-
Call Now
1800-102-2727
Interhalogen Compounds - Introduction, Preparation, Properties, Uses, Practice Problems & FAQs
We have all performed laboratory experiments. Generally, we use aqueous solvents. Right?
An aqueous solution is one in which water serves as the solvent. In many of our common experiments, we use aqueous solutions.
Do you know non-aqueous solvents? I guess many of you don’t have much idea about such solvents.
A non-water solvent that is inorganic in nature is one that is not an organic substance. Chemical research and industry employ these solvents for reactions that cannot take place in aqueous solutions or call for a particular environment. We all know halogen compounds very well but I guess many of you were not aware of interhalogen compounds. They are a very good example of a non-aqueous solvent.

Today we will discuss such compounds in detail, more specifically about the preparation and properties, and applications.
Table of Content:
- Introduction
- Preparation of Interhalogen compounds
- Physical properties of Interhalogen compounds
- Chemical properties of Interhalogen compounds
- XX',XX3'and XX5' compound
- Uses of Interhalogen compounds
- Practice Problems
- Frequently asked questions-FAQs
Introduction:
When members of the halogen group interact with one another, interhalogen compounds are created. They can be classified as having general compositions, XX' , XX3' and XX7', where X is a larger-sized halogen and X' is a smaller-sized halogen, and X is more electropositive than X'. These are interhalogen chemicals, which are covalent compounds.
Preparation of interhalogen compounds:
The direct combining of interhalogen compounds or the action of halogen on lower interhalogen compounds can be used to create them. The product created depends on a few particular circumstances.
Examples:

Physical properties of interhalogen compounds:
- These molecules are covalent and generally diamagnetic in behavior.
- Compared to diatomic halogen bonds, the bonds produced between these compounds are more reactive.
- With the exception of ClF , which is a gas, they are all volatile solids or liquids at 298 K.
- These molecules' physical characteristics vary depending on which of their constituents they are.
- The size of the individual halogens determines the bond length.
|
Type |
Formula |
Physical state and colour |
|
XX' |
ClF BrF IF BrCl ICl IBr |
Colourless gas Pale brown gas Detected spectroscopically Gas Ruby red solid (α-form) brown red solid (β-form) Black solid |
|
XX3' |
ClF3 BrF3 IF3 ICl3 |
Colourless gas Yellow green liquid Yellow powder Orange solid |
|
XX5' |
IF5 BrF5 ClF5 |
colourless gas but solid below 77 K colourless liquid colourless liquid |
|
XX7' |
IF7 |
colourless gas |
Chemical Properties of interhalogen compounds:
- They behave chemically similar to the individual halogens.
- Interhalogen compounds typically exhibit higher reactivity than halogens (except fluorine). This is so because all halogens, with the exception of F-F bonds, have X-X' bonds that are weaker than X-X bonds.
- All of them are hydrolyzed to produce halide ions from the smaller halogen and hypohalite (XX'), halite(XX3'), halate (XX5'), and perhalate anion (XX7')from the bigger halogen.
Examples:
- Many metal oxides, metal halides, and metals will fluorinate in the presence of interhalogen chemicals.
Examples:
XX',XX3'and XX5' compounds:
- It is commonly known that iodine monochloride exists. It serves as Wij's reagent in the calculation of a fat or oil's iodine number. The amount of double bonds, or the degree of unsaturation of the fat, is measured by the iodine number. In the fat, the ICl strengthens double bonds. When an unsaturated fat is introduced to the brownish ICl solution, the colour gradually fades away until all the double bonds have broken. The volume (mL) of a standard solution of ICl that reacts with a set weight of fat is what the term "iodine number" refers to.

