-
Call Now
1800-102-2727
Isocyanide: Structure, Nomenclature, Preparation, Properties and Uses of Isocyanide, Practice Problems & Frequently Asked Questions
When purchasing shoes, comfort should always come first, right? Even though we adore the appearance of the shoe, we would not choose it if it were not comfy for our feet. The soothing feel of the shoes is a result of the shoe soles.
Do you know what these shoe soles are made of?
Polyurethane, which is created from isocyanides, is used to make shoe soles.

For hundreds of years, isocyanides were recognised to exist in natural resources. Penicillium notatum Westling's isocyanide chemical, Xanthoxylin, was isolated and utilised as an antibiotic. It was first made by combining silver cyanide and an alkyl halide. Additionally, they are more effective at producing fungicides like fludioxonil, heterocyclic organic compounds, and pharmaceuticals like ACE inhibitors.
On this concept page, let's learn more about isocyanides.
Table of Contents:
- Structure of Isocyanide
- Nomenclature of Isocyanide
- Preparation of Isocyanides
- Properties of Isocyanide
- Uses of Isocyanide
- Practice Problems
- Frequently Asked Questions(FAQs)
Structure of Isocyanide:
In the year 1867, the organic chemical isocyanide was discovered. The functional group of this molecule is - N ≡ C. Isocyanide is so called because it is an isomer of the cyanide - C ≡ N, functional group. In organic chemistry, the same functional group is referred to as Carbylamine, which is obsolete now.
An organic molecule known as an isocyanide has a triple bond between carbon and nitrogen as well as an alkyl or aryl group attached to the nitrogen. Two resonance structures can be used to characterise isocyanides: one has a triple bond between carbon and nitrogen, while the other has a double bond. The second structure is required due to the linearity of the carbon-nitrogen bond angle, which is close to 180 degrees, even though tests indicate that the structure with the double bond in carbon is a more significant resonance contributor.

Nomenclature of Isocyanide:
An atom or group of atoms known as a functional group governs the properties and unusual chemical reactions of an organic molecule.
When identifying compounds with functional groups, a secondary suffix is added after the primary suffix to the IUPAC name of the compound to specify the kind of functional group that is present in the organic compound.
The IUPAC Naming System's fundamental skeleton is
Secondary prefix + Primary prefix + Root word + Primary suffix + Secondary suffix
Secondary prefix - corresponds to the substituents connected to the parent chain
Primary prefix - corresponds to the nature of the parent chain (cyclo, bicyclo, spiro)
Root Word - corresponds the number of carbon atoms in the parent chain
Primary Suffix - corresponds the saturation of the parent chain
Secondary Suffix - corresponds the functional group
The compounds are named as alkyl cyanides if the -N-C group is at the terminal end, of a hydrocarbon chain. In accordance with the same, iso is prefixed to the name of the corresponding cyanide compound according to IUPAC nomenclature for the isocyanide group. For instance, methyl isocyanide or methyl carbylamine is the term given to CH3NC in the IUPAC nomenclature.
Secondary prefix - isocyano, if more priority group is present
Secondary suffix - isocyanide if isocyanide group is the only or most preferred substituent
|
Formula |
Common Name |
Secondary Prefix |
Primary Prefix |
Root Word |
Primary Suffix |
Secondary Suffix |
IUPAC name |
|
CH3NC |
Methyl isocyanide |
- |
- |
Meth |
ane |
isocyanide |
Methyl Isocyanide |
|
CH3CH2NC |
ethyl isocyanide |
- |
- |
Eth |
ane |
isocyanide |
Ethyl Isocyanide |
|
CH3CH2CH2NC |
Propyl isocyanide |
- |
- |
Prop |
ane |
isocyanide |
Propyl isonitrile |
|
C6H5NC |
Benzyl Isocyanide |
- |
- |
Phenyl |
- |
isocyanide |
Phenyl isocyanide |
Preparation of Isocyanides:
- Alkyl isocyanides are prepared in the laboratory by heating an alcohol solution containing the corresponding alkyl halide and silver cyanide.
C2H5Cl + AgCN → C2H5NC + AgCl
- Alkyl and aryl isocyanides are formed by heating the alkyl or aryl primary amines with chloroform in the presence of ethanolic potassium hydroxide.
Chloroform forms an active electrophilic reagent carbene with ethanolic potassium hydroxide. Carbene reacts with the amine to form an aryl or alkyl isocyanide.
RNH2 + CHCl3 + 3KOH → RNC + 3KCl + 3H2O
The reaction is referred to as the carbylamine reaction.
- By the dehydration of the corresponding formamide using a dehydrating agent like phosphoryl chloride. Dehydration phenyl formamide produces phenyl isocyanide.

