-
Call Now
1800-102-2727
IUPAC Nomenclature of Carboxylic Acids, Acid Anhydrides, Esters, Acyl Halides, Amides, Cyanides, Aldehydes, Ketones, Alcohols, Amines, and Ethers
When we change our clothes, we realise that our personalities change too. Similarly, replacing an "H" atom in any hydrocarbon with another atom or group of atoms affects the hydrocarbon's personality, i.e., its physical and chemical properties. This atom or group of atoms that define the properties of an organic compound is called a functional group. Each functional group has a different identity which is represented by a different prefix or suffix given by IUPAC.
A common question that comes to mind is “why is it required to name chemical molecules?”
The IUPAC system of nomenclature was formed to create a universal standard for naming substances to improve communication.
TABLE OF CONTENTS
- Functional Groups
- Rules to Assign IUPAC Names to Functional Groups
- IUPAC Naming of Carboxylic Acids
- IUPAC Naming of Acid Anhydrides
- IUPAC Naming of Esters
- IUPAC Naming of Acyl Halides
- IUPAC Naming of Amides
- IUPAC Naming of Cyanide
- IUPAC Naming of Isocyanide
- IUPAC Naming of Aldehyde
- IUPAC Naming of Thial
- IUPAC Naming of Ketone
- IUPAC naming of Alcohol
- IUPAC naming of Thiols
- IUPAC Naming of Amines
- IUPAC naming of Ethers
- Practice Problems
- Frequently Asked Questions - FAQ
Functional Groups
An atom or group of atoms that define the properties and unique chemical reactions of an organic compound is called a functional group.
For the naming of compounds containing functional groups, a secondary suffix is added after
the primary suffix in the IUPAC name of the compound to indicate the nature of the functional
group in the organic compound.
The basic skeleton of IUPAC Naming System
Secondary prefix + Primary prefix + Root word + Primary suffix + Secondary suffix
Secondary prefix - denotes the substituents attached to the parent chain
Primary prefix - denotes the nature of the parent chain (cyclo, bicyclo, spiro)
Root Word - denotes the number of carbon atoms in the parent chain
Primary Suffix - denotes the saturation of the parent chain
Secondary Suffix - denotes the functional group
Rules to Assign IUPAC Names to Functional Groups
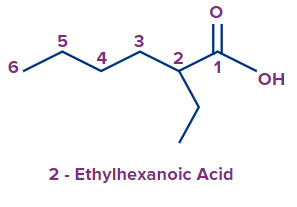
- The carbon atom of the functional group or the carbon bearing the functional group has to be included in the principal carbon chain even if it violates the longest chain rule.
Example:
|
|
|
|
Incorrect |
Correct |
- Priority order: Functional Group > double/triple bond > substituent.
Example:
|
|
|
|
Incorrect |
Correct |
- The locant of the functional group has to be written either before the name of the hydrocarbon (root word + primary suffix) or after the name of the hydrocarbon followed by the suffix of the functional group (drop the letter "e" from the primary suffix).
Example:
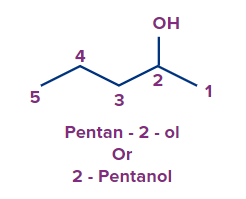
- If there is more than one identical functional group present in the molecule, then we can use di, tri, tetra, etc.
Example:
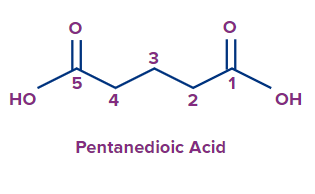
IUPAC Naming of Carboxylic Acids
This functional group is of the highest priority. While naming the compound, the C of -COOH is also considered in the name of the word root.
