-
Call Now
1800-102-2727
Isochoric Process - Definition, Thermodynamic Changes, P vs V Plot, T vs V Plot and Work Done in Isochoric Processes, Practice Problems and FAQs
Have you been to your kitchen? You must have seen utensils like a pressure cooker, and Tawa (open pan) there. The pressure cooker is used for softening hard items like rice, dhal, millet, etc. Tawa for hot mixing. This is not a cookery class, but to know in what way both of these utensils are utilised under heating.


The whole idea of cooking in a pressure cooker is to break a hard material easily which would otherwise take a long time at high temperatures. The pressure cooker is a closed vessel, where no mass can either enter or leave the system. The volume available for the contents is the same irrespective of the temperature of the vessel. The pressure inside the vessel shall increase proportionately with the heating or temperature for a volatile content inside the container. Cooking here is a constant volume process or also referred to as an isochoric process. Automobile engines and industrial reactors work on this constant volume process.
Tawa on the other hand is for materials that get cooked at atmospheric conditions. Here the pressure is the same at all temperatures, equal to the atmospheric pressure of one atmosphere, while the volume is expandable and hence not a constant. This is a constant pressure process. All open-air processes or natural processes are constant pressure or isobaric processes.
Lets us know something more about this isochoric process.
Table of Content
- Definition of Isochoric Process
- Thermodynamic Changes During an Isochoric Process
- Pressure vs Temperature(P vs T) Plot for Isochoric Process
- Pressure vs Volume( P vs V) Plot for Isochoric Process
- Work Done in Isochoric Processes
