-
Call Now
1800-102-2727
Nucleophilic Substitution Reaction- SN1: What is SN1 Reaction? Mechanism, Energy profile diagram, Rate of the SN1 Reaction, Examples, Stereochemistry, Effect of solvent
Let us consider a small story of the cat and the comfortable chair to understand this topic better. Suppose a cat (nucleophile) lying on the floor wants to sit on a comfortable chair, but it needs to wait until the brown cat leaves it. Once it's empty, the other cat (nucleophile) occupies the comfortable chair. Now the cat owner calls the brown cat in order to give her something to eat. She immediately gets up and leaves the chair. And as soon as she leaves the chair, a grey cat(nucleophile) occupies the comfortable chair.

Considering this as an analogy for SN1 mechanism, we get a hint of what happens in the SN1 reaction. What we understood is that in SN1 reaction for substitution reaction to occur there must be some leaving group and nucleophile can attack only after leaving group has left. Now, let's get into the details of SN1reaction.
TABLE OF CONTENTS
- Introduction
- What is an SN1 Reaction?
- Mechanism of SN1 Reaction
- Energy Profile Diagram of SN1 Reaction
- Rate of the SN1 Reaction
- Examples of SN1 Reaction
- Stereochemistry of SN1 Reaction
- Effect of Solvent
- Practice Problems
- Frequently Asked Questions - FAQs
Introduction
SN1, The reaction mechanism follows a step-by-step process wherein first, the carbocation is formed by the removal of the leaving group. Then the carbocation is being attacked by the nucleophile. Finally, the deprotonation of the protonated nucleophile takes place to give the required product. The rate-determining step of this reaction depends on the reactivity of alkyl halide and is not impacted at all by the concentration of the nucleophile.
What is a SN1 Reaction?
SN1 reaction is a nucleophilic substitution reaction with an unimolecular rate-determining step. It's an organic substitution reaction of some sort. Thus, the SN1 reaction mechanism follows a step-by-step process wherein first, the carbocation is formed by the removal of the leaving group. The carbocation intermediate formed, is attacked by the nucleophile. Finally, the deprotonation of the protonated nucleophile takes place to give the required product.
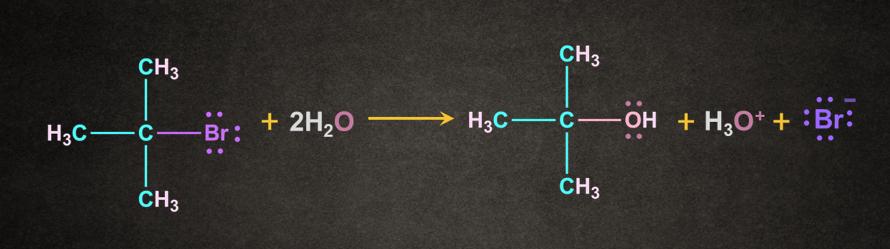
A carbocation intermediate is formed during this reaction. It is generally given by tertiary or secondary alkyl halides. Example of SN1 reaction is shown below.
Mechanism of SN1 Reaction
SN1 reaction involves two steps.
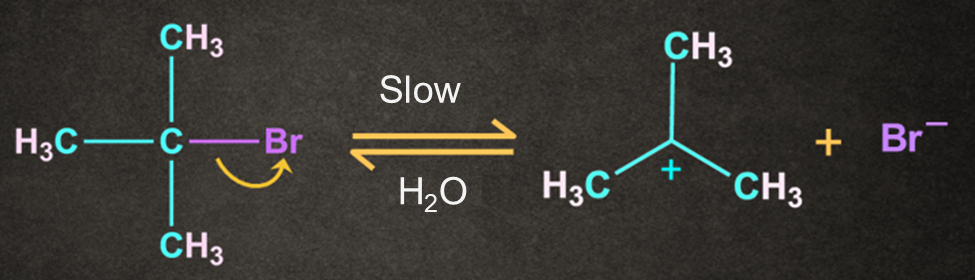
Step 1: Formation of carbocation
The bond between carbon and bromine is a polar covalent bond. The cleavage of this bond permits the leaving group to be removed as a bromide ion.
A carbocation intermediate is generated when the bromide ion departs from the tertiary butyl bromide.
As previously stated, this is the SN1 mechanism's rate-determining step.