Secondary suffix - oic acid
Secondary prefix - carboxy
Special suffix - carboxylic acid
Examples:
|
Serial number |
IUPAC nomenclature |
Secondary Prefix |
Primary Prefix |
Word Root |
Primary Suffix |
Secondary Suffix |
|
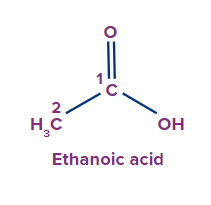
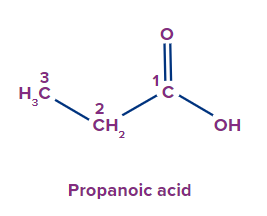
1 |
|
- |
- |
Eth |
ane |
-oic acid |
|
2 |
|
- |
- |
Prop |
ane |
-oic acid |
|
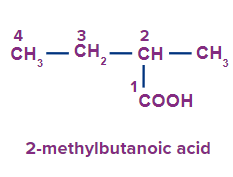
3 |
|
2-methyl |
- |
But |
ane |
-oic acid |
|
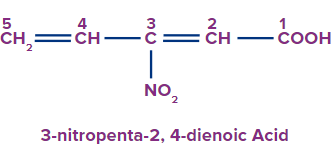
4 |
|
3-nitro |
- |
penta |
2,4-diene |
-oic acid |
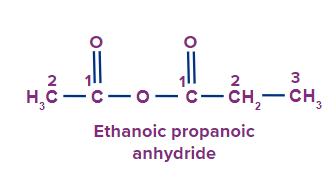
IUPAC Naming of Acid Anhydrides
An acid anhydride is obtained when two carboxylic acids react with each other with the removal of water.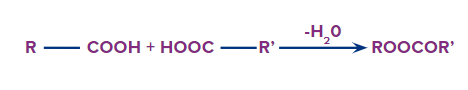
Formation of Acid Anhydride
The functional group next in priority (highest among the derivatives of carboxylic acids) is that of
acid anhydride(-COOCOR').
Secondary suffix: - oic anhydride
Examples:
|
Serial number |
IUPAC nomenclature |
Word Root |
Primary Suffix |
Secondary Suffix |
|
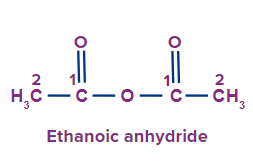
1 |
|
Eth |
ane |
-oic anhydride |
|
2 |
|
Eth Prop |
ane |
-oic anhydride |
|
3 |
|
But |
ane |
-dioic anhydride |
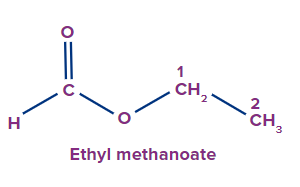
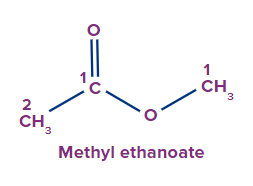
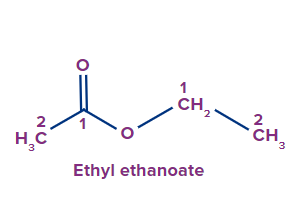
IUPAC Naming of Esters
An ester is obtained when a carboxylic acid reacts with alcohol with the removal of water.
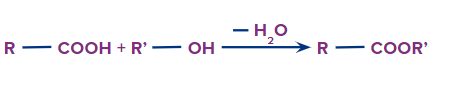
Formation of Esters
The functional group next in priority is that of ester (-COOR'). Generally, esters are named alkyl alkanoate. The alkyl group that is bonded to O (as OR) is termed alkyl, whereas the rest of the molecule is termed alkanoate.
Secondary suffix - oate
Special suffix - carboxylate
Examples:
|
Serial number |
IUPAC nomenclature |
Secondary Prefix |
Primary Prefix |
Word Root |
Primary Suffix |
Secondary Suffix |
|
1 |
|
ethyl |
- |
meth |
ane |
-oate |
|
2 |
|
methyl |
- |
eth |
ane |
-oate |
|
3 |
|
ethyl |
- |
eth |
ane |
-oate |
|
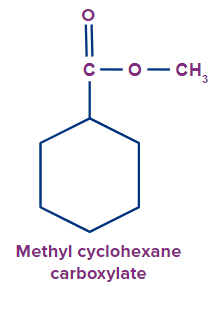
4 |
|
methyl |
cyclo |
hex |
ane |
carboxylate |
Secondary prefix for esters:
- Alkoxy carbonyl: When the C-atom of the ester (carbonyl carbon) is directly attached to the parent chain.
Example:
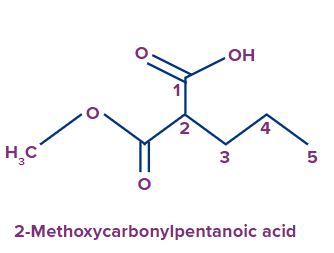
In the given compound, the ester group (methyl group attached to the oxygen) is directly attached to the parent chain at the second position from its carbonyl carbon. The IUPAC name of the compound is
2-methoxycarbonylpentanoic acid.
- Alkanoyloxy: When the O-atom of the ester is directly attached to the parent chain.
