-
Call Now
1800-102-2727
Non-Aromatic Compounds - Non-Aromatic Compounds, Their Examples, Practice Problems and FAQs
Let's imagine that the 11th grade will be divided into sections based on these three criteria.
(i) Grades for the 10th grade
(ii) Performance on the basis of Quarterly science quizzes
(iii) Performance on the basis of Mid Semester

Students are considered for Section A if they receive an A+ on all of the aforementioned examinations or quizzes, Section B if they receive an A+ on any two of the exams or quizzes. Students who score A+ on any of the tests or quizzes may be considered for section C, while the remaining students will be placed in section D.
In a similar way, we can classify our organic compounds based on their aromaticity.
Let's consider the circumstances in which non-aromatic molecules arise!
Table of Contents
- Non-Aromatic Compounds
- Examples
- Practice Problems
- Frequently Asked Questions
Non-Aromatic Compounds:
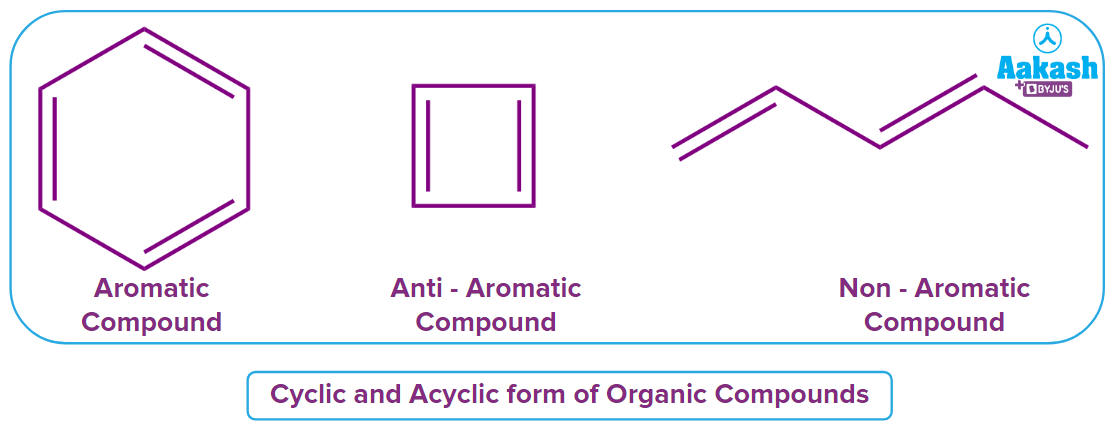
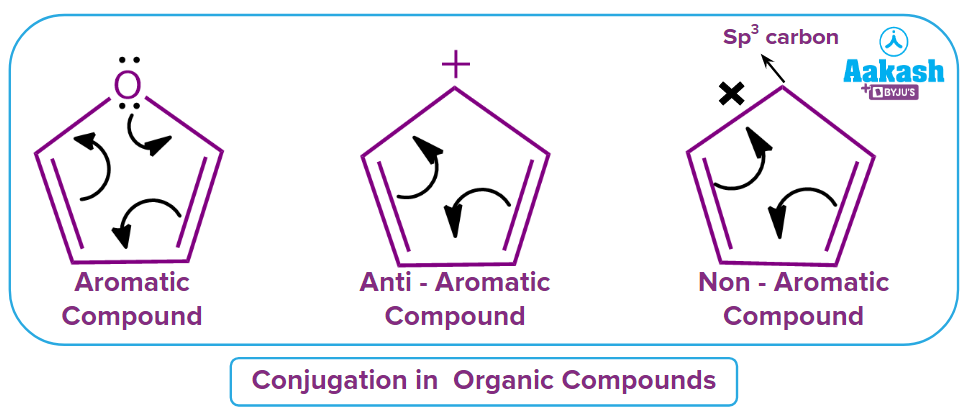
Cyclic compounds have the characteristic of aromaticity, which results in more stable molecules due to the planar structure and delocalized electrons in the orbitals. These substances fall under the categories of aromatic and anti-aromatic substances.
