-
Call Now
1800-102-2727
Industrial Methods for the Synthesis of Methanol: Methanol, Structure, Industrial Methods for the Synthesis of Methanol, Uses, Practice Problems & FAQs
You might have known, Grignard reagents, amines, acids etc., as useful resources to prepare a variety of other organic molecules. Some, among them, can be prepared in multiple ways also, adding to their importance. Still, one among them is unique, for it can be prepared from a perennial natural source and also can supply energy.
Can you guess? It is difficult. It is methanol.
Methanol is synthesised from biomass and methane perennially and is used either as fuel or in the preparation of many large-volume chemicals such as formaldehyde. An additional advantage is that the production of methanol is an exothermic reaction, so as not only to satisfy the energy requirement of its production but also in electrical energy production.
Methanal or formaldehyde are frequently produced by catalytic oxidation using methanol as a chemical feedstock.
The production of plastics, including those used in construction materials, automobile parts, paints, explosives, and wrinkle-resistant imitation textures, heavily relies on the use of formaldehyde, which is produced from methanol.

Formaldehyde is used to preserve cadavers and lab specimens by both researchers and funeral directors.
Table of Contents:
- Introduction to Methanol
- Structure of Methanol
- Industrial Methods for the Synthesis of Methanol
- Uses of Methanol
- Practice Problems
- Frequently Asked Questions(FAQs)
Introduction to Methanol:
Methanol, or methyl alcohol, is the first member of the alkyl alcohols homologues. A flammable, colourless liquid with an ethanol-like odour. The original method of producing methanol was through the destructive distillation of wood, which gave rise to the term "woody alcohol." Methanol is now manufactured industrially by hydrogenating carbon monoxide. Methanol is mostly used as a chemical raw material in synthesising other compounds, but it can also be used as a fuel in some internal combustion engines. The chemical reaction formula for methanol combustion is given by,
As a result of its reactivity with metals, which can result in engine corrosion, methanol is not the best fuel to use. Methanol is a great option for the marine industry because it significantly reduces the emission of sulphur oxides, nitrogen oxides, and particulate matter into the atmosphere with only minor engine modifications. It is a substitute fuel.
In order to stop the abuse of recreational ethanol, methanol is also used as a denaturing substance. The licensed and licenced sale of denatured alcohol is governed by local governments all over the world. Methanol is also used as a solvent and antifreeze for pipe and windshield washer fluid daily.
Structure of Methanol:
Methanol has a chemical formula CH3OH. It is sometimes written as MeOH, where Me stands for Methyl, due to the fact that its chemical structure consists of a linkage between a methyl
(-CH3) group and a hydroxyl (-OH) group. Methanol has a molecular weight (or molar mass) of 32.04 g mol-1. The molecular structure of methanol can be represented as:
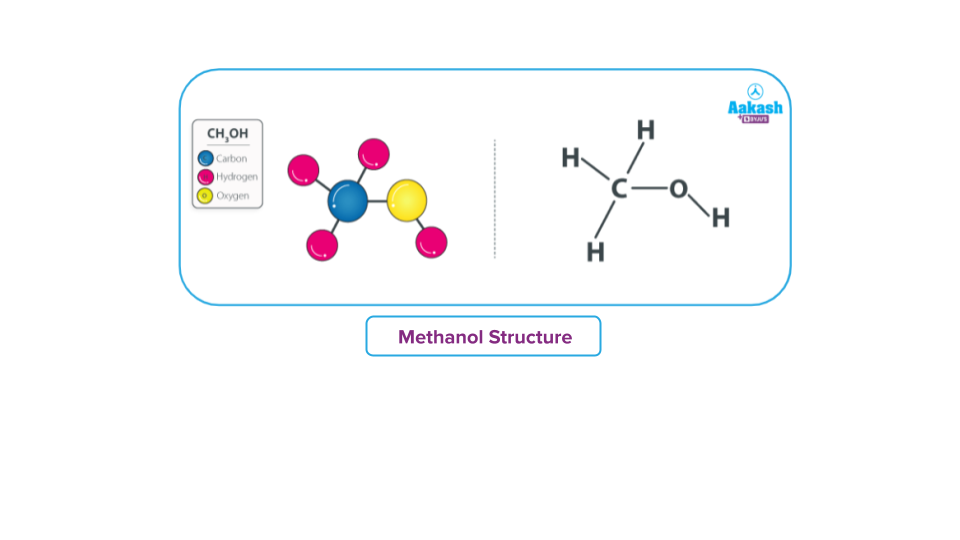
One carbon, one oxygen, and four hydrogen atoms constitute methanol (CH3OH). Since carbon (atomic number = 6) has a valency of 4, it requires four electrons to become stable. By forming covalent bonds with three hydrogen atoms and one oxygen atom of the hydroxyl group, carbon
in methanol is able to maintain its stability.