The breaking of a carbon-bromine bond is an endothermic reaction.
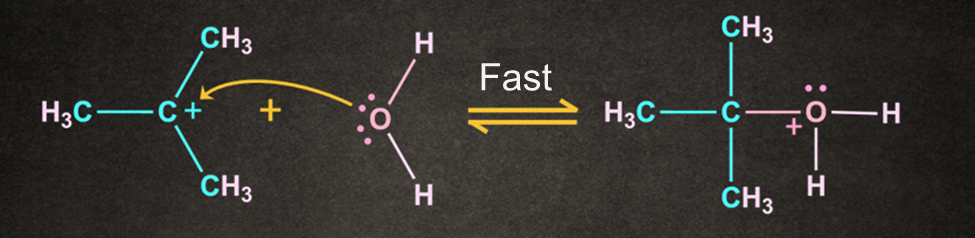
Step 2: Attack of the nucleophile
The nucleophile attacks the carbocation in the second step of the SN1 reaction mechanism.
The use of water as a nucleophile results in the formation of an oxonium ion intermediate. In this step, the positive charge on the carbocation was transferred to the oxygen.
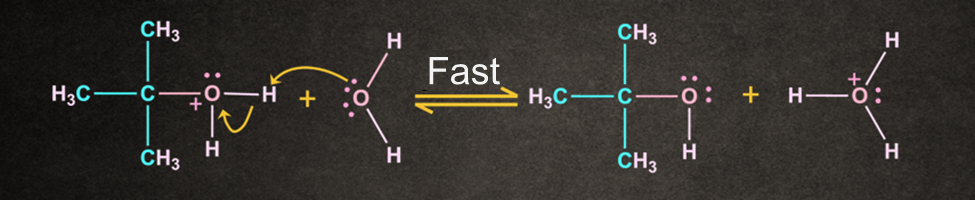
Because the solvent is neutral, deprotonation is required.
The water solvent now works as a base, deprotonating the oxonium ion and producing the required product alcohol as well as a hydronium ion as a result.
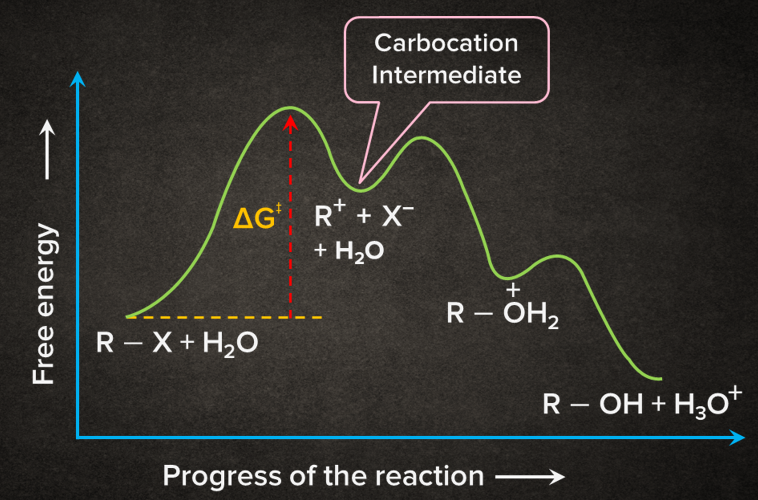
Energy Profile Diagram of SN1 Reaction
Rate of the SN1 Reaction
In SN1 reaction, the first step being the slowest step is the rate-determining step.
|
Rate ∝ [Alkyl halide] |
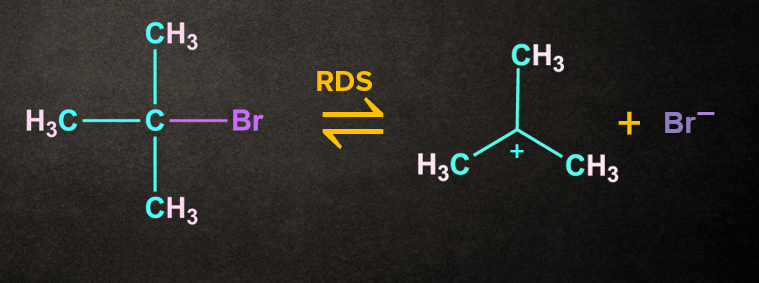
Since in the rate-determining step only alkyl halide is involved, it is a unimolecular reaction.