Example:
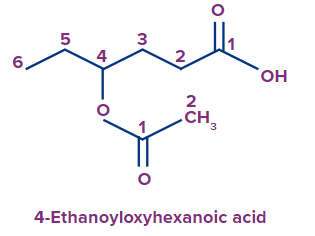
In the given compound, since the ester group is directly attached to the parent chain from the oxygen atom at the fourth position, the prefix ethanoyloxy is used for it. Hence, the IUPAC name of the compound is 4-ethanoyloxyhexanoic acid.
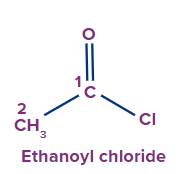
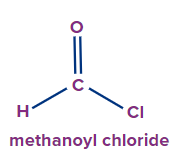
IUPAC Naming of Acyl Halide
An acyl halide is obtained when a carboxylic acid reacts with a halogen acid (HX).
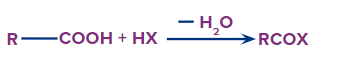
Formation of Acyl Halides
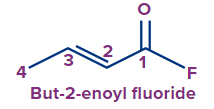
The functional group next in priority is that of acid halide (-COX). Here, X is a halogen (F, Cl, Br or I).
Secondary suffix - oyl halide
Secondary prefix - halo carbonyl
Examples:
|
Serial number |
IUPAC nomenclature |
Secondary Prefix |
Word Root |
Primary Suffix |
Secondary Suffix |
|
1 |
|
- |
eth |
ane |
-oyl chloride |
|
2 |
|
- |
meth |
ane |
-oyl chloride |
|
3 |
|
- |
but |
2-ene |
-oyl fluoride |
|
4 |
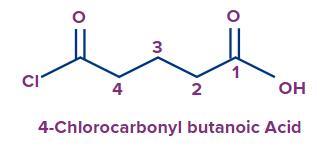
|
4-chloro carbonyl |
but |
ane |
-oic acid |
IUPAC Naming of Amides
An amide is obtained when a carboxylic acid reacts with ammonia (NH3).
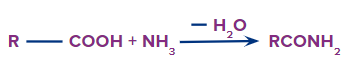
Formation of Amides
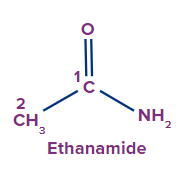
The functional group next in priority is that of amide (-CONH2, -CONHR, -CONR2).
Secondary suffix - amide
Secondary prefix - amino
Degree of amides:
Amides can be classified as 30 (tertiary), 20 (secondary), and 10 (primary) based on the number
of carbon atom(s) directly attached to the N atom.
Examples:
|
Serial number |
IUPAC nomenclature |
Secondary Prefix |
Primary Prefix |
Word Root |
Primary Suffix |
Secondary Suffix |
|
1 |
|
- |
- |
eth |
ane |
-amide |
|
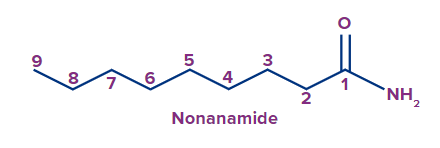
2 |
|
- |
- |
non |
ane |
-amide |
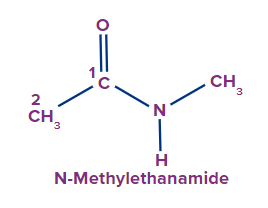
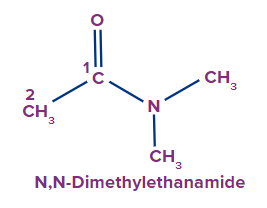
If H attached to N in amide is replaced by alkyl groups.
If one H attached to N is replaced by an alkyl group. It is written as an N-alkyl prefix. If two H are
replaced by two alkyl groups, then N,N-dialkyl prefix is used.
|
Serial number |
IUPAC nomenclature |
Secondary Prefix |
Primary Prefix |
Word Root |
Primary Suffix |
Secondary Suffix |
|
1 |
|
N-methyl |
- |
eth |
ane |
-amide |
|
2 |
|
N,N-Dimethyl |
- |
eth |
ane |
-amide |
|
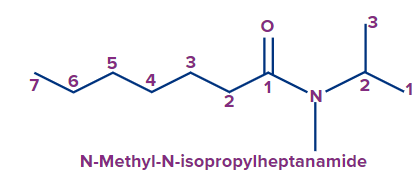
3 |
|
N-methyl- N-isopropyl |
- |
hept |
ane |
-amide |
Secondary Prefix for amide:
- Alkylaminocarbonyl: For amides (-CONHR), when the carbonyl C‒atom of the amide is directly attached to the parent chain, the prefix used is alkylaminocarbonyl.
