-
Call Now
1800-102-2727
Lanthanoids: Discovery, General characteristics, Uses, Comparison Between Lanthanoids and Actinoids, Practice problems, FAQs
Many of you generally when feel bored, you choose watching television for entertainment. Am I right?
Even children are so addicted to their shows that they even miss completing their important work.
This may happen to you while watching television, this question comes up how coloured is the screen looking. Have you thought about these things? What is there on the screen which makes us able to see this beauty?
My motive was to let you know about the special features of lanthanoids. In colour television sets, the lanthanides serve as phosphors as well. Chemicals called phosphors to shine in a variety of colours when struck by electrons. For instance, the red colour on television screens is made using yttrium and europium oxides.

You must have seen nowadays, science and technology are developing at a rapid speed.
Have you seen elements lie at the bottom of the periodic table? These are Lanthanides, or rare-earth metals, which are elements in the 4f-block. 5f-block elements are also known as actinides or actinones.
Let’s begin with the study of some interesting facts about lanthanoids and many of their important properties and general uses.
TABLE OF CONTENTS
- What are lanthanoids?
- General Characteristics of lanthanoids
- Important Physical and Chemical Properties of Lanthnoids
- Uses of Lanthanoids
- Comparison between lanthanoids and Actinoids
- Practice Problems
- Frequently Asked questions-FAQs
What are Lanthnoids?
Because of their striking resemblance to the element lanthanum (atomic no: 57), the f-block (inner transition) elements with partially filled 4f-subshells are known as Lanthanides or Lanthanones Lanthanides are a group of fourteen elements with atomic numbers ranging from 58 to 71.
Victor Goldschmidt coined the term "lanthanide" in 1925. The technical term "lanthanides," which derives from the Greek word lanthanein, which means "to lay hidden," is understood to represent a sense of elusiveness on the part of these elements despite their abundance.
The term refers to their ability to "hide" behind other minerals, rather than to their inherent abundance.
General Characteristics of Lanthnoids:
Electronic configuration:
For lanthanoids starting with cerium (Z=58) and finishing with lutetium (Z = 71), the electronic configuration of the inner transition elements is .
Example: Electronic configuration of Lanthanoids are mentioned below:
|
Atomic Number |
Element |
Symbol |
Configuration |
|
57 |
Lanthanum |
La |
|
|
58 |
Cerium |
Ce |
|
|
59 |
Praseodymium |
Pr |
|
|
60 |
Neodymium |
Nd |
|
|
61 |
Promethium |
Pm |
|
|
62 |
Samarium |
Sm |
|
|
63 |
Europium |
Eu |
|
|
64 |
Gadolinium |
Gd |
|
|
65 |
Terbium |
Tb |
|
|
66 |
Dysprosium |
Dy |
|
|
67 |
Holmium |
Ho |
|
|
68 |
Erbium |
Er |
|
|
69 |
Thulium |
Tm |
|
|
70 |
Ytterbium |
Yb |
|
|
71 |
Lutetium |
Lu |
Atomic size and Ionic size:
- The additional electron reaches the 4f-subshell in lanthanides but not the valence-shell or outermost shell.
- Because the shape of the f-subshell is very diffused, there is no similar rise in the mutual shielding effect of 4f-electrons, the shielding effect of one electron in the same subshell by another in the same subshell is very modest, even smaller than that of d-electrons.
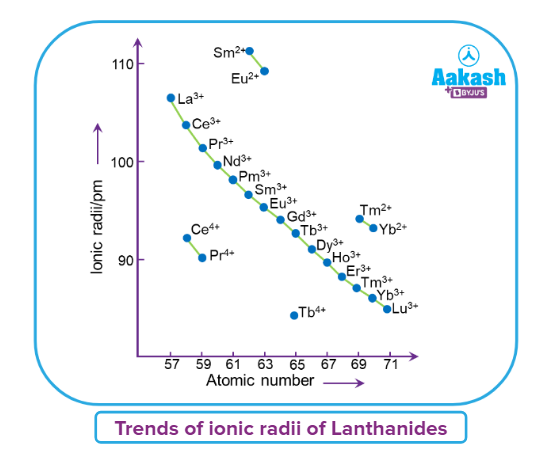
- The outermost shell electrons in the nucleus suffer more significant nuclear attraction as a result of this. As a result, as we go from La (57) to Lu (71), the atomic and ionic radii continue to decrease.