- Practice Problems
- Frequently Asked Questions
Definition of Isochoric Process
It is a thermodynamic process in which the volume of the system remains constant throughout a thermodynamic (heat or work) changes occurring on the system, i.e., the change in volume (dV) throughout the process is zero.
A thermodynamic process taking place at constant volume is known as the isochoric process. It is also sometimes called an isometric process or constant-volume process. The term isochoric is a Greek word, where “iso” means “constant” or “equal” and “choric” means “space” or “volume.”
An isochoric process is one for which, the initial and final volumes of a process are equal.
Vf = Vi i.e. V =0
Thermodynamic Changes during an Isochoric Process
Consider a vessel fixed with an immovable piston. Heating the vessel containing a volatile substance creates evaporation of the substance into vapour. As the temperature increases the kinetic energy of the (vapour) gas particles, will result in an increased impact per collision. Thus, the pressure exerted by the gas particles will increase (as indicated by the barometer), trying to push the piston in an upward direction and increase the pressure on it.
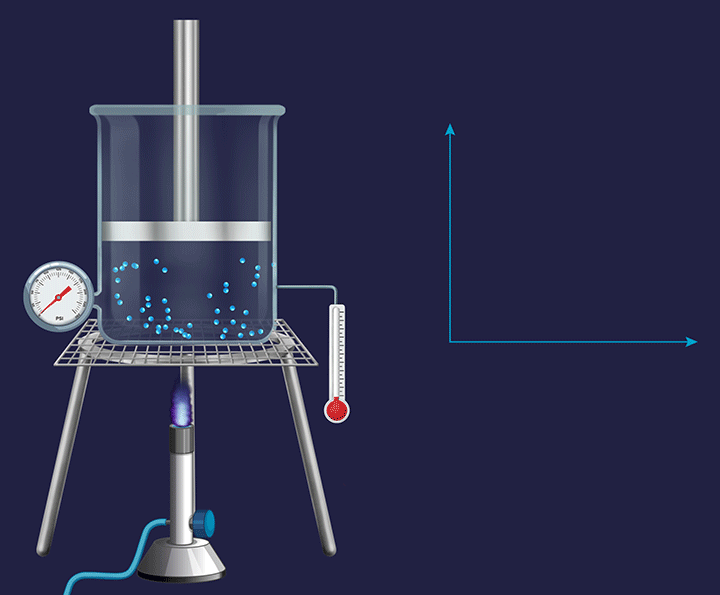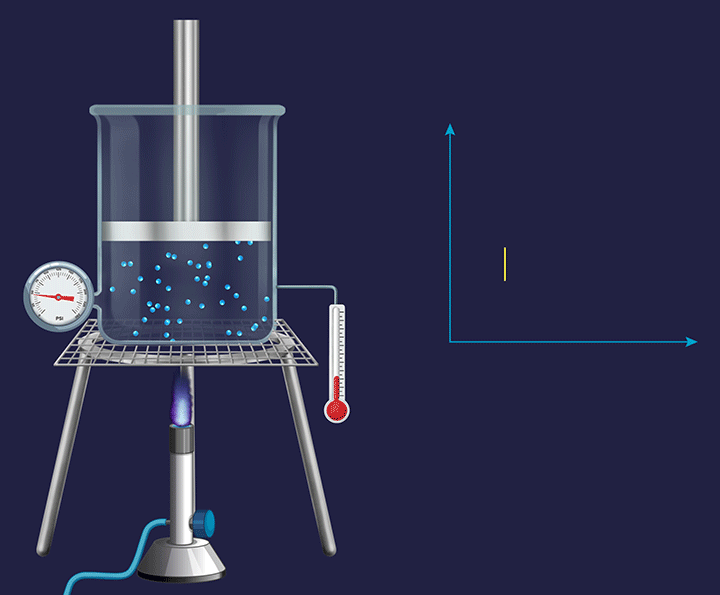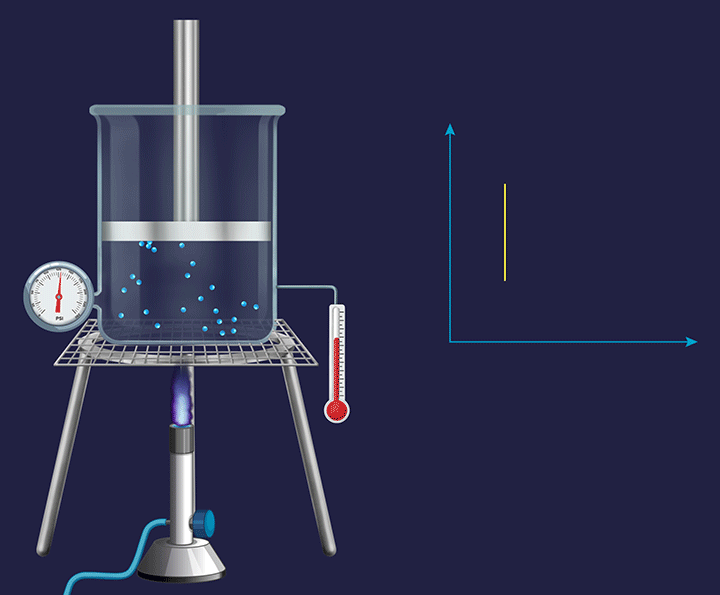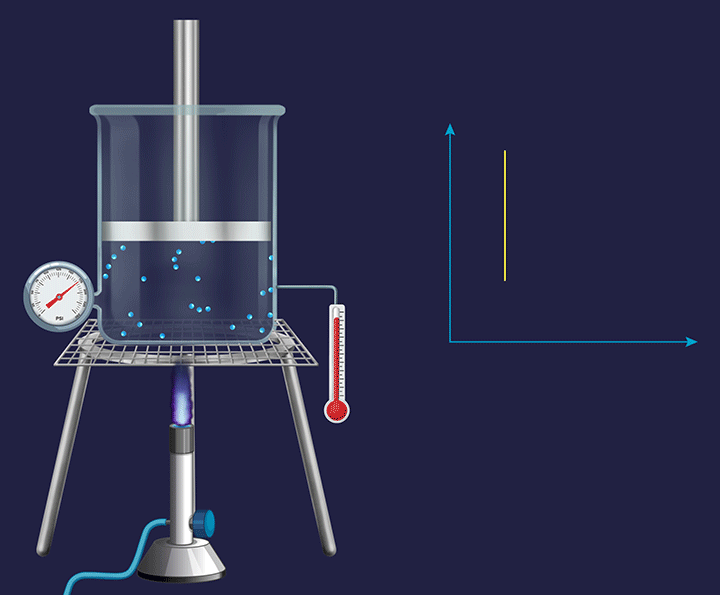

Pressure vs Temperature (P vs T) Plot for Isochoric Process
In a constant volume system the pressure inside varied directly in proportion to the changes in the temperature, which is shown in the P vs T plot given below.
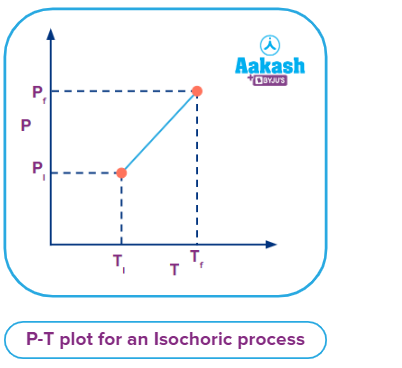
This can be correlated with the equation of a straight line.
y = mx + c
T = kV
If above two equations are compared, the y-intercept (c) would be zero and with a slope (m) = k on a T-V curve.