Example:
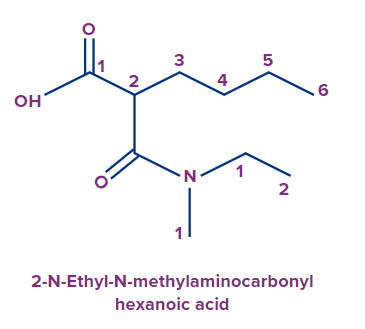
The IUPAC name of the compound is 2-N-Ethyl-N-methylaminocarbonylhexanoic acid.
- Alkanoylamino - For amides(-CONHR), when the N‒atom of amide is directly attached to the parent chain, the prefix used is alkanoylamino.
Example:
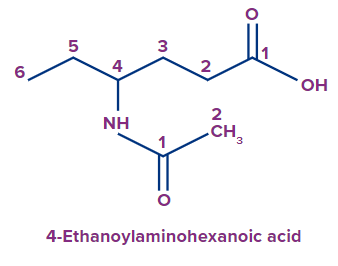
The IUPAC name of the compound is 4-Ethanoylaminohexanoic acid.
IUPAC Naming of Cyanide
The functional group next in priority is that of cyanide (—CN).
Secondary prefix - cyano
Secondary suffix - nitrile
Special suffix - carbonitrile
Examples:
|
Serial number |
IUPAC nomenclature |
Secondary Prefix |
Primary Prefix |
Word Root |
Primary Suffix |
Secondary Suffix |
|
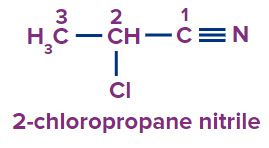
1 |
|
2-chloro |
- |
prop |
ane |
-nitrile |
|
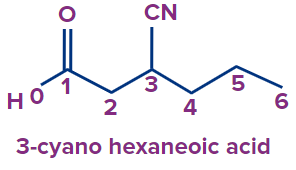
2 |
|
3-cyano |
- |
hex |
ane |
-oic acid |
|
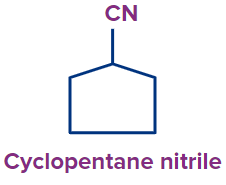
3 |
|
- |
cyclo |
pent |
ane |
-nitrile |
IUPAC Naming of Isocyanide
The functional group next in priority is that of isocyanide (—NC).
Secondary suffix - isonitrile
Examples:
|
Serial number |
IUPAC nomenclature |
Secondary Prefix |
Primary Prefix |
Word Root |
Primary Suffix |
Secondary Suffix |
|
1 |
|
- |
- |
meth |
ane |
-isonitrile |
|
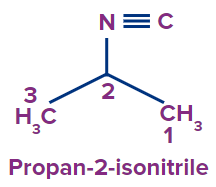
2 |
|
- |
- |
prop |
ane |
-isonitrile |
IUPAC Naming of Aldehyde
The functional group next in priority is that of aldehyde (—CHO). Here, we count the carbon of the
aldehyde group in the parent chain.
Secondary suffix - al
Secondary prefix for aldehyde:
- Oxo: Oxo is used as the prefix when C of CHO is included in the parent chain.
- Formyl: Formyl is used as the prefix when C of CHO is not included in the parent chain.
Examples:
|
Serial number |
IUPAC nomenclature |
Secondary Prefix |
Primary Prefix |
Word Root |
Primary Suffix |
Secondary Suffix |
|
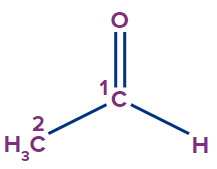
1 |
Ethanal |
- |
- |
prop |
ane |
-al |
|
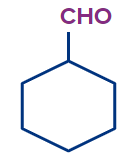
2 |
Cyclohexanecarbaldehyde |
- |
cyclo |
hex |
ane |
-carbaldehyde |
|
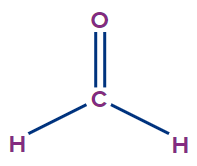
3 |
Methanal |
- |
- |
meth |
ane |
-al |
|
4 |
|
6-methyl-7-oxo |
- |
hept |
ane |
-oic acid |
|
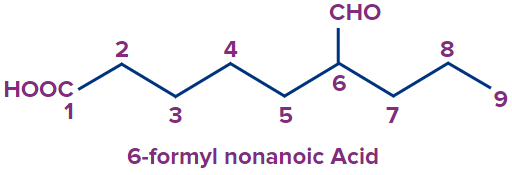
5 |
|
6-formyl |
- |
non |
ane |
-oic acid |
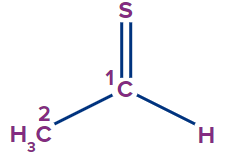
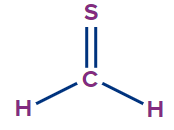
IUPAC Naming of Thial
The functional group thial is similar to aldehyde, in which the oxygen atom of aldehyde is replaced by sulphur atom.