The rate of SN1 reaction is independent of the concentration of the nucleophile. A carbocation intermediate is formed, so rearrangement is possible in SN1 the reaction. Since carbocation is formed in the RDS so rate of SN1 reaction depends on the stability of carbocation.
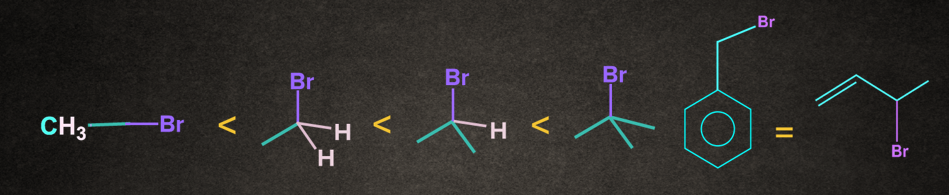
Nature of alkyl halide decides the reactivity of SN1 reaction. Increasing order of reactivity of alkyl halides is:
CH3–X <1o Alkyl halide <2o Alkyl halide < 3o Alkyl halide <Benzylic halide =Allylic halide
|
Alkyl bromide |
Class of alkyl bromide |
Relative rate of alkyl bromide |
|
|
Tertiary |
12,00,000 |
|
|
Secondary |
11.6 |
|
|
Primary |
≈ 0 |
Also, rate of reaction depends on R-X bond, for a given alkyl group. Weaker the R-X bond, more easily it can be cleaved for further nucleophile to attack.
|
Bond |
C-X Bond enthalpy (kJ mol-1) |
Bond length(pm) |
|---|---|---|
|
CH3-I |
234 |
214 |
|
CH3-Br |
293 |
193 |
|
CH3-Cl |
351 |
178 |
|
CH3-F |
452 |
139 |
Order of reactivity of R-X bond towards SN1 nucleophilic substitution is: R-I > R-Br > R-Cl >R-F
Note: SN1 nucleophilic substitution is a regioselective reaction, where the most stable carbocation gives the major product.
Examples of SN1 Reaction
1. 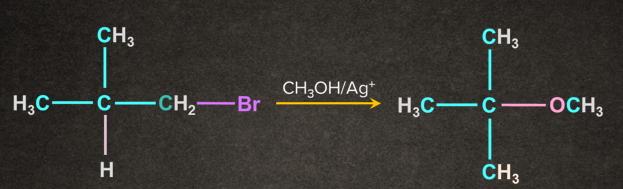
2. 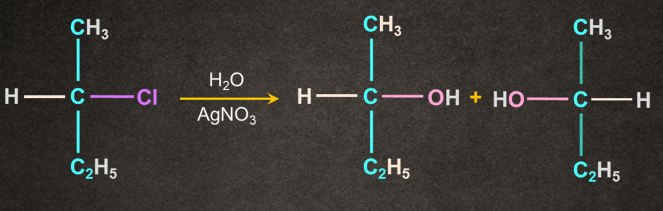
Note: If the solvent itself acts as a nucleophile in the absence of any other reagent, the reaction is known as solvolysis.
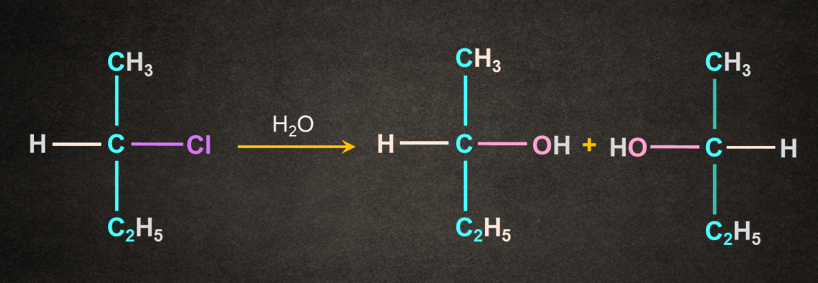
In this case water acts as both solvent and nucleophile.