Huckel's rule of aromaticity states that cyclic and planar compounds are aromatic if the total number of electrons participating in conjugations is (4n+2) electrons in the given cyclic ring structure, where n is any natural number. If a cyclic and planar compound exhibits (4n) electrons participating in conjugation in the given ring structure, then the given compound is Anti-Aromatic in nature.
A compound is non-aromatic in nature if any of these given rules is not followed by it.
First and foremost, an organic compound must have a cyclic structure.
Acyclic organic molecules, which do not exist in a cyclic structure but in a linear or branched form, are non-aromatic compounds.
According to rule two, the cyclic ring must be planar.
If the cyclic compound is non-planar, then it is non-aromatic in nature.
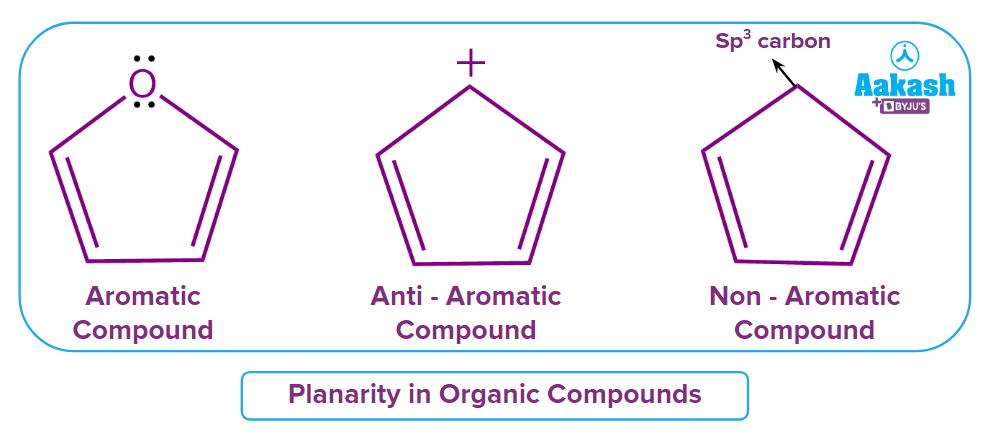
Rule three states that all electrons must be conjugated. There should be complete delocalisation in the compound.