Secondary suffix - thial
Example:
|
Serial number |
IUPAC nomenclature |
Secondary Prefix |
Primary Prefix |
Word Root |
Primary Suffix |
Secondary Suffix |
|
1 |
Ethanethial |
- |
- |
eth |
ane |
-thial |
|
2 |
Methanethial |
- |
- |
meth |
ane |
-thial |
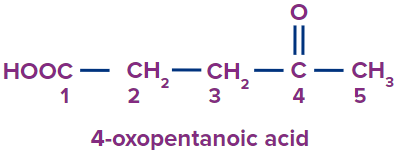
IUPAC Naming of Ketone
We count the number of carbons of the ketone group in the parent chain.
Secondary suffix - one
Secondary prefix for Ketone: Keto/oxo
Examples:
|
Serial number |
IUPAC nomenclature |
Secondary Prefix |
Primary Prefix |
Word Root |
Primary Suffix |
Secondary Suffix |
|
1 |
pentan-3-one |
- |
- |
pent |
ane |
-one |
|
2 |
butan-2-one |
- |
- |
but |
ane |
-one |
|
3 |
|
4-oxo |
- |
pent |
ane |
-oic acid |
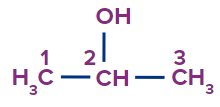
IUPAC Naming of Alcohol
The hydrocarbons containing the –OH groups are known as alcohols.
Prefix (if any) + Word root + Primary suffix (-e) + ol
While numbering the parent chain, give the lowest possible number to this functional group (−OH).
Secondary suffix - ol
Secondary prefix - hydroxy
If the primary suffix ends with a vowel, then it (vowel) is dropped while writing the IUPAC
nomenclature.
Examples:
|
Serial number |
IUPAC nomenclature |
Secondary Prefix |
Primary Prefix |
Word Root |
Primary Suffix |
Secondary Suffix |
|
1 |
Propan-2-ol |
- |
- |
prop |
ane |
-ol |
|
2 |
Butan-2-ol |
- |
- |
but |
ane |
-ol |
|
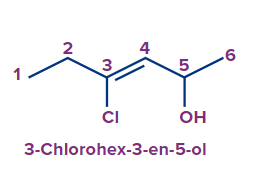
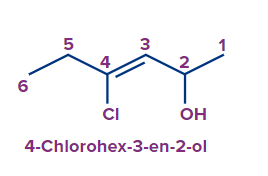
3 |
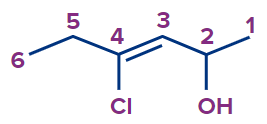
4-Chlorohex-3-en-2-ol |
4-chloro |
- |
hex |
3-ene |
-ol |
|
4 |
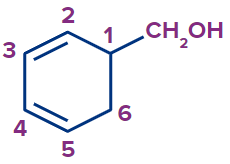
Cyclohexa-2,4-dienylmethanol |
hexa-2,4-dienl |
cyclo |
meth |
ane |
-ol |
IUPAC Naming of Thiols
The hydrocarbons containing the –SH groups are known as thiols.
IUPAC nomenclature: Add the suffix thiol after the primary suffix
Prefix (if any) + Word root + Primary suffix + thiol
Secondary suffix - thiol
Secondary prefix - mercapta
Examples:
|
Serial number |
IUPAC nomenclature |
Secondary Prefix |
Primary Prefix |
Word Root |
Primary Suffix |
Secondary Suffix |
|
1 |
Ethanethiol |
- |
- |
eth |
ane |
-thiol |
|
2 |
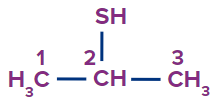
Propane-2-thiol |
- |
- |
prop |
ane |
-thiol |
|
3 |
Butane-2-thiol |
- |
- |
but |
ane |
-thiol |
IUPAC Naming of Amines
Amines may be regarded as the derivatives of ammonia (NH3) formed by the replacement of one or
more hydrogen atoms by the corresponding number of alkyl groups.