Stereochemistry of SN1 Reaction
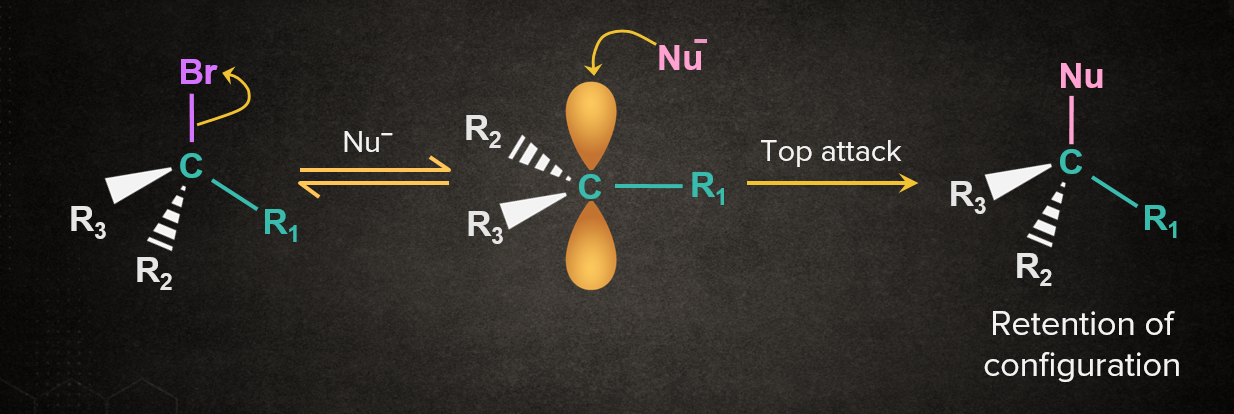
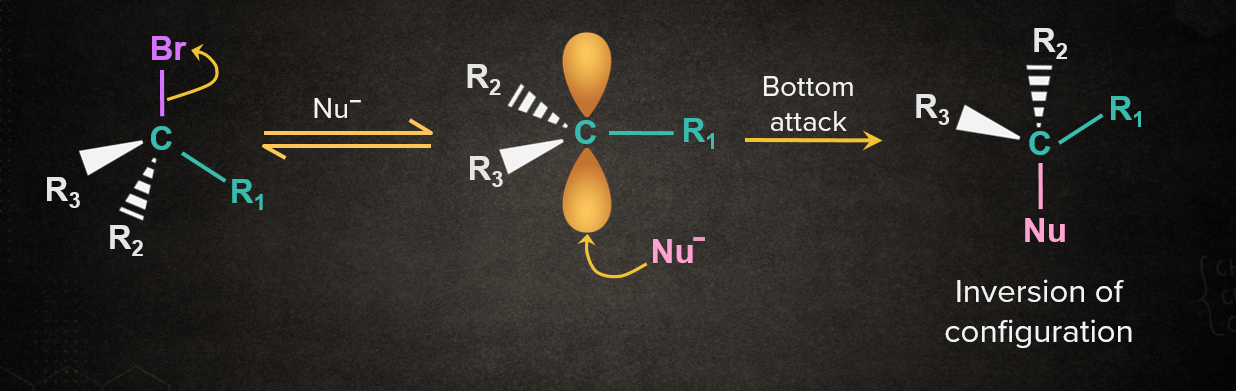
In the SN1 mechanism, the carbocation intermediate is sp2 hybridized and planar. A nucleophile can attack the carbocation from either face. If the reactant is chiral, then the attack of the nucleophile from both faces gives enantiomers as the product, which is known as racemisation. Both the cases are discussed below.
Case 1: When Nu- attacks from the same side as the leaving group.
Case 2: When Nu- attacks from the opposite side with respect to the leaving group.
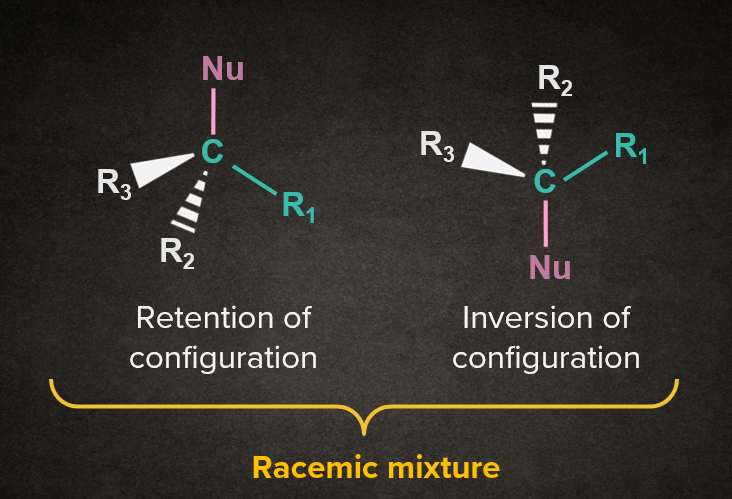
|
Note: Practically, we get slightly higher proportion of inverted product in SN1 reaction. Since we get one out of the two enantiomers as a major product, it is called partial racemization. |
Effect of Solvent
The rate-determining step of the SN1 reaction can be speed up by using a solvent that facilitates the formation of the carbocation intermediate.