- ICl frequently iodinates organic substances when it combines with them, while under certain circumstances it may also chlorinate them.
For example:
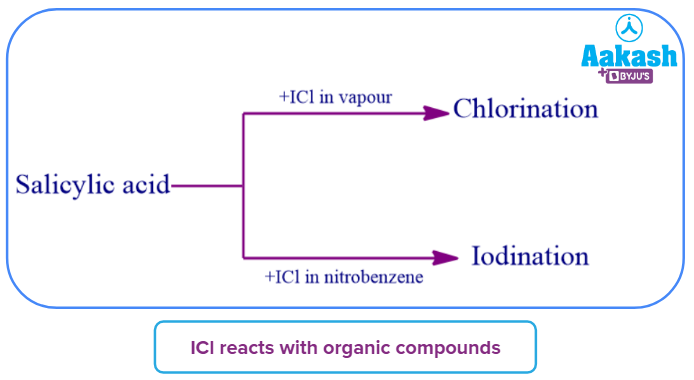
- The order of reactivity of the interhalogens compounds can be written as:
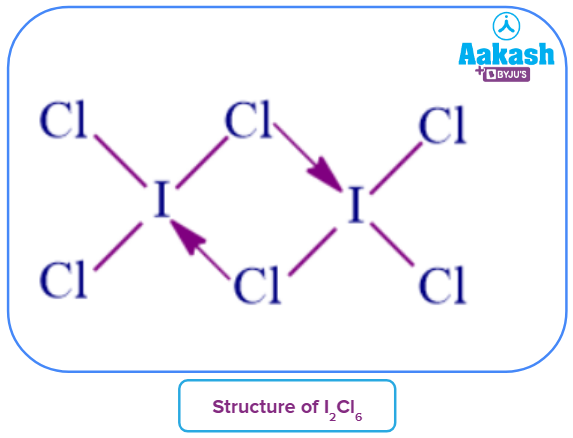
- ICl3 does not exist, but the dimer exists in the form of I2Cl6 which is a bright yellow solid. It has a flat/planar structure. I-Cl bonds at the terminal positions are regular single bonds with lengths of 2.38Å and 2.39 Å. Significantly longer I-Cl bonds (2.68 Å and 2.72 Å ) reveal delocalized bonding as opposed to straightforward halogen bridges made of coordinate bonds from Cl to I. Due to self-ionization, the liquid exhibits an appreciable electrical conductivity.
Self ionisation of
Uses of Interhalogen Compounds:
- These are employed as non-aqueous solvents.
- In a few reactions, they serve as a catalyst.
- Compounds that include interhalogens make excellent fluorinating agents.
- ClF3 and BrF3 are used to create UF6, which is used to enrich U-235.
Practice Problems:
Q1. What is true regarding interhalogen compounds?
A. They are highly stable
B. They have unpaired electrons
C. They are paramagnetic
D. They are diamagnetic
Answer: D
Solution: Interhalogen chemicals are inherently diamagnetic. They have bond pairs and lone pairs, which explains this. Except for the F-F bond, the X-X' link in interhalogen compounds is substantially weaker than the X-X bond in halogens.
Q2. What is the purpose of interhalogen compounds?
A. Aqueous solvents
B. Reducing agents
C. To decrease reactivity
D. Non-aqueous solvents
Answer: D
Solution: When it comes to interhalogen compounds' usage, one of its primary functions is to act as halogenating agents, which means that they are utilized to create additional halogen compounds likeBrF5. Fluorine interhalogen compounds are excellent fluorinating agents.
Q3. What is the type of XX3' (Bent T-shaped) interhalogen compound hybridization?
A. sp3d2
B. sp
C. sp3
D. sp3d
Answer: D
Solution: All interhalogen compounds of type XX3' have trigonal bipyramidal geometries or T-shaped molecules because their structures have sp3d hybridization of the core atom X.
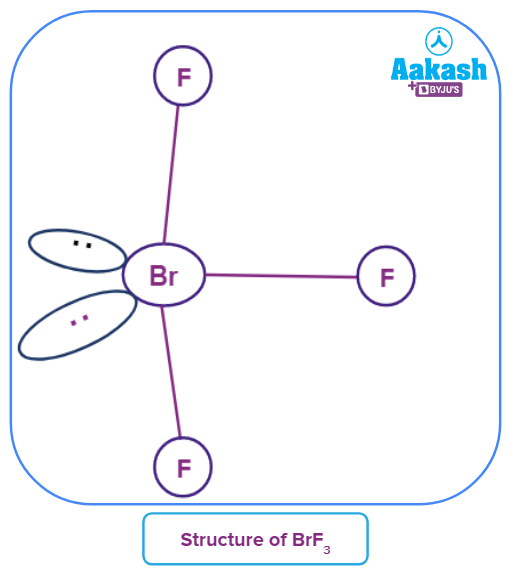
Q4. What is the molecular shape of BrF3 molecules?
A. see-saw
B. linear
C. Bent
D. tetrahedral
Answer: C
Solution: Seven electrons make up the valence shell of the core atom Br. Four electrons will be left behind after three of these form electron-pair bonds with three fluorine atoms. As a result, there are two lone pairs and three bond pairs. These will occupy the corners of a trigonal bipyramid, according to VSEPR theory. To reduce lone pair-lone pair and bond pair-bond pair repulsions that are larger than lone pair-lone pair repulsions, the two lone pairs will occupy the equatorial positions. In order to reduce the lone-pair-lone pair repulsions, the axial fluorine atoms will also be bent in the direction of the equatorial fluorine. The form would resemble a slightly bent "T."
Frequently Asked Questions-FAQs:
Q1. Can you explain pseudohalogens?
Answer: Pseudo-halogens are another name for polyatomic similar halogens. They can replace halogens in numerous kinds of chemical compounds thanks to their similar chemistry to real halogens. Some of the functional groups of the well-known pseudo-halogens contain cyanide, cyanate, thiocyanate, and others.
Q2. What causes interhalogens to be unstable?
Answer: Interhalogens are all easily hydrolyzed, ionize, and produce polyatomic ions. With the exception of F Interhalogens are often more reactive than halogens. This is so because the X-X bonds in dihalogen molecules are stronger than the X-X' bonds in interhalogens.
Q3. What practical use do halogens have?
Answer: Every member of the halogen family is widely utilized in daily life. Your teeth's enamel is strengthened against decay by fluorine in compounds. In acid form, it is also used to etch glass. Our drinking water and swimming pools both include chlorine to prevent bacterial development.
Q4. Are interhalogen compounds explosive in nature?
Answer: Despite being quite unstable, none of them are explosive. Their nature is covalent. They are extremely volatile and have low boiling points. Interhalogen chemicals are not very volatile and are typically liquid or solid at normal temperature.