Methanol molecules have sp3 hybridisation in both carbon and oxygen. The molecule has a tetrahedral structure as a result of sp3 hybridization, but the bond angle H-O-C is less than 109.5o because of the repulsion due to oxygen's lone pair of electrons.
Industrial Methods for the Synthesis of Methanol:
- From Natural Gas.
Methanol is synthesised industrially in two phases in a typical plant.
- Production of CO, CO2 and H2 mixture and
- Production of methanol from the mixture.
The first stage is to transform the feedstock natural gas into a synthesis gas stream composed of carbon monoxide, carbon dioxide water and hydrogen. This is normally performed through the catalytic reformation of feed gas and steam. Another possibility is partial oxidation. The second stage is the catalytic synthesis of methanol from the synthesis gas. Each of these phases can be completed in a variety of ways, and diverse technologies provide a range of options that may be most appropriate for any given application.
i) By Steam Reforming:
Step-1: Production of synthesis gas:
The most basic and extensively used method of producing synthesis gas (CO+H2) is conventional steam reforming:
As can be observed, this procedure produces a significant hydrogen excess.
Step-2: Catalytic synthesis of methanol from synthesis gas:
The extra hydrogen can be used to produce more methanol if an external source of CO2 is accessible. The most advantageous gasification technologies "burn" the excess hydrogen to water and achieve steam reformation by the following partial oxidation reaction:
The excess hydrogen from the upper sequence of reactions would then combine with the carbon dioxide and hydrogen from the last equation to make more methanol. The utmost efficiency is provided, although there may be an additional capital expense.
The synthesis of methanol is very exothermic and occurs over a catalyst bed at moderate temperatures, in contrast to the reforming process. The majority of plant designs utilise this additional energy to produce the necessary amount of power.
ii) By Catalytic Hydrogenation:
The reaction between carbon monoxide and hydrogen is the oldest and most widely used method for producing methanol. The catalyst zinc or cupric oxide, which is coated on the surface of alumina, facilitates the direct combustion reaction between carbon monoxide and hydrogen. Because of its high selectivity, the reaction is also used in the commercial production of methanol.
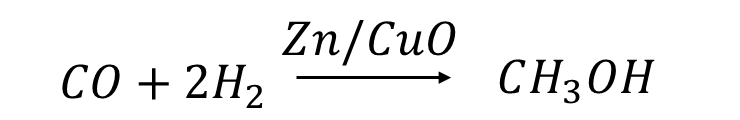
Traditionally, coal, oil, or natural gas were used as carbon monoxide sources in this method. This method, however, was not environmentally friendly and resulted in the depletion of these limited natural resources; thus, it has been replaced by a more environmentally friendly method known as "green" methanol production, which uses agricultural, industrial, and other urban wastes as a source of carbon monoxide. Another "green" way to produce Methanol is to use carbon dioxide as a waste product from a variety of processes.