Both polar and protic solvents are recommended for this type of reaction.
The polarity of the solvent helps in the stabilization of ionic intermediates formed in the reaction, whereas the protic nature of the solvent helps in the solvation and departure of the leaving group.
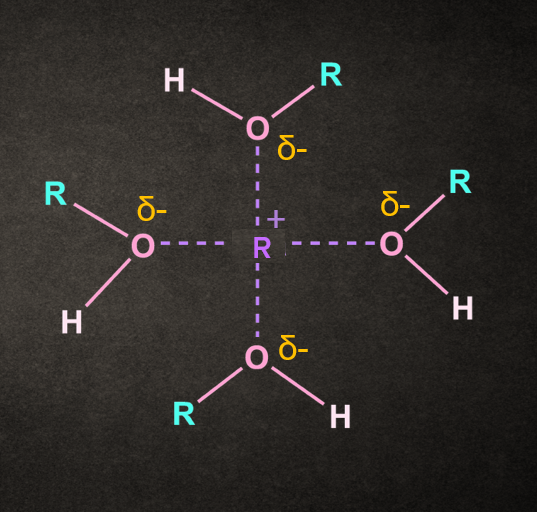
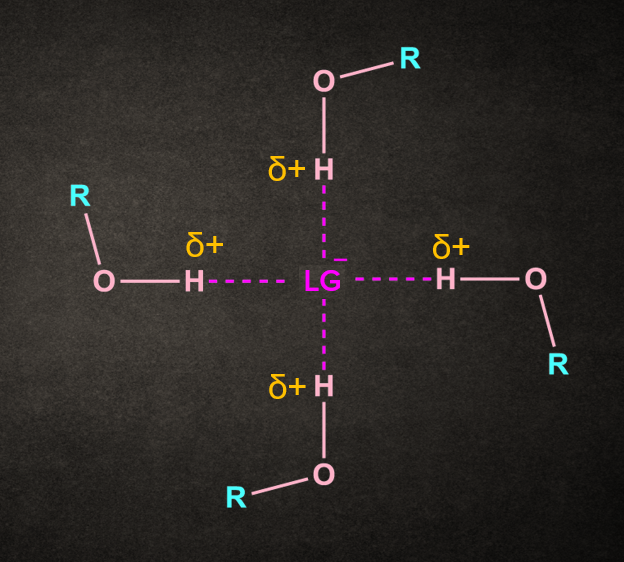
Water and alcohol are examples of solvents used in SN1 reactions. These solvents also have nucleophilic properties and act as a nucleophile.
Practice problems
1. Product of the following reaction is:
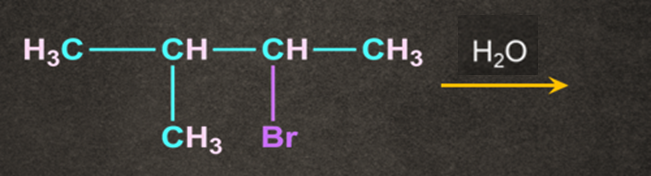
A).
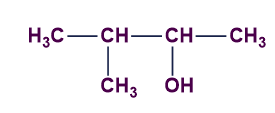
B).
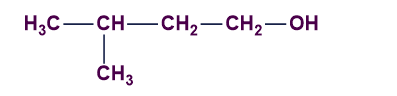
C).
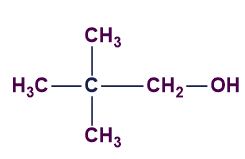
D).
