-
Call Now
1800-102-2727
Hydroboration Oxidation - Hydroboration-Oxidation Reaction of Alkenes and Alkynes, Their Mechanism, Practice Problems & FAQs
Have you ever experienced cinnamon's lovely aroma? It is simply amazing. Right???
It also has numerous medicinal uses, including in addition to being hydrating, cinnamon has anti-inflammatory, anticancer, and antioxidant characteristics that improve heart health, decrease cholesterol, and may even help with weight loss.
According to certain studies, cinnamon may benefit diabetics also.

Wow! Such a magical spice it is.
Are you curious to understand the makeup of the chemicals found in this powerful spice?
Cinnamaldehyde, an aldehyde that provides flavorings and fragrances, is one of the most significant active components of Cinnamon. Let's talk about the hydroboration oxidation process which is used to make these kinds of important aldehydes.
TABLE OF CONTENT
- What is Hydroboration Oxidation reaction
- Hyboration-Oxidation Reaction of Alkene:
- Mechanism of Hydroboration-Oxidation of Alkene
- Hydroboration Oxidation reaction for Alkynes
- Practice Problems
- Frequently Asked Questions-FAQs
What is Hydroboration Oxidation reaction?
When unsaturated hydrocarbons are transformed into neutral alcohols or ketones or aldehyde using borane in the presence of THF followed by the addition of hydrogen peroxide in the presence of a base, this is known as the hydroboration-oxidation reaction.
This is accomplished via a two-stage procedure that entails both an oxidation and a hydroboration step.
As per the anti-Markovnikov rule, a hydroxyl group is bonded to the carbon atom that is less substituted in the hydroboration oxidation reaction mechanism. The American chemist Herbert Charles Brown, who was born in England, published the article regarding the Hydroboration Oxidation Reaction and received the Nobel Prize in Chemistry.
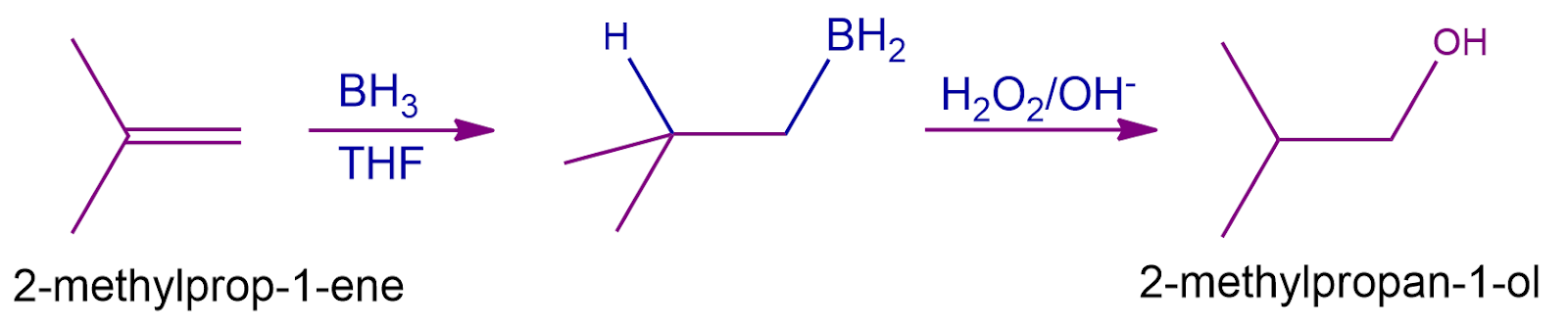
Hyboration-Oxidation Reaction of Alkene:
The hydroboration oxidation of alkene is a two-step process that involves both a hydroboration step and an oxidation step. An anti-Markovnikov rule is used to achieve the addition of products across the entire double bond.