If a cyclic system lacks conjugation, then the system is non-aromatic in nature. Because carbon is sp3 hybridized and has a full octet, the last transfer of electrons in the example is impractical, making delocalization impossible throughout the ring.
Examples:
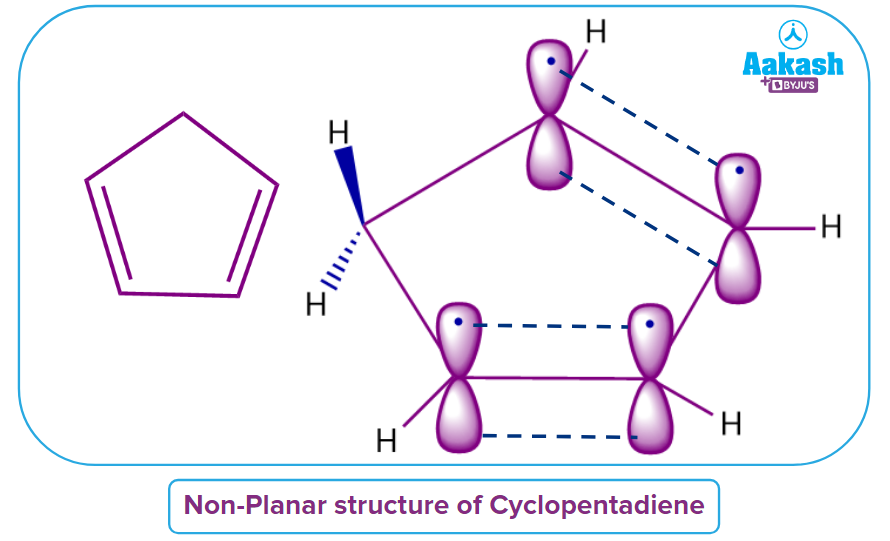
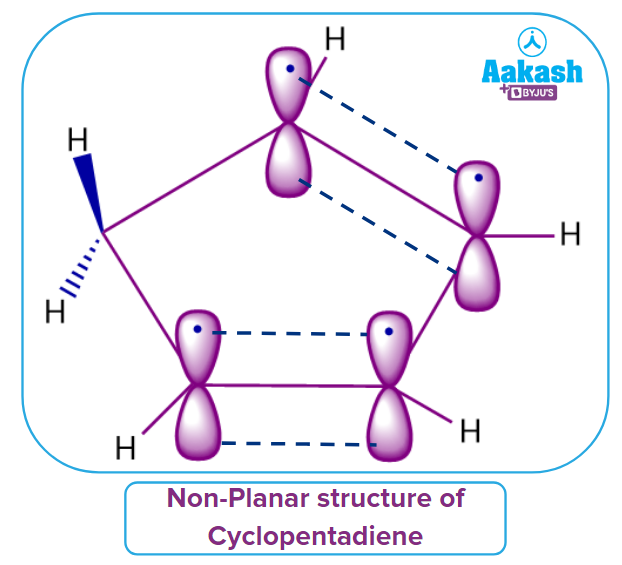
Cyclopentadiene
Let us discuss whether cyclopentadiene is a non-aromatic compound or not. See, Cyclopentadiene has a cyclic structure but not a planar molecule because it contains one sp3 hybridized carbon atom. Hence, it does not follow Rule 2. Cyclopentadiene fails to adhere to one of the established rules. The compound given is therefore Non-Aromatic in nature.
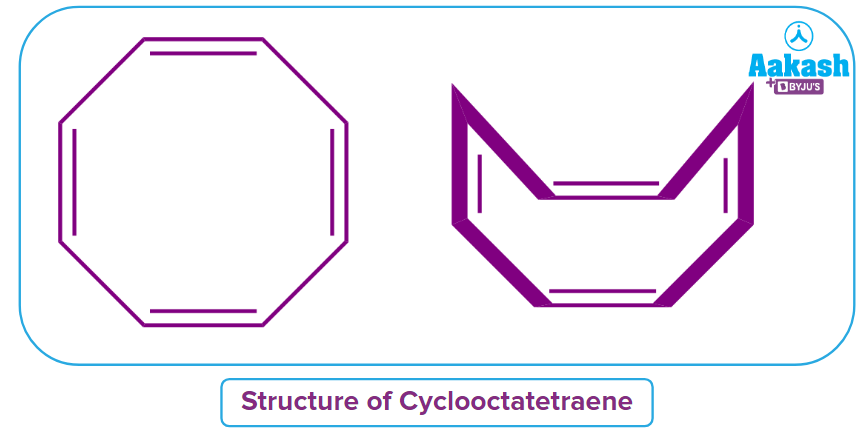
Cyclooctatetraene
We all know that non-aromatic compound is less stable than aromatic compounds. The given compound, cyclooctatetraene, appears to be Anti-aromatic in nature but it is actually a Non-aromatic compound. In the higher membered rings (C > 7), the compound loses its planarity to become non-aromatic and become a non-planar structure. It looks like a tub as shown in the image and is also referred to as a tub-shaped structure.