B) Biochemical Method of Production of Methanol:
Fermentation is one of the simplest and oldest processes still in use today because it produced alcohol for the first time in nature. The catalytic conversion of methane to methanol can be carried out by specific enzymes, such as methane monooxygenases. Although the production of methanol by oxygenation is accompanied by the formation of other by-products like water and NAD+, this is not a problem because these can be separated by final processing steps. These enzymes are oxygenases by origin and perform a mixed function. The following is the biochemical process that produces methanol from methane gas with the aid of enzymes:
An energetic co-factor called NADPH contributes electrons and hydrogen atoms to processes that are catalysed by enzymes. During photosynthesis, the NADPH takes high-energy electrons and transports or transfers them to other molecules. In the initial phase of photosynthesis, it is created by the addition of electrons. Receiving or releasing electrons, aid in the oxidation and reduction of molecules during the process of photosynthesis. To create the carbon-carbon bonds in the sugar molecule, they transport protons and electrons.
The energy for the Calvin cycle in plants is also provided by NADPH, which is created during photosynthesis. Cellular respiration refers to the group of chemical metabolic processes that take place in living things to produce energy through the breakdown of food or sugar molecules by the action of oxygen. Oxygen and glucose are the reaction's reactants. The end results include ATP (Adenosine Triphosphate) and waste products like carbon dioxide and water. These processes start in the cell's cytoplasm and move on to the double-membrane organelle known as the mitochondria. The three metabolic pathways of glycolysis, the Krebs cycle, and the electron transport chain reaction are where it occurs.
A dinucleotide is nicotinamide adenine dinucleotide (NAD). It is made up of two nucleotides linked together by phosphate groups. Adenine base is present in one of them, while nicotinamide is present in the other.
3. Commercial Bioreactors:
Large fixed bed reactors that operate at high temperature (roughly 550- 575o C) and high pressure (about 100 atm) and alumina-coated copper and zinc oxides as a catalyst are now used to produce methanol on a commercial scale. It is discovered that using this method results in a 97 per cent conversion of the synthesis gas reactants into methanol. Numerous theories have been proposed to explain how methanol is created by this process, despite the fact that the mechanism by which it does so is still unknown.
Uses of methanol:
- Methanol has been used as a fuel, particularly biofuel, since ancient times.
- It is frequently used in car racing competitions or stunt driving performances because it is relatively less flammable than other fuel options and because it is easily soluble in water.
- Due to its polar nature, methanol is frequently used as a laboratory solvent for a variety of reactions.
- Other industrially useful chemicals like formaldehyde, acetic acid, and some heavy chemicals like methyl tertiary butyl ethers and other polymers are produced commercially using methanol.
- Alkyl halides, a class of compounds from which other significant compounds are easily formed through nucleophilic substitution reactions, are made using methanol.
- In many cold areas, including Europe, methanol is used as an anti-freeze agent.
- In order to prevent factory workers from having access to ethanol, methanol is used as an alcohol denaturant during commercial ethanol production.
- Methanol can undergo condensation reactions that are catalysed by heterogeneous catalysts like zeolites to produce hydrocarbons like gasoline, olefins, and other higher complex aromatic compounds.
- Since methanol thermally decomposes to produce carbon dioxide and water, it is occasionally used as an automobile fuel. The carbon dioxide created serves as a source of thermal energy for automobile combustion engines.
- Methanol works as a solvent for inks, adhesives, resins, and dyes.
- Methanol is frequently added to ethanol to stop alcohol abuse.
- Additionally, it serves as a solvent in the pharmaceutical industry. It is also used as an antifreeze for car radiators and in screenwash.
Practice problems:
Q1. Methanol on reaction with a carboxylic acid in the presence of sulphuric acid gives
(A) Ester
(B) Ether
(C) Amine
(D) None of the above
Answer: (A)
Solution: Alcohol converts into esters, a group of organic compounds that are found in organic chemistry when it reacts with carboxylic acids. Below is an illustration of how methanol and acetic acid react to form an ester (methyl ethanoate).
