-
Call Now
1800-102-2727
Maxwell-Boltzmann Distribution Curve - Maxwell Equation, Observation, Effect of Temperature and Molar Mass on the Distribution Curve, Types of Molecular Speeds
What if all the students present in the classroom of a school are asked to run a 100-metre race? You will notice that some students will run very fast, some will run slowly, and some will run very slowly or just walk. A similar type of observation is seen in the gas molecules present in the closed container where particles of gas move randomly in all directions but, do you think particles present in the container will have the same speed? No! Some will move comparatively with higher speed but some will move with lower speed. Two scientists James Maxwell and Ludwig Boltzmann proposed a theory showing how the speed of the particles of a gas is distributed at a particular temperature and plotted a curve between the fraction of molecules vs molecular speeds. Let’s understand in detail what type of molecular speeds gas particles can have when present in a container at a specific temperature.

Table of content
- Maxwell-Boltzmann equation and distribution curve
- Observation of Maxwell-Boltzmann distribution curve
- Effect of temperature in Maxwell-Boltzmann distribution curve
- Effect of change in molar mass of gas on the Maxwell-Boltzmann distribution curve
- Types of molecular speeds
- Practice problems
- Frequently asked questions-FAQs
Maxwell-Boltzmann equation and distribution curve
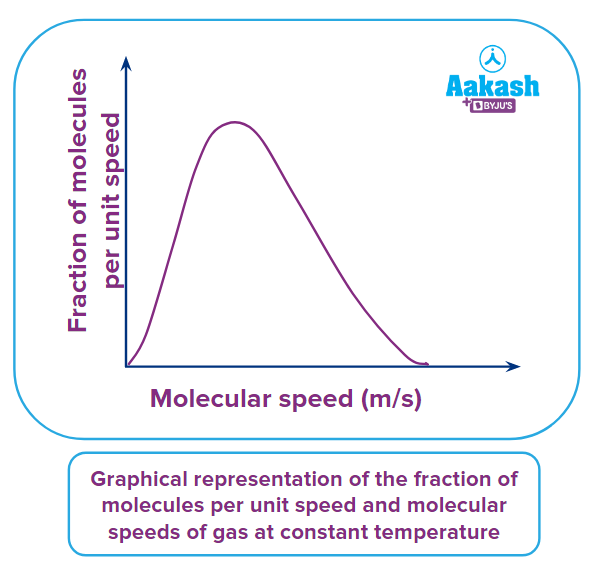
If we consider gas present in an enclosed chamber which consists of a large number of the molecule and at given temperature molecules are moving randomly. Gas molecules are colliding with each other and with the wall of the container due to which the speed of individual molecules is changing and it is, therefore, difficult to calculate the speed of individual molecules. Two scientists James Maxwell and Ludwig Boltzmann proposed a theory and plotted a curve between fractions of molecules per unit speed vs and molecular speed by using an equation known as the maxwell equation.
Where,

“k”, represent the Boltzmann constant
“m” represent the mass of the molecule
“T”, represent the temperature of the gas molecule
“u” represent the speed of the molecules

Observation from Maxwell-Boltzmann distribution curve

Few observations from the above Maxwell-Boltzmann distribution curve between the fraction of a number of molecules vs molecular speeds are as follows:
- The fraction of molecules in the extremely low-speed range and in the extremely high-speed range is very less.
- The speed possessed by most of the molecules is in between the very high and very low-speed range and this speed is known as the most probable speed.
- The total area under the curve represents the total number of molecules present in the container for a particular gas.
Types of molecular speeds
Molecular speeds are classified into three types:
Most probable speed (Ump)
Most probable speed is defined as the speed possessed by the maximum number of molecules at a given temperature.
Let us consider that at 100 ℃, out of 1000 molecules in a container if we consider, 700 molecules possess a speed of 50 ms-1, 100 molecules possess a speed of 50 ms-1
200 molecules possess a speed of 70 ms-1. Since the most probable speed is the speed possessed by a maximum number of molecules. Therefore, (Ump) in the case will be 50 ms-1.
Mathematically most probable speed(Ump) can be calculated using the formula:
“R”, represents the universal gas constant
“T”, represents the temperature of the gas in the Kelvin scale
“M”, represents the molar mass of the gas
Average speed (Uavg)
It is defined as the average of the speed possessed by all molecules present in the container.
In general, mathematically it can be represented as:
(∵ n1+n2+n3...........+nN=N)
Where,
u1 is the speed possessed by n1 molecules
u2 is the speed possessed by n2 molecules
uN is the speed possessed by nN molecules
Average speed of the gas molecules can also be calculated by using the formula:
Average speed (Uavg)
Where,
“R”, represents the universal gas constant
“T”, represents the temperature of the gas in the Kelvin scale
“M”, represents the molar mass of the gas
Root mean square speed (Ur.m.s)
It is defined as the square root of the mean of squares of speeds of the individual gas molecule.
In general, mathematically it can be represented as:
Where,
u1 , u2 , u2 …..uN represent the speed of the molecules
"N”, the total number of the gas molecules present in the container
Root mean square speed (Ur.m.s) of the gas can also be calculated using the formula:
Root mean square speed(Ur.m.s)
Where,
“R”, represents the universal gas constant
“T”, represents the temperature of the gas in the Kelvin scale
“M”, represents the molar mass of the gas

Recommended video:
Effect of temperature on Maxwell-Boltzmann distribution curve
When the Maxwell distribution curve is plotted between the fraction of molecules vs molecular speeds at different temperatures for the same gas. It has been seen that with the increase in temperature of the gas from T1 to T2 the curve get broadens up and molecular speeds (i.e.most probable speed, root mean square speed, average speed) of gas also increases as all type of molecular speeds of gas is directly proportional to 
Note: The total area under the curve in both cases will remain the same as it represents the total number of molecules present in the gas.

