
When the grey cat (leaving group) is about to leave, the brown cat (nucleophile) occupies the comfortable chair, hence giving us a transition phase of one cat coming in and the other going out at the same time. It shows the formation of the transition state.
It shows the final stage, where the grey cat (leaving group) left and the brown cat (nucleophile) occupied the comfortable chair.

Considering this as an analogy for mechanism, we get a hint of what happens in the reaction. What we understood is that in reaction for substitution reaction to occur there must be some leaving group, and nucleophile will attack only after the leaving group has left. Now, let's get into the details of reaction.
TABLE OF CONTENTS
- What is an SN2 Reaction?
- Mechanism of SN2 Reaction
- Energy Profile Diagram of SN2 Reaction
- Rate of the SN2 Reaction
- Stereochemistry of SN2 Reaction
- Effect of Solvent
- Practice Problems
- Frequently Asked Questions - FAQs
What is an Reaction?
The SN2 reaction is a bimolecular nucleophilic substitution reaction because the rate-determining step involves two reacting species, i.e. the alkyl halide and the nucleophile. It is a single-step reaction in which a bond is broken and a new one is created simultaneously.

Mechanism of SN2 Reaction
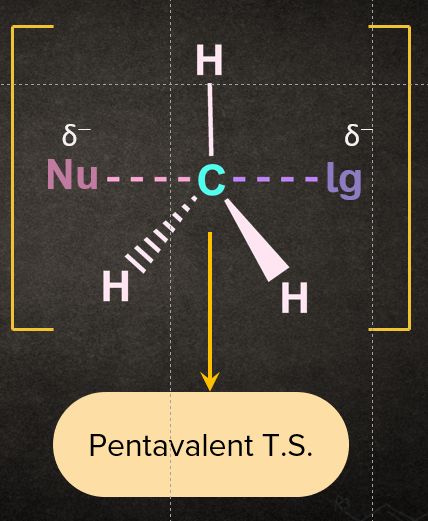
The SN2 reaction mechanism follows a single step in which a nucleophile attacks the substrate from the backside, which initiates the reaction. At an angle of 1800to the carbon-leaving group bond, the nucleophile approaches the supplied substrate through a transition state, the carbon-nucleophile bond is formed and the carbon-leaving group bond breaks at the same time. Now, on the other side of the carbon-nucleophile bond, the leaving group is forced out of the transition state, generating the corresponding product.. It's worth noting that the final product is created by inverting the tetrahedral geometry at the central atom. Below is a diagram of the reaction mechanism for the nucleophilic substitution of chloromethane with hydroxide ion as the nucleophile.
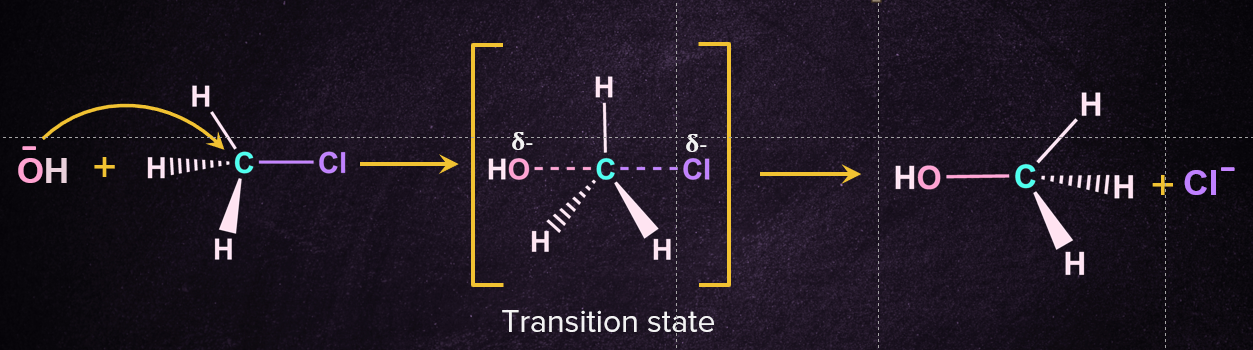
Energy Profile Diagram of Reaction
In the transition state, a bond is partially formed between the nucleophile and the carbon, and the bond between the carbon and the leaving group is partially broken as it involves simultaneous breaking and formation of bond.

Rate of the SN2 Reaction
The rate of reaction depends on the concentration of both alkyl halide and the nucleophile.
|
Rate ∝ [Alkyl halide][Nucleophile] |
It is a bimolecular, one-step process.
The nature of alkyl halide decides the reactivity of the reaction. Very large and bulky groups can often hinder the formation of the required transition state. The crowding raises the energy of the transition state and slows down the rate of reaction.
Decreasing order of reactivity of alkyl halides is:
|
Substituent |
Compound |
Relative rate |
|
Methyl |
30 |
|
|
10 |
1 |
|
|
20 |
0.02 |
|
|
30 |
∽0 |
Also, rate of reaction also depends on R-X bond, for a given alkyl group. Weaker the R-X bond, more easily it can be cleaved for further nucleophiles to attack.
|
Bond |
C-X Bond enthalpy (kJ mol-1) |
Bond length(pm) |
|
CH3-I |
234 |
214 |
|
CH3-Br |
293 |
193 |
|
CH3-Cl |
351 |
178 |
|
CH3-F |
452 |
139 |
Order of reactivity of R-X bond towards SN2 nucleophilic substitution is: R-I > R-Br > R-Cl >R-F
Strong anionic nucleophiles speed up the rate of the SN2 reaction, And mostly anionic nucleophiles give SN2 reaction. Example-
Stereochemistry of SN2 Reaction
In an SN2 mechanism, the nucleophile attacks from the back side, that is from the side directly opposite to the leaving group. This causes an inversion of configuration at the chiral carbon atom. Also known as Walden inversion.