This process occurs in an acidic medium (preferably a dilute sulfuric acid solution is commonly used). Because methanol is simple alcohol, it can easily combine with any carboxylic acid to create an ester with a fruity scent. Esters are extremely useful in a variety of commercial and industrial contexts, including the food and fragrance industries.
Q2. What does producer gas consist of?
(A) Mixture of CO+N2
(B) Mixture of CH4+H2+CO2
(C) Mixture of CO+H2
(D) Mixture of H2+O22
Answer: (A)
Solution: Synthesis gas is a mainly a mixture of CO+H2. The name derives from its use as an intermediate in the production of SNG (Synthetic natural gas). A mixture of CO+N2 is called producer gas. A mixture of CH4+H2+CO is called coal gas.
Q3. Methyl alcohol on reaction with ethyl magnesium bromide (Grignard reagent)
(C2H5MgBr) gives
(A) Methane (B) Ethane
(C) propane (D) Butane
Answer:(B)
Solution: Methyl alcohol on reaction with ethyl magnesium bromide (Grignard reagent) (C2H5MgBr) gives methane. The reaction is as follows
Q4. Methyl alcohol on oxidation with potassium dichromate in the presence of an acidic medium gives
(A) Formaldehyde
(B) Acetaldehyde
(C) Propanone
(D) Propionaldehyde
Answer: (A)
Solution: Formaldehyde, a chemical used in industry and commercially, is formed through the oxidation reaction of methanol. Methanol reacts with oxygen in the presence of potassium dichromate in an acidic medium to produce formaldehyde and water. The reaction is expressed below.
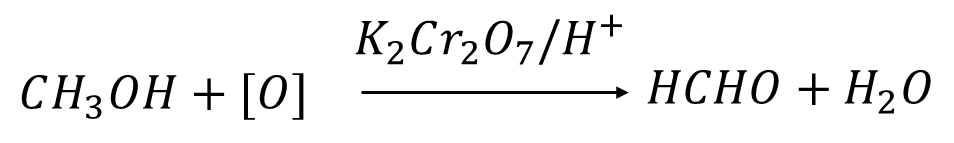
Frequently asked questions(FAQs):
Q1. How are methanol and ethanol distinguished from one another?
Answer: Methanol and ethanol can be distinguished from one another using the iodoform test. While methanol does not respond favourably to the Iodoform test when heated with iodine in the presence of sodium hydroxide(NaOH), ethanol does produce a yellow-coloured precipitate
(CHI3).
Q2. What is methanol poisoning?
Answer: Methanol consumption by an individual is a rare occurrence, but its harmful effects on the body cannot be disregarded. Although methanol is not toxic in and of itself, it becomes formic acid when consumed. Due to the high toxicity of this formic acid, it can cause severe metabolic acidosis. Additionally, 10 mL Methanol at low concentrations can damage the optic nerve and result in permanent blindness. Since ethanol is much safer than methanol, it is used to make hand sanitisers.
Q3. Is methanol acidic or basic?
Answer: Methanol is both acidic and basic. Proton donors are acids in the Bronsted-Lowry method, while proton acceptors are bases. Methanol is a proton donor, readily donating O-Hprotons to strong bases such as sodium hydride. Because it absorbs protons from strong acids such as sulfuric acid, methanol is a proton acceptor. Electron-pair acceptors are acids, and electron-pair donors are bases, according to Lewis' acid-base principle. Methanol is also referred to by this term. Because the proton is an electron pair acceptor on the O-H group and oxygen is an electron donor with two unpaired electrons, methanol is an acid. This type of substance, which is both acid and base, is known as amphoteric.
Q4. What causes methanol to be more acidic than ethanol?
Answer: Methanol has higher acidity than ethanol. This is due to the fact that in ethanol, a higher alkyl group, namely ethyl, is attached to the -OH group, increasing the electron density on oxygen via the +I effect and thus strengthening the O-H bond. The stronger the O-H bond, the weaker the acid.