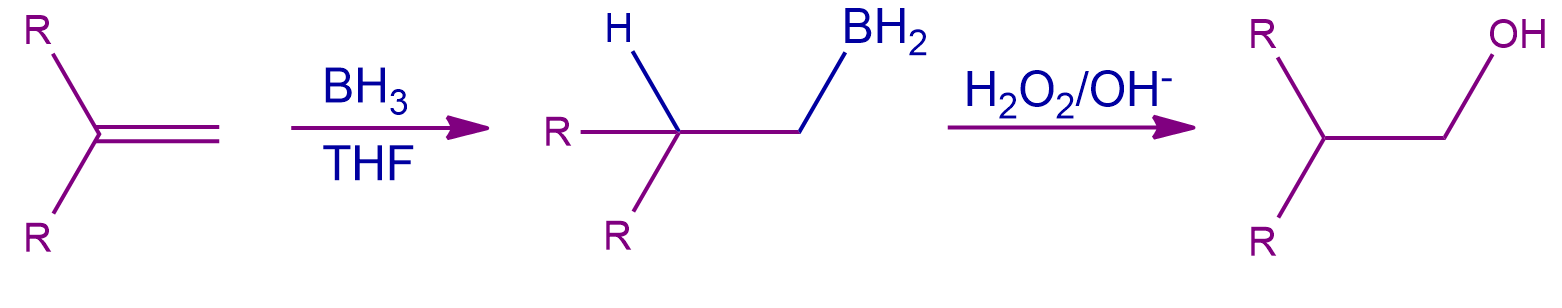
Mechanism of Hydroboration-Oxidation of Alkene:
An anti-Markovnikov reaction in which a hydroxyl group attaches to the less substituted carbon can be used to conceptualize the mechanism of hydroboration oxidation. Here, alkenes undergo conversion to primary alcohols. Two steps, as described below, can be used to summarize the entire reaction.
Step-1: The Hydroboration Process
The initial step is the stereospecific-syn addition of BH3to the given double bond. As a result, an atom of hydrogen is transferred to the carbon