All three types of amines (primary, secondary, and tertiary) act as different functional groups.
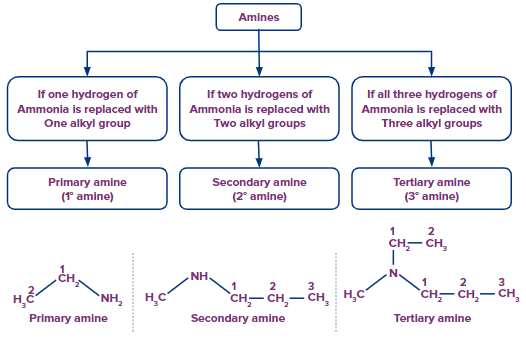
- Primary Amines
In the IUPAC system, the primary amines are named by replacing the final –e in the parent
hydrocarbon by -amine. Therefore, the IUPAC name of the primary amine is alkanamine.
Prefix (if any) + Word root + Primary suffix (-e) + amine
Secondary prefix for amine is amino.
Example:
|
Serial number |
IUPAC nomenclature |
Secondary Prefix |
Primary Prefix |
Word Root |
Primary Suffix |
Secondary Suffix |
|
1 |
Propan-2-amine |
- |
- |
prop |
ane |
-amine |
|
2 |
Butan-1-amine |
- |
- |
but |
ane |
-amine |
|
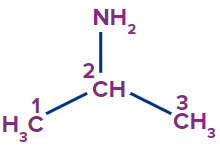
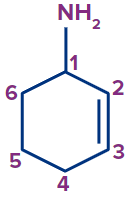
3 |
Cyclohex-2-en-1-amine |
- |
cyclo |
hex |
-2-ene |
-amine |
|
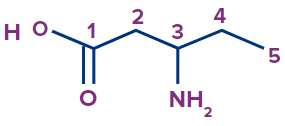
4 |
|
3-amino |
- |
pent |
ane |
-oic acid |
- Secondary and Tertiary Amines
Secondary and tertiary amines are named as N-substituted derivatives of primary amines.
- The name of the groups attached to the nitrogen of amine should be written alphabetically and the prefix ‘N-’ should be used for it as a position.
- The largest group attached to the nitrogen atom is taken as the alkyl group of the primary amine with suffix amine.
- The other groups are written as the alkyl groups with the prefix ‘N-’.
Note:
- If the secondary suffix is starting with a vowel and the primary suffix is ending with a vowel, then the vowel in the primary suffix is dropped while writing the nomenclature.
- While numbering the parent chain, give the lowest possible number to these functional groups (-NH2, -NHR, -NR2 )
Examples:
|
Serial number |
IUPAC nomenclature |
Secondary Prefix |
Primary Prefix |
Word Root |
Primary Suffix |
Secondary Suffix |
|
1 |
N-Ethylbutanamine |
N-ethyl |
- |
but |
ane |
-amine |
|
2 |
N-Methylpropanamine |
N-methyl |
- |
prop |
ane |
-amine |
|
3 |
|
N-ethyl-N- methyl |
- |
prop |
-2-ene |
-amine |
IUPAC Naming of Ethers
The hydrocarbons containing the –OR groups are known as ethers.
Ethers are always used as a substituents.
General IUPAC naming of ether is alkoxy alkane where the parent chain is alkane and alkoxy is used for substituents.
Secondary prefix - alkoxy
Examples:
|
Serial number |
IUPAC nomenclature |
Secondary Prefix |
Primary Prefix |
Word Root |
Primary Suffix |
Secondary Suffix |
|
1 |
Methoxymethane |
methoxy |
- |
meth |
ane |
- |
|
2 |
Methoxyethane |
methoxyl |
- |
eth |
ane |
- |
Practice Problems
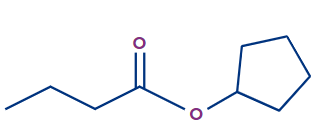
Q. 1. Write the IUPAC name of the given compound.
Solution:
The given compound is an ester.
The general IUPAC name of an ester is Alkyl alkanoate.
The alkyl group that is bonded to O (as OR) is termed 'alkyl', whereas the rest of the molecule is termed 'alkanoate'.