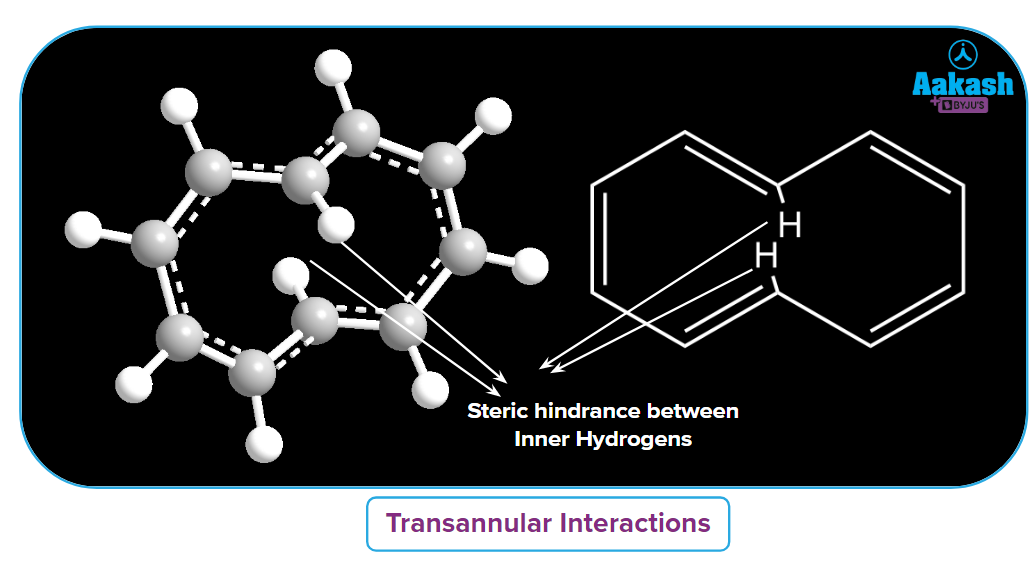
10-[Annulene]
Another case is [10]-annulene, which contains 10 electrons and satisfies the (4n+2) rule of Huckel. But it cannot adopt a planar geometry, hence it is non-aromatic. How?
Let's understand!
Transannular strain, also known as Prelog strain, is the unfavorable interaction of ring substituents on non-adjacent carbons in organic chemistry. These interactions result from the inner hydrogens of the ring being forced into conflict with one another due to a lack of space. Due to conflict between the inner hydrogen, the ring system loses its planarity and the system becomes non-aromatic in nature.
Related Video link: Aromaticity | General Organic Chemistry | Class 11 Chemistry | NEET 2022 & 2023 | Aakash NEET
Practice Problems:
Q1. Which of the following points is true for Non-aromatic compounds?
- A compound can be non-aromatic if it is not cyclic in nature.
- A compound can be non-aromatic if it is non-planar in nature.
- A compound can be non-aromatic if it is branched or linear in nature.
- All of these
Answer: (D)
Solution: Compounds can be regarded as Non-aromatic compounds if they are non-cyclic or in branched or linear form. If the compound is a non-planar or non-conjugated system, then the compound is also regarded as a non-aromatic compound.
Hence, all the given options hold true for non-aromatic compounds and the correct answer is option (D).
Q2. Which of the following compounds is a non-aromatic compound?
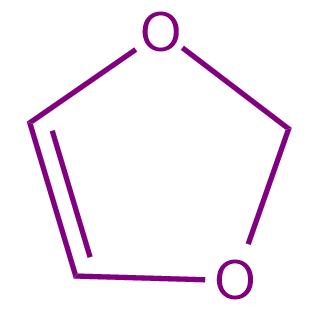
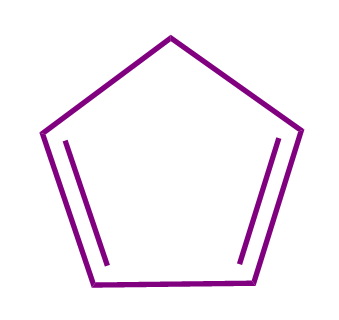
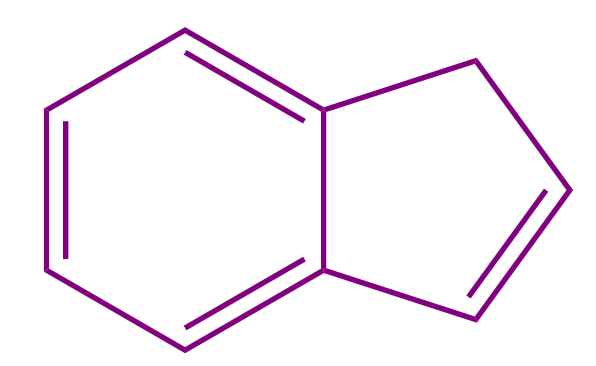
- All of these
Answer: (D)
Solution: All the given compounds contain one sp3 hybridized carbon due to which the compounds are non-planar in nature. If the organic compound is non-planar in nature, then it is regarded as non-aromatic in nature. Hence, all the given compounds are non aromatic and the correct answer is option (D).