atom next to the one that is bound to the boron atom. After two further repetitions of the hydroboration procedure, the initial BH3 is converted into three alkenes attached with a boron atom. Hence, Trialkyl borate is created by repeating this procedure three times.
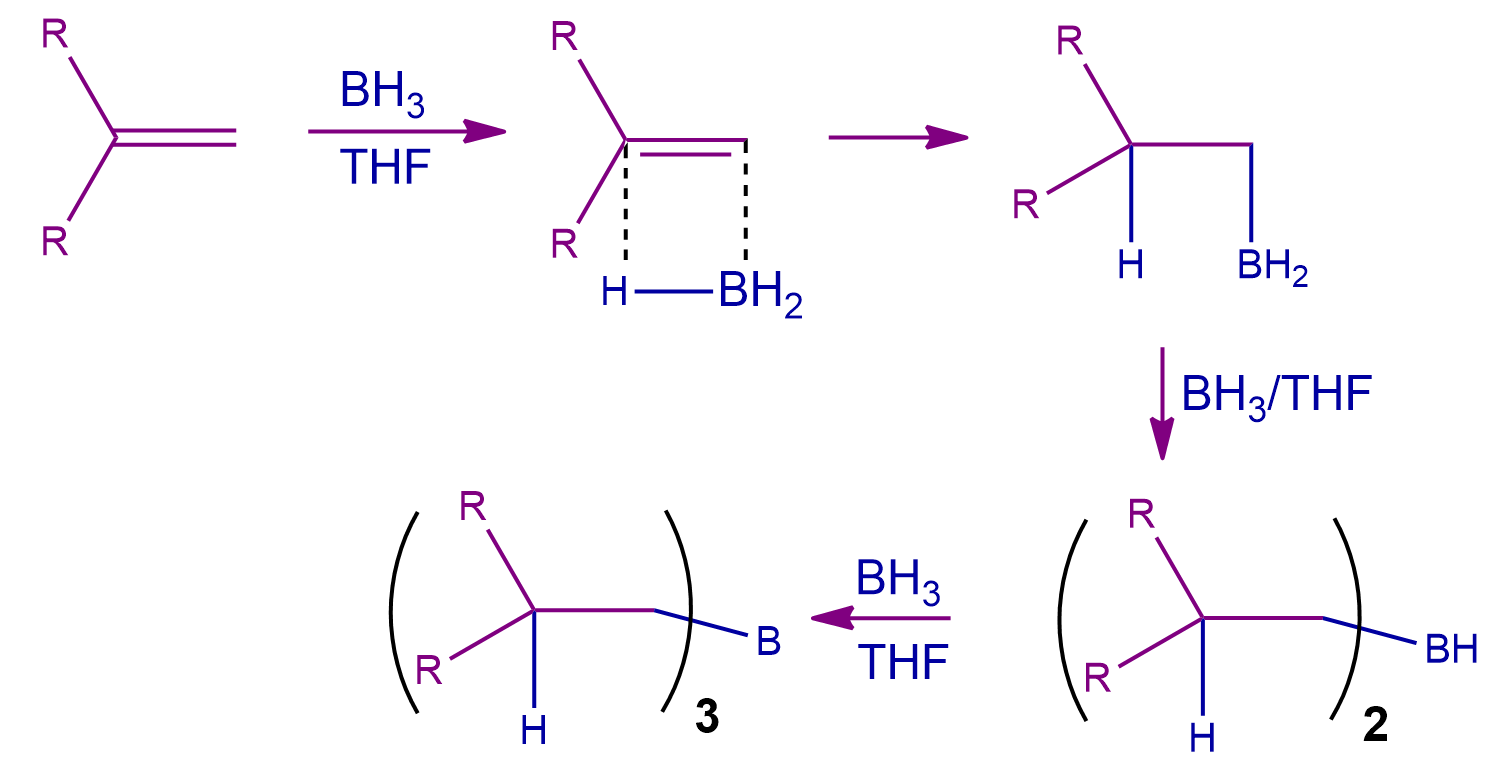
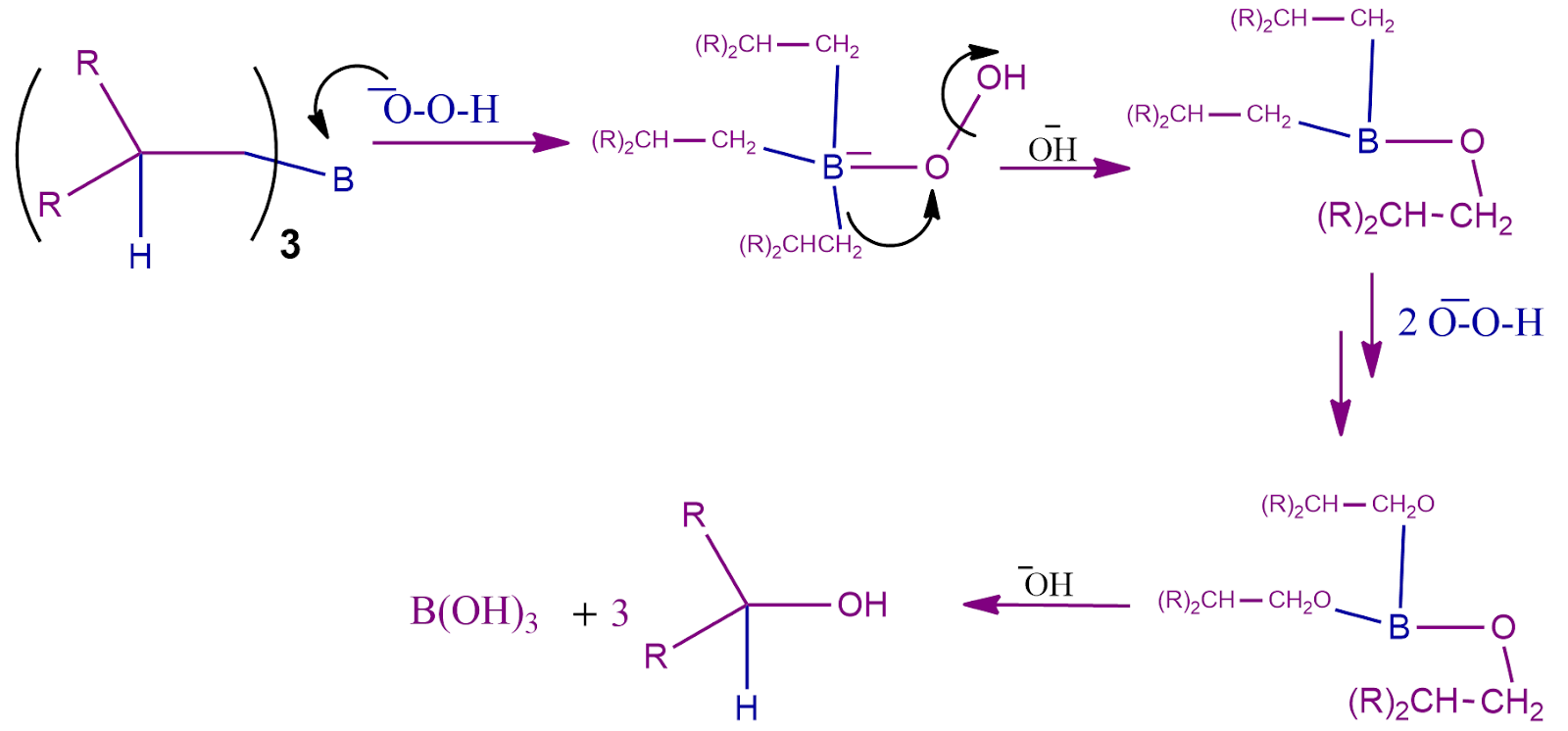
Step-2: The Oxidation Process
The second part of the hydroboration procedure can now start since the trialkyl borane has been created. The hydroperoxide ion, a nucleophilic ion, attacks the boron atom in this reaction.