- Below is the graph which is showing the atomic radii of Lanthanides.
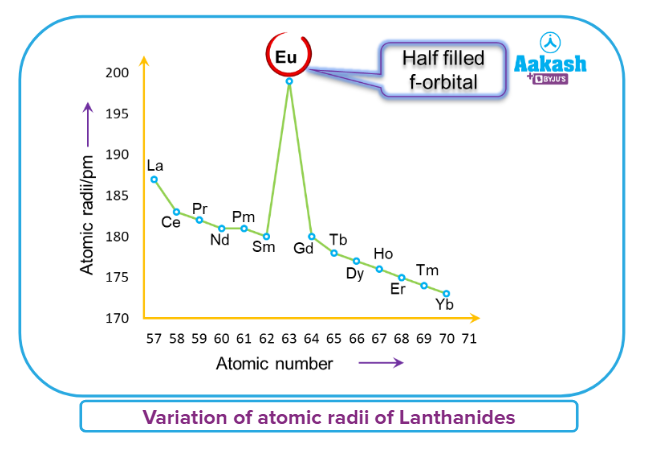
- The lanthanoid contraction (a decrease in atomic and ionic radii from lanthanum to lutetium) is a distinctive aspect of the lanthanoids' chemistry.
- It has far-reaching implications in the chemistry of the elements' third transition series. The drop in atomic radii (derived from metal structures) is not as consistent as it is in M+3 ions.
- Of course, this contraction is comparable to that seen in a regular transition series and is due to the same cause i.e; improper shielding of one electron by another in the same subshell. With the increase in nuclear charge down the series, however, the shielding of one 4f electron by another is smaller than that of one d electron by another. With rising atomic numbers, the diameters shrink in a pretty regular pattern.
Oxidation state:
- The +III is the most common oxidation state in lanthanoids. However, +2 and +4 ions are occasionally found in solution or in solid compounds.
- The increased stability of empty, half-filled, or filled f subshells causes this irregularity in ionisation enthalpies. The noble gas structure favours the creation of CeIV, although it is a strong oxidant that reverts to the common +3 state. Ce+4/ Ce+3 has a E value of + 1.74 V, indicating that it may oxidise water. However, because the reaction rate is slow, Ce(IV) is an excellent analytical reagent.
- Pr, Nd, Tb, and Dy have a +4 state as well, but only in oxides, MO2. Eu+2 is created by losing two s electrons, and its f7 configuration is responsible for its production. Yb+2, which has the f14 configuration, is also a reductant. Tb+4 is an oxidant with half-filled f-orbitals. Samarium behaves similarly to europium in that it has both +2 and +3 oxidation states.
Coloured ions:
Many lanthanide ions are coloured both in solid form and in solutions. Partially filled f-orbitals allow for f-f transitions, hence the colour. Colourless M3+ ions are those with 4f0, 4f7, or 4f14configurations. La3+,Gd3+,Lu3+ are colourless in nature.
The colour of pairs of M3+ ions with the same amount of unpaired electrons in 4f-orbitals are the same.