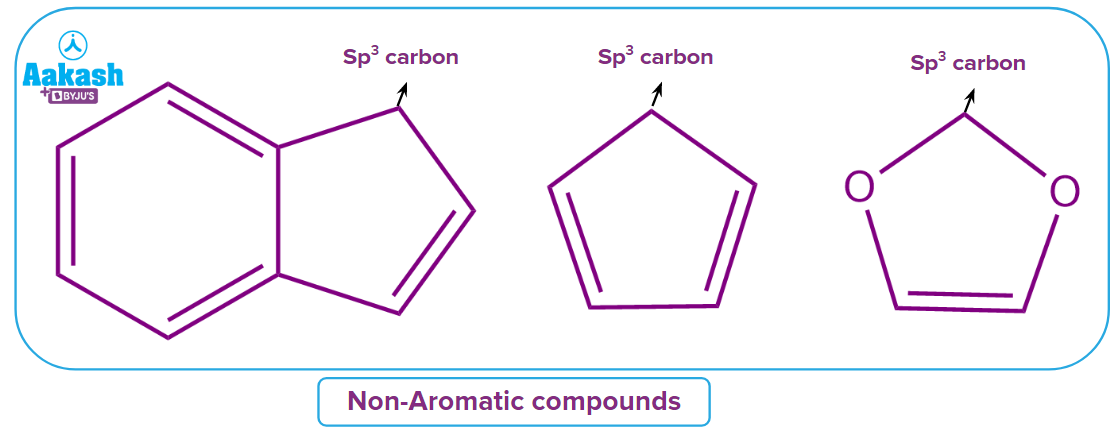
Q3. Predict the nature of a given compound?
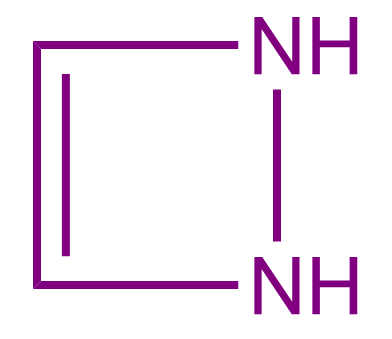
- Aromatic Compound
- Anti-Aromatic compound
- Non-Aromatic compound
- All of these
Answer: (C)
Solution: The given compound is non-aromatic in nature because delocalisation or conjugation throughout the ring is not possible here. Since nitrogen has its own complete octet, shifting electrons from double bonds to those between carbon and nitrogen is not feasible. Hence, if there is no conjugation in a cyclic compound, then the compound is considered a Non-aromatic compound.
Hence, the correct answer is option (C).
Q4. What should be the correct stability order for Anti-aromatic, aromatic, and non-aromatic compounds?
- Non-aromatic compound > Aromatic compound > Anti-aromatic compound
- Aromatic compound > Non-aromatic compound > Anti-aromatic compound
- Aromatic compound > Anti-aromatic compound > Non-aromatic compound
- Anti-aromatic compound > Aromatic compound > Non-aromatic compound
Answer: (B)
Solution: The correct order of stability order for Anti-aromatic, aromatic, and non-aromatic compounds can be decided on the basis of Huckel’s MOT(Molecular orbital theory).
Aromatic compound > Non-aromatic compound > Anti-aromatic compound
Hence, the correct answer is option (B).