Properties of Isocyanides:
- A triple bond between carbon and nitrogen makes up the chemical compound isocyanide. Isocyanide is an extremely hazardous molecule since this sort of bond is very reactive. Isocyanide gas inhalation can quickly result in fatality through paralysis of the respiratory system.
- It has been looking into developing non-lethal isocyanide weapons in the past. The isocyanides that smell like aged wood, malt, distinctive elastic, creosote, and cherry are among the least toxic isocyanides.
- An example of a non-volatile derivative without an odour is tosylmethyl isocyanide.
- Because of their high electron density, isocyanides are extremely reactive. Water, alcohols, and amines are just a few of the chemicals they can react with. Isocyanides turn into cyanides when they combine with water. Cyanide gas, a chemical weapon, is created through this reaction.
- Polyurethanes are also created using isocyanides. A form of plastic called polyurethane is utilised in many different products, such as furniture, insulation, and vehicle parts.
- Numerous plants and foods, including almonds, apricots, and honey, contain trace levels of isocyanide.
Uses of Isocyanides:
- Isocyanates are widely used and can be found in a variety of projects and professions, such as painting, construction, shipbuilding, upholstery manufacturing, and firefighting.
- Isocyanide-based multicomponent reactions can encompass a variety of substantial mixtures because of their useful gathering resilience and indisputable levels of chemo, stereo, and regioselectivity.
- As a fumigant and industrial chemical, isocyanide is used. An organic substance with the formula -NC is isocyanide. It is an unpleasant-smelling, colourless gas that is toxic. It is used on a big scale for the production of polyurethanes and serves as a structural component of some polyurethanes. Medicines, herbicides, and insecticides are all made using it.
- Globally, polyurethane foam is a type of insulation that is mostly utilised in coolers and refrigerators. The ability of polyurethane foams to insulate effectively is a crucial quality for maintaining low food temperatures during production, storage, and transportation to consumers. A significant impact on our daily lives would result from the approximately 50% of food produced worldwide that would spoil without polyurethane-insulated cooling.
- The best application for polyurethane in modern buildings is as an insulation material. A reliable, long-lasting, and secure method of reducing the CO2 emissions that contribute to global warming is polyurethane. In actuality, effective insulation retains the heat outside during warm months and maintains the interior temperature during cold days.
- Isocyanides are a broad category of metabolites that are produced by a variety of organisms on Earth, including aquatic life and plants.
Isocyanide Intoxicity:
When a human is exposed to excessive doses of isocyanide, isocyanide poisoning happens. This can happen either through ingesting, inhaling, or skin absorption. A rapid heartbeat, chest pain, difficulty breathing, headache, nausea, and confusion are signs of isocyanide intoxication. Isocyanide poisoning can, in extreme circumstances, result in coma or death.
Practice Problems:
Q1. What is the following compound’s IUPAC name?
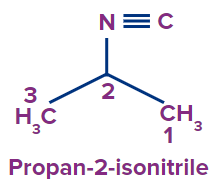
(A) Propyl-2-isocyanide
(B) Butane-2- isonitrile
(C) Isopropyl cyanide
(D) None of the above
Answer: (A)
Solution: The following compound has an isocyanide functional group.
Root Word: The parent chain is of three carbon atoms. So, prop will be the root word.
Primary suffix: -yl
Secondary suffix: isocyanide
Hence the compound’s IUPAC name is Propyl-2-isocyanide.
Q2. Type of isomerism in cyanide and isocyanide is
(A) Functional
(B) Positional
(C) Tautomerism
(D) Chain
Answer: (A)
Solution: As both cyanides and isocyanides have the same chemical formula but different functional groups -CN and -NC , they exhibit functional isomerism.
Q3. The Bhopal gas tragedy in 1984 was brought on by the leakage of
(A) Methyl Isocyanate
(B) Sodium bicarbonate
(C) Potassium iodide
(D) Sodium dioxide
Answer: (A)
Solution: As a pesticide, MIC gas is employed. The Bhopal disaster commonly referred to as the Bhopal gas tragedy, included a MIC gas leak in India. An organic substance with the chemical formula CH3NCO is methyl isocyanate. It is also known as MIC, isocyanatomethane, and methyl carbylamine.
Q4. Why does the carbylamine test fail for secondary (2o) amines and tertiary (3o) amines?
(A) They undergo a reaction with alcoholic potassium hydroxide (KOH)
(B) on reaction with chloroform, they produce a stable complex.
(C) Both A and B
(D) None of the above
Answer: (A)
Solution: Isocyanide, commonly known as carbylamine, is produced when chloroform is heated in the presence of a primary amine and an alcoholic potassium hydroxide (KOH). This reaction is also known as an isocyanide test since it is used to test for primary amine and chloroform. The carbylamine test fails for secondary and tertiary amines because of their reactions with alcoholic potassium hydroxide (KOH).
Frequently Asked Questions(FAQs):
Q1. Are compounds of isocyanides and cyanides the same?
Answer: Cyanides and isocyanides are isomers of one another. Through its carbon atom, the cyanide functional component binds to proteins and other organic substances. On the other hand, the related organic component is joined to the isocyanides component by its nitrogen species.
Q2. What are the purpose and applications of isocyanide?
Answer: When it comes to their reactivity with various kinds of chemicals, isocyanides exhibit a dynamic nature. These isocyanides are suitable reagents for a variety of processes due to their versatility in reactivity. Applications of isocyanides have been seen in a variety of academic fields, such as the material sciences, astronomy, and biological sciences. The synthesis of heterocyclic compounds is a significant application in the study of organic chemistry.
Q3. What's an illustration of isocyanides that happen naturally?
Answer: An antibiotic called xanthocillin is the first isocyanate chemical to exist naturally. From the fungus P. notatum, this was extracted. In the years that followed, a number of naturally occurring chemicals with the functional group isocyanide were found. Among these are primarily marine animal metabolites. The majority of them are terpene isocyanides, and they are frequently seen in combination with other functional groups like isothiocyanate and formamide.
Q4. What are the general characteristics of isocyanide?
Answer: Isocyanides are exceedingly poor electrophiles that can only react with solid nucleophiles like organometallic compounds. The isocyano group in aliphatic isocyanides, like the ortho-methyl bunch in fragrant isocyanides, has an electron-withdrawing function that enhances the acidity of the a-C-H bond group.