Colours of Lanthanides ions are mentioned in the below table:
|
M3+(M = lanthanide ion) |
Number of 4f electrons |
Colour |
|
La3+ |
0 |
colourless |
|
Ce3+ |
1 |
colourless |
|
Pr3+ |
2 |
Green |
|
Nd3+ |
3 |
Lilac |
|
Pm3+ |
4 |
Pink |
|
Sm3+ |
5 |
Yellow |
|
Eu3+ |
6 |
Pale pink |
|
Gd3+ |
7 |
Colourless |
|
Tb3+ |
8 |
Pale pink |
|
Dy3+ |
9 |
Yellow |
|
Ho3+ |
10 |
Pale yellow |
|
Er3+ |
11 |
Pink |
|
Tm3+ |
12 |
Pale pink |
|
Yb3+ |
13 |
Colourless |
|
Lu3+ |
14 |
Colourless |
Magnetic property
Ions with unpaired electrons are paramagnetic, whereas diamagnetic ions have all paired orbitals. Except for La3+ (4f0) and Lu3+ (4f14), all lanthanide ions (M3+) are paramagnetic due to the presence of unpaired electrons.
Complex formation
Due to their huge size and low charge density, lanthanides do not have a strong tendency to form complexes. However, as the atomic number rises, However, as the atomic number rises, the chances of complex formation and their stability also increase.
Some important Physical and Chemical Properties of Lanthnoids:
- Lanthanoids are silvery-white soft metals that tarnish quickly when exposed to air.
- With rising atomic numbers, the hardness of metal rises with samarium being steel hard metal.
- They have a conventional metallic structure and are good heat and electricity conductors.
- Except for Eu and Yb, and infrequently Sm and Tm, density and other characteristics shift smoothly.
- In the solid-state and in aqueous solutions, many trivalent lanthanoid ions are coloured. The existence of f electrons accounts for the colour of these ions.
- Neither the La+3 nor the Lu+3 ion exhibit any colour because of empty f-orbitals and fully filled f-orbitals respectively.
- Because of the excitation within the f level, the absorption bands are narrow.
- Other than the f0 type (La+3 and Ce+4) and the f14 type (Yb+2 and Lu+3), all lanthanoid ions are paramagnetic.
- The general half-cell reaction for lanthanides is
- When metals are heated with carbon, carbides such as Ln3C, Ln2C3, and LnC2 are produced.
- They react with halogens like F2,Cl2,Br2 to produce halides (LnX3) .
- On reacting with dilute acids, they liberate hydrogen gas.
- Lanthanides when burned in presence of air(O2) forms lanthanide oxides(Ln2O3).
Uses of Lanthanods:
The metals are rarely utilized in their pure form. Because lanthanides' physical and chemical characteristics are so similar, they are typically employed in alloys. Here are some frequent applications for lanthanides and related compounds:
- Misch Metal (an alloy) made up primarily of lanthanide metals (94–95%), iron (5%), with traces of sulphur, carbon, silicon, calcium, and aluminium.
- Cerium salts are frequently employed in lead accumulators, cotton dyeing, volumetric analysis, petroleum cracking (cerium phosphate), and other applications.

- These alloys are utilized to create ignition components including tracer bullets, shells, and lighter flints.
- Any alloy of 30% Misch metals (alloys of lanthanoids) and 1% Zr is utilised to create components for jet engines Powders for flashlights are made of cerium-magnesium alloys.
- Cerium salts are employed as a catalyst, in lead accumulators, and for dyeing cotton.
- Glass polishing is accomplished with lanthanum oxide. Goggles with coloured lenses are made using neodymium and praseodymium oxides. Gas mantles use CeO2 as a fuel.
- In volumetric analysis, ceric sulphate is a well-known oxidising agent.
- The phosphors in colour TV tubes is made up of a variety of lanthanide oxides.
- Different lanthanide compounds are employed as catalysts in the hydrogenation, dehydrogenation, oxidation, and petroleum cracking processes.
- Due to their paramagnetic and ferromagnetic properties, lanthanide compounds are employed to create magnetic and electrical devices.
Comparison between Lanthanides and Actinides:
Both lanthanides and actinides are f-block elements, meaning that their atoms contain anti-penultimate shells which contain a f-subshell. They have the following general electronic configuration:
where n for actinides is 7, and n for lanthanides is 6. They exhibit commonalities in features due to comparable electronic configurations, but they also exhibit differences in certain of their characteristics.