Alkyl branch: The alkyl group bonded to O is cyclopentyl. So, the cyclopentyl prefix will be used.
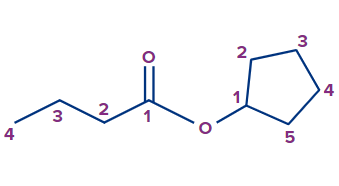
Alkanoate branch: Since the alkanoate branch has four carbon atoms, butanoate will be used for it.
Therefore, the IUPAC name of the compound is cyclopentylbutanoate.
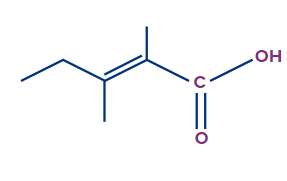
Q. 2. Write the IUPAC name of the compound.
Solution:
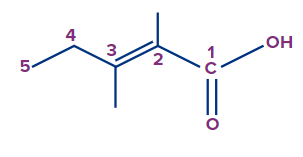
The given compound is a carboxylic acid.
Word root: The parent chain is of five carbon atoms. So, the word root will be pent.
Prefix: The compound contains two methyl substituents at the second and third carbons. So, the prefix 2,3-dimethyl will be used.
Primary Suffix: The compound contains a carbon-carbon double bond at the second position, so 2-ene will be used as the primary suffix.
Secondary Suffix: The compound contains carboxylic acid as a functional group, so -oic acid will be used as the secondary suffix.
Therefore, the IUPAC name of the compound is 2,3-dimethylpent-2-enoic acid.
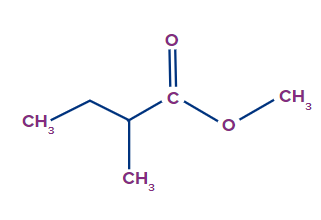
Q. 3. Write the IUPAC name of the compound.
Solution:
The given compound is an ester.
The general IUPAC name of an ester is alkyl alkanoate.
The alkyl group that is bonded to O (as OR) is termed 'alkyl', whereas the rest of the molecule is termed 'alkanoate'.
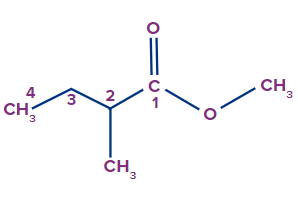
Alkyl branch: The alkyl group, which is bonded to O, is methyl. So, the methyl prefix will be used.
Alkanoate branch: Since the alkanoate branch is of four C-atoms with methyl connected to the second position, 2-methylbutanoate will be used for it.
Therefore, the IUPAC name of the compound is methyl-2-methylbutanoate.
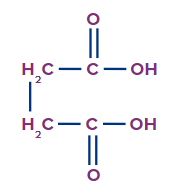
Q. 4. Write the IUPAC name of the compound.
Solution:
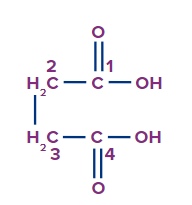
The given compound is a carboxylic acid.
Word root: The parent chain is of four carbon atoms. So, the word root will be but.
Prefix: There are no substituents attached to the parent carbon chain.
Primary Suffix: The compound contains Carbon-carbon single covalent bond, so ane will be used as a primary suffix.
Secondary Suffix: The compound contains carboxylic acid as a functional group, so -oic acid will be used as a secondary suffix.
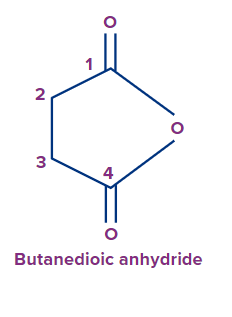
Therefore, the IUPAC name of the compound is Butan-1,4-dioic acid or 1,4-Butanedioic acid.
Q. 5. Write the IUPAC name of the compound.
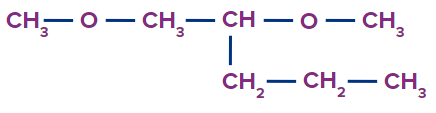
Solution :

The given compound is an ether. The general IUPAC name of ether is alkoxy alkane.
Word root: The parent chain is of five carbon atoms. So, the word root will be pent.
Prefix: 1,2-dimethoxy
Primary suffix: ane,
Therefore, the IUPAC name of the compound is 1,2-dimethoxypentane.