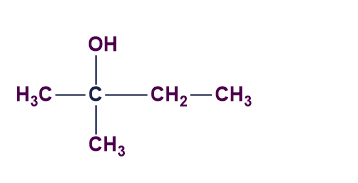
Answer: D
Solution:

2. Which of the following solvent can be preferred in SN1 reaction?
A). DMSO
B). NH3
C). H2O
D). Both B and C
Answer: D
Solution: SN1 reaction is facilitated by polar protic solvent. Polar protic solvents like NH3and H2O are preferred solvents for SN1 reaction.
3. Which of the following gives the fastest SN1 reaction?
A). 
B). 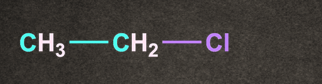
C). 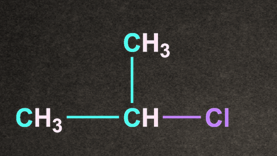
D). 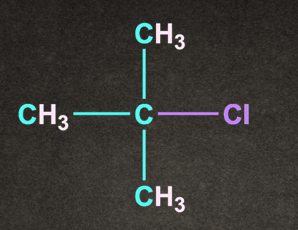
Answer: D
Solution: Degree of alkyl halide decides the reactivity of SN1 reaction.
CH3–X <1o Alkyl halide <2o Alkyl halide < 3o Alkyl halide <Benzylic halide =Allylic halide
More substituted the alkyl halide, it will rise to more stable carbocation.
Therefore option D, (CH3)3C-Cl generates more stable carbocation and will give the fastest SN1 reaction.
4. Which of the following will give stable carbocation intermediate?
A). 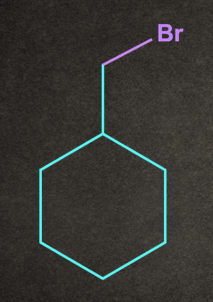
B). 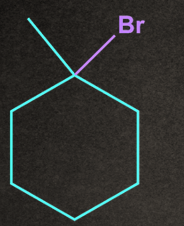
C). 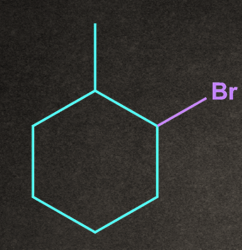
D). 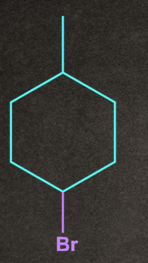
Answer: B
Solution: Tertiary carbocation will be formed in option b, so it will be the most stable among all by +H and +I effects.
Frequently Asked Questions-FAQs
1. What is a nucleophilic substitution reaction?
Answer: The replacement of an atom or group by any other atom or group in a molecule is known as a substitution reaction. If a substitution reaction is brought about by a nucleophile, then it is known as a nucleophilic substitution reaction.
2. What is a leaving group?
Answer: The part of the reactant molecule which gets cleaved is called a leaving group. Generally poor bases are a good leaving group.
1. Nucleofuge: Leaving group that carries away an electron pair or negative charge. For nucleofuge, weaker bases are good leaving groups.
2. Electrofuge: Leaving group that comes away without an electron pair or having positive charge.
Generally, the leaving group carries away an electron pair.
Example:
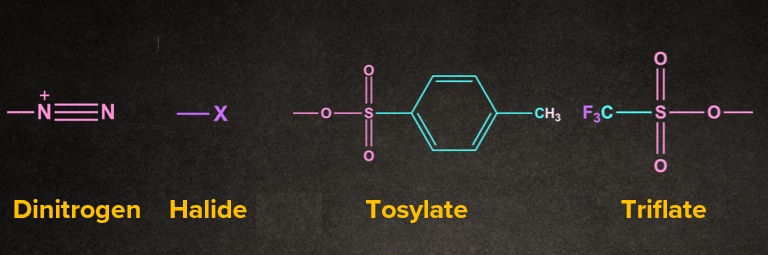
3. What is a rate-determining step (RDS)?
Answer: If one step in a multistep reaction is intrinsically slower than all the others, then the rate of the overall reaction will be essentially the same as the rate of this slowest step. This slowest step is known as the rate-limiting step or the rate-determining step.
4. What is a regioselective reaction?
Answer: A reaction which has more than one region to attack, which results in the formation of two or more products is called a regioselective reaction. Products obtained are constitutional (structural) isomers of each other. But in this mixture of products, one is the major product.