Similarities:
- Three of the outermost shells in the atoms of the elements in each series are only partially filled, whereas the remaining inner shells are fully filled.
- The outermost energy shell contains two electrons. The penultimate shell has 18 or 19 electrons, and the shell immediately after it has 18 to 32 electrons.
- The f-subshell is entirely filled in two elements, lutetium and lawrencium. These are the final elements in these two series respectively.
- Both series of elements primarily have a +3 oxidation state.
- Both series' constituents have an electropositive character. They function as potent reducing agents and reactive metals.
- As the atomic number rises, there is a contraction in atomic and ionic (M3+ ions) sizes in both of the series.In other words, actinide contraction is similar to lanthanide contraction. The weak shielding between the electrons dwelling in (n-2)f orbitals is the cause of these contractions.
- Cations in both series with unpaired electrons are paramagnetic.
- Actinides and lanthanides both exhibit ion exchange behaviour.
Dissimilarities:
The table compares some of the features of lanthanides with actinides.
|
Lanthanides |
Actinides |
|
All of the other lanthanides, with the exception of promethium, are non-radioactive. |
Each and every actinide is radioactive. |
|
In addition to the +3 oxidation state, lanthanides can occasionally exhibit the +2 and +4 oxidation states. |
In addition to the +3 oxidation state, Actinides can exhibit the +2, +4,+5,+6 and +7 oxidation states. |
|
Lanthanide oxides and hydroxides have lower basicity. |
Actinides oxides and hydroxides have higher basicity. |
|
It is simple to predict magnetic behaviour. |
Magnetic behaviour is difficult to understand. Typically, observed values and expected values do not match. |
Related link: https://www.youtube.com/watch?v=EAQGKdam8sE
Practice Problems:
Q1. The electronic configuration of cerium in the +3 oxidation state is
A.
B.
C.
D.
Answer: C)
Solution: The electronic configuration of cerium is . Cerium in the +3 oxidation state loses +3 electrons and thus gets a configuration of .
Q2. In flashlight powder alloy of ___________ is used.
A. Cerium-calcium
B. Cerium-magnesium
C. Samarium-magnesium
D. Samarium-calcium
Answer: B)
Solution: Flashlight powders contain cerium-magnesium alloys. Magnesium-based alloy is the world's strongest and lightest metal, and it has the potential to revolutionise the world.
Q3. The general electronic configuration of Lanthanides can be written as
Answer: C)
Solution: The general electronic configuration of lanthanides can be written as
Q4. _________________ acts as a strong reducing agent .
A. Europium
B. Cerium
C. Terbium
D. Lanthanum
Answer: A)Solution: Europium (atomic number 63) has the electronic configuration , in which it loses two electrons from the 6ssubshell and achieves the very stable, half-filled configuration, from which it quickly forms the Eu+2 ion. The typical oxidation states of lanthanides (+3) are then converted to Eu+3, which acts as a powerful reducing agent.
Frequently asked questions- FAQ
Q1. Are lanthanides good conductors of heat or electricity?
Answer: The lanthanides are good conductors of heat or electricity because the resistivity of lanthanides is relatively high.
Q2. What are the major lanthanides' sources?
Answer: The main source of lanthanides is monazite sand, which is made up of phosphates of lanthanum, thorium, cerium, and neodymium. The phosphate portion of monazite also contains trace amounts of other lanthanide ions. Promethium is the only lanthanide that is produced artificially through a nuclear reaction.
Q3. Which lanthanide is utilised in the production of magnets?
Answer: High-strength neodymium magnets, a form of a potent permanent magnet, are made using neodymium-based alloys. The strongest rare earth magnets like neodymium, combined with transition metals are utilised in some electric car motors and wind turbines.
Q4. Mention some lanthanide elements which are beneficial for health?
Answer: Erbium (Er) and Cerium (Ce) salts increase the number of red blood cells and the amount of haemoglobin in the blood, hence they are beneficial for health.