Effect of change in molar mass of the gas on the Maxwell-Boltzmann distribution curve
When the maxwell distribution curve is plotted for different gases at a constant temperature it is observed that gas having lower molecular mass has a higher most probable speed and the curve gets broadened and shifts to the right side as compared with the gas of higher molecular mass which have low most probable speed. But the total average translational kinetic energy per mole of gas will be the same for both lighter and heavier gas as it does not depend on the type of gas.

Practice problems
Q1. For the given Maxwell-Boltzmann distribution curve of the gas at different temperatures select the correct option.

A. Temperature of the gas with curve a is more than the curve c
B. Temperature of the gas with curve c is more than the curve a
C. Temperature of gas in all the three curves is equal
D. Temperature of the gas with curve b is maximum followed by curve a then curve c
Answer: (B)
Solution: Maxwell-Boltzmann distribution curve is plotted for fraction of molecules vs molecular speeds at different temperatures for the same gas. When the temperature of the gas increased, the curve gets flattened and the most probable speed of the gas also increases. So, the temperature of gas with curve c is greater than curve b and then curve a.
Q2. The ratio of Urms of O2(g) when temperature changes from 273 K to 546 K is:
Answer: (A)
Solution: Root mean square speed(Ur.m.s) of the gas can be calculated by using the formula:
Root mean square speed(Ur.m.s)
Let,
The root mean square speed of O2(g) at 273 K be U(r.m.s)1
The root mean square speed of O2(g) at 273 K= U(r.m.s)1
Let,
The root mean square speed of O2(g) at 546 K be U(r.m.s)2
The root mean square speed of O2(g) at 546 K = U(r.m.s)2
Dividing equation (i) by (ii) we get,
Q3. Select the correct option for the temperature at which Uavg of H2(g) is equal to Ur.m.s O2(g) at O℃.
A. 20.09 K
B. 10.09 K
C. 29.3 K
D. 120.9 K
Answer: (A)
Solution: Let the temperature at which Uavg of H2(g) is equal to Ur.m.s O2(g) be T(K)
Molar mass of H2(g)=2 gmol-1
Molar mass of O2(g)=32 gmol-1
Temperature of O2(g)= 273 K
Root mean square speed(Ur.m.s) of the gas can be calculated using the formula,
Root mean square speed (Ur.m.s) of O2(g)
Putting the value in the equation (i) we get,
Q4. What will be Ur.m.s of N2(g) at STP condition?
A. 5.59 ms-1
B. 14 ms-1
C. 3.59 ms-1
D. 15.59 ms-1
Answer: (D)
Solution: At STP condition, pressure exerted by the gas is equal to 1 bar and temperature corresponds to O ℃.
Molar mass of N2(g) = 28 g mol-1
Root mean square speed(Ur.m.s) of the gas can be calculated using the formula,
Root mean square speed(Ur.m.s)
Putting the given values in the equation (i) we get,
Ur.m.s=15.59 ms-1
Q5. In a container containing 10000 molecules of gas, 700 molecules have a speed of 400 ms-1, 500 have a speed of 600 ms-1, 2300 have a speed of 120 ms-1and the remaining 6500 have a speed of 500 ms-1.
The most probable speed of the gas molecules is:
A. 400 ms-1
B. 600 ms-1
C. 500 ms-1
D. Cannot be computed
Answer:(C)
Solution:
Most probable speed is defined as that speed which is possessed by the maximum number of molecules.
Since 6500 molecules out of the 10000 molecules have a speed of 500 m/s, the most probable speed must be 500 m/s.
Frequently Asked Question(FAQs)
Question 1. What is the use of the maxwell-Boltzmann distribution law?
Answer: Maxwell-Boltzmann law and its distribution curve are used to determine the distribution of speed for gas at a constant temperature. It also helps us in determining the factors that affect the molecular speeds of gas.
Question 2. What are the assumptions to be considered for the Maxwell-Boltzmann distribution curve?
Answer: The assumption to be considered for the Maxwell-Boltzmann distribution curve are:
- There is no interaction present between the gas molecules.
- Molecules of gases move randomly in all directions.
- The volume of individual molecules is very small as compared to the container in which it is kept.
Question 3. What is the significance of Maxwell-Boltzmann distribution?
Answer: Maxwell-Boltzmann distribution is also known as a probability distribution curve and helps us to determine the fraction of gas molecules having a particular speed at a specific temperature.
Question 4. Among three different molecular speeds of gas, which speed is the highest?
Answer: We know that there are three types of molecular speeds.
Therefore, from the above relation we get, Urms will have the highest value as compared to other two speeds.

Related topics
|
Kinetic theory of gases |
Gay-Lussac’s Law |
|
Avogadro’s Law |
Dalton's Law of Partial Pressure |
|
Graham’s law of diffusion |
Real Gas |


