SN2 is a stereospecific reaction. Stereospecific reaction is a reaction in which a particular stereoisomeric form of the reactant reacts in such a way that it leads to a specific stereoisomeric form of the product
Effect of Solvent
Polar aprotic solvents have a crowded positive center, so they do not solvate the anion appreciably. So they don’t help in the breaking of R-X bond or in the removal of leaving group. This rules out the possibility of SN1 reaction in this case. Hence, the rate of SN2 reactions increases when they are carried out in a polar aprotic solvent.

As we can see here, there is a repulsion between the methyls of DMSO and the nucleophile. As a result, the nucleophile remains unsolvated.
DMSO and acetone are commonly used solvents in SN2 reactions.
Practice problems
Q1. Write the product of the following reaction:
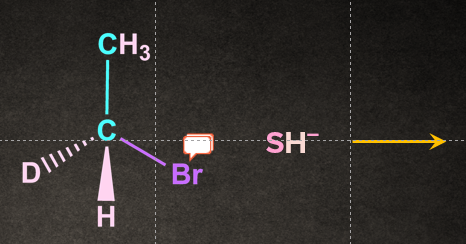
A). 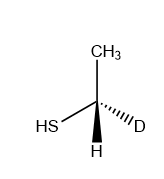
B). 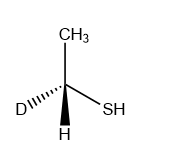
C). Both A and B
D). None of the above
Answer: A
Solution: In an SN2 reaction nucleophile attacks from the back side, that is from the side directly opposite to the leaving group. Such that the product formed is always inversion product.
Q2. In an SN2 reaction of alkyl halide on chiral centres there is:
A). 100 % racemization
B). inversion more than retention leading to partial racemization
C). 100 % retention
D). 100 % inversion
Answer: D
Solution: In an SN2 reaction nucleophile attacks from the back side, that is from the side directly opposite to the leaving group. Such that the product formed is always 100 % inversion.
Q3. Arrange the following alkyl chlorides in the order of decreasing reactivity in an SN2 reaction?

A). P > Q > R > S
B). S > R > Q > P
C). S > Q > R > P
D). R > Q > S > P
Answer: B
Solution: Nature of alkyl halide decides the reactivity of SN2 reaction. Bulky groups often hinder the formation of the required transition state. The crowding raises the energy of the transition state and slows down the rate of reaction.
The decreasing order of reactivity of alkyl halides is:
Hence, the decreasing order of alkyl chloride is: S > R > Q > P
Q4. Which of the following statements are correct about SN2 reaction?
A). It is a concerted reaction.
B). No intermediate is formed during the course of reaction.
C). It is a one step reaction.
D). All of the above
Answer: D
Solution: SN2 reaction is a bimolecular concerted reaction, such that breaking and formation of bonds occur simultaneously. Since it is a one step reaction, no intermediate is formed instead reaction occurs via formation of a single transition state.
Frequently Asked Questions-FAQs
Q1. What is a nucleophilic substitution reaction?
Answer: The replacement of an atom or group by any other atom or group in a molecule is known as substitution reaction. If a substitution reaction is brought about by a nucleophile, then it is known as a nucleophilic substitution reaction.
Q2. What is a leaving group?
Answer: The part of the reactant molecule which gets cleaved is called a leaving group. Generally poor bases are a good leaving group.
1. Nucleofuge: Leaving group that carries away an electron pair. For nucleofuge, weaker bases are good leaving groups.
2. Electrofuge: Leaving group that comes away without an electron pair.
Generally, leaving group carries away an electron pair.
Example: 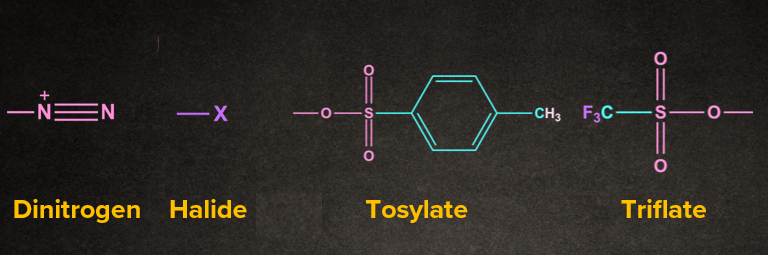
Q3. What is a rate determining step(RDS)?
Answer: If one step in a multistep reaction is intrinsically slower than all the others, then the rate of the overall reaction will be essentially the same as the rate of this slowest step. This slowest step is known as the rate-limiting step or the rate-determining step(RDS).
Q4. Why will the energy of the transition state be maximum?
Answer: In the transition state, carbon is simultaneously bonded to the incoming nucleophile and the outgoing leaving group such that carbon becomes pentavalent. This pentavalent atom carbon is highly unstable, therefore increasing energy of the transition state.