In the case of expansion of gas at isobaric conditions, as the temperature of the gas increases, the volume occupied by the gas also increases. During compression, the temperature of the gas decreases and will result in a subsequent decrease in volume.
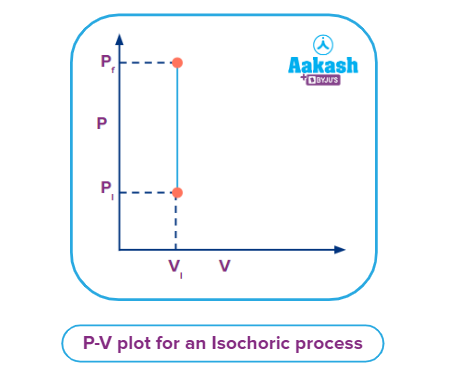
Pressure vs Volume (P vs V) Plot for Isochoric Process
From the illustration, it is observed that the piston remains in its stationary position with the increase in pressure, i.e., the volume is constant throughout the process. The P vs V plot for an isochoric process is a straight line parallel to the pressure axis.
Work Done in Isochoric Processes
In thermodynamics we consider only the work of expansion or compression accompanying changes against a constant external pressure ie PdV.
w = ∫dw = -∫Pext dV ... (1)
since the volume remains constant throughout the process in an isochoric process,
dv = 0. Thus, work done during an isochoric process is zero, i.e., dw = 0
From the first law of thermodynamics, change in internal energy (dU) of a system is given as follows:
dq = dU + dw,
Where, dq is the heat transferred and dw is the work done.
Hence from the first law of thermodynamics
dq = dU (isochoric process)
The total heat supplied or rejected is also equal to the increase or decrease in the internal energy of the system.
Hence, the change in internal energy for an ideal gas sample in an isochoric process is brought about by heat transfer.
Practice Problems
Q1. The isochoric process is one in which
A. Free expansion takes place
B. Very little mechanical work is done by the system
C. No mechanical work is done by the syste
D. All parameters remain constant
Answer: Option C
Solution: Since the volume remains constant throughout the process in an isochoric process,
dV = 0. Thus, work done during an isochoric process is zero.
Q2. Which of the following is constant in an isochoric process ?
a. Volume
b. Pressure
c. Temperature
d. None of these
Answer: Option A
Solution: Isochoric process is the process that occurs at constant volume. In other words, the change in volume is zero.
Q3. For which of the following processes is the relation dQ=dU true?
a. Isothermal
b. Isochoric
c. Adiabatic
d. None
Answer: Option B
Solution: Using the first law we see that the relation dQ=dU implies that the work done is zero. Work done being W=∫PdV is zero for no change in volume.
Thus, the correct answer is an isochoric process.
Q4. What will be the work done when pressure changes from Pi to Pf as shown in graph below?
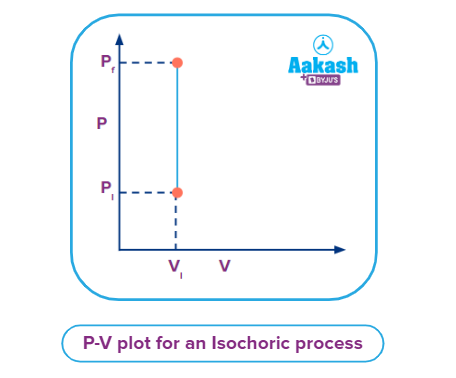
a. Maximum
b. Infinite
c. Zero
d. Negative
Answer: Option C
Solution: Since the volume remains constant throughout the process as shown in the graph above, dV = 0. Thus, the work done during this process will be zero.
Frequently Asked Questions
Q1. Why the relationship between pressure and temperature maintains a constant value in the isochoric process?
Answer: An isochoric process is a thermodynamic process that occurs at a constant volume. Because the volume doesn't change, the relationship between pressure and temperature maintains a constant value.
Q2. Can an isochoric process be reversible?
Answer: Yes, an isochoric process is reversible. For the isochoric process to be reversible every moment system has to be in thermal equilibrium and the supply of heat should mostly be in a quasi-static manner.
Q3. Is heat added during the isochoric process?
Answer: During an isochoric process, heat enters (or leaves) the system and increases (or decreases) the internal energy.
Q4. What happens when a gas is heated at constant volume?
Answer: When the gas is heated at constant volume, the heat supplied increases the internal energy of the gas.