Q. 6. Write the IUPAC name of the compound.
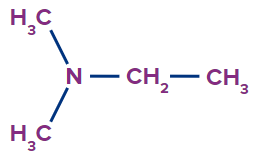
Solution:
The given compound is a tertiary amine.
Word root: The parent chain is of two carbon atoms. So, the word root will be eth.
Prefix: N,N-dimethyl
Primary suffix: ane
Secondary suffix: amine,
Therefore, the IUPAC name of the compound is N,N-dimethylethanamine.
Q. 7. Write the IUPAC name of the compound.
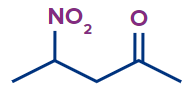
Solution :
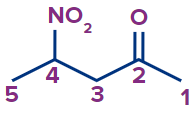
The given compound is a ketone.
Word root: The parent chain is of five carbon atoms. So, the word root will be pent.
Prefix: 4-nitro
Primary suffix: ane
Secondary suffix: -one,
Therefore, the IUPAC name of the compound is 4-nitropentan-2-one.
Frequently Asked Questions - FAQ
Q1. Which bond should be given more priority, double bond or triple bond?
Answer: While naming an organic compound that contains double and triple bonds, the double bond is given priority over the triple bond.
Q2. What is the priority order if a molecule contains carboxylic acid, ester and amide?
Answer: When a molecule contains polyfunctional groups, IUPAC naming is done according to the priority order.
Priority order of some common functional groups: Carboxylic acid > Ester > Acyl Halide > Amide > Aldehyde > Ketone > Alcohol > Amine.
Carboxylic acid comes first, followed by esters, and finally amide.
Q3. When is the longest carbon chain selection rule violated?
Answer: When at least two functional groups or multiple bonds are present, it has to be included in the principal carbon chain even though it violates the longest chain rule.
Q4. What is the need for the selection of the longest chain as a principal carbon chain?
Answer: Every organic compound is either a hydrocarbon or a derivative of hydrocarbon. The reason behind the selection of the principal carbon chain is to select the highest possible principal carbon chain by which the compound can be prepared.
Q5. When is ‘buta’ used in place of ‘but’ as a word root?
Answer: When more than one multiple bond is present in the principal carbon chain that contains at least four carbon atoms, then we use an extra letter "a" with the word root.
Q6. Can we arrange substituents in any order in IUPAC naming?
Answer: In IUPAC naming, the substituents are always arranged in their alphabetical order with the locants.
Q7. Are two different prefixes used for the same functional group?
Answer: Yes, in polyfunctional groups containing compounds, there will be two different prefixes used for a functional group depending on the connectivity of the group. Formyl and oxo are used in aldehydes, oxo and keto are used in ketone, and alkoxy carbonyl and alkanoyloxy are used in esters.



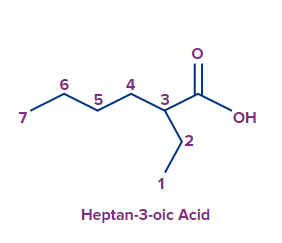
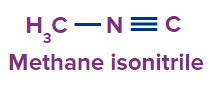
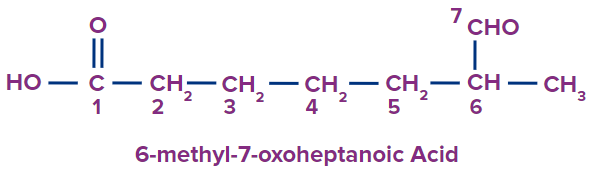
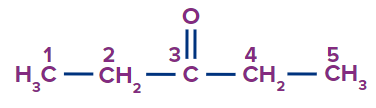
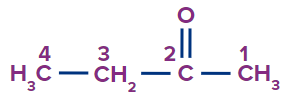
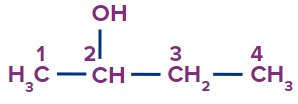
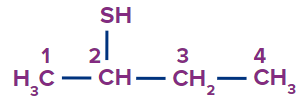
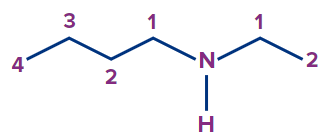
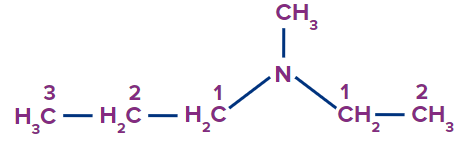 N-Ethyl-N-methylpropanamine
N-Ethyl-N-methylpropanamine