Frequently Asked Questions-FAQs:
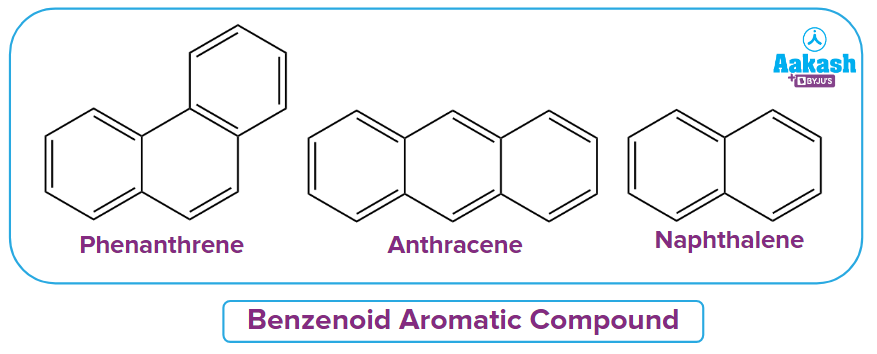
1. What are Aromatic benzenoid systems?
Answer: Aromatic compounds with two or more fused rings and at least one benzene ring are known as benzenoid aromatic systems. The entire system may be regarded as aromatic even if just one component (ring) is aromatic. Examples include phenanthrene, anthracene, and naphthene.
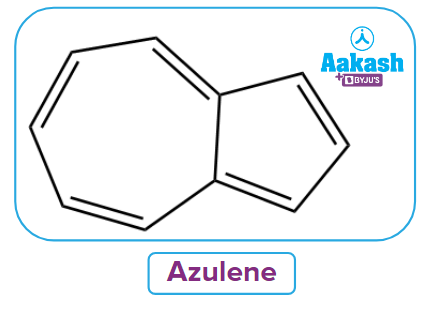
2. What are Aromatic non-benzenoid systems?
Answer: The term "non-benzenoid aromatic systems" refers to aromatic compounds without a benzene ring that has two or more fused rings. An example of a fusible aromatic non-benzenoid compound is azulenes. In anti-blemish cosmetic creams, these components are present.
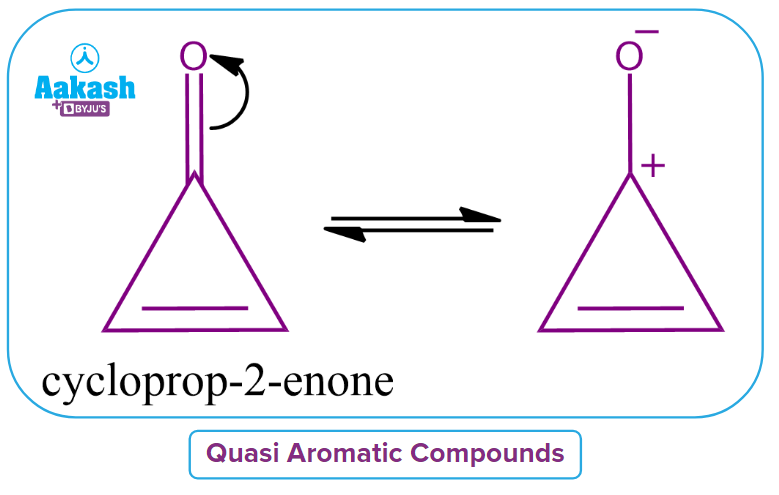
3. What are quasi aromatic compounds?
Answer: "Quasi-aromatic compounds" refer to aromatic species whose charge contributes to the compound's aromaticity. In their charged state, quasi-aromatic compounds are aromatic but not in their neutral state. As an example of cyclopropenone, its dipolar ionic form is aromatic.
4. What are heterocyclic compounds?
Answer: Heterocyclic compounds are cyclic compounds having an extra element, such as oxygen, nitrogen, phosphorus, or sulfur, attached to the carbon ring. A heterocyclic compound has at least two different atoms in its ring.