The necessary primary alcohol is now created by treating this trialkyl borate with perhydroxide ion. The mechanism's steps are shown in the mechanism scheme below.
Hydroboration-Oxidation reaction for Alkynes:
The hydroboration oxidation reaction is a type of organic chemical reaction that converts alkynes to ketones or aldehydes. The hydroboration oxidation of prop-1-yne is a two-step process that involves both a hydroboration step and an oxidation step. An anti-Markovnikov rule is used to achieve the products across the entire triple bond.
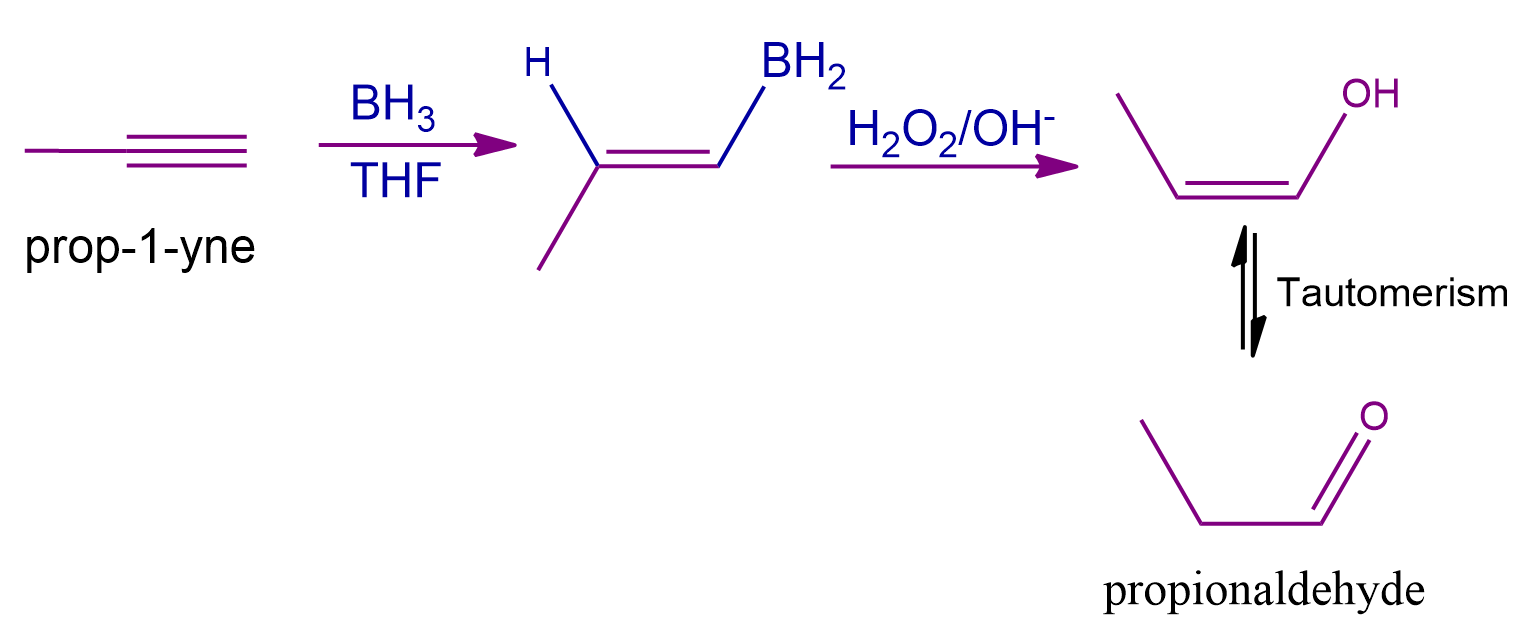
Mechanism:
In an anti-Markovnikov reaction, a hydroxyl group bonds to the less substituted carbon, and this is how hydroboration oxidation works. The complete reaction can be divided into the following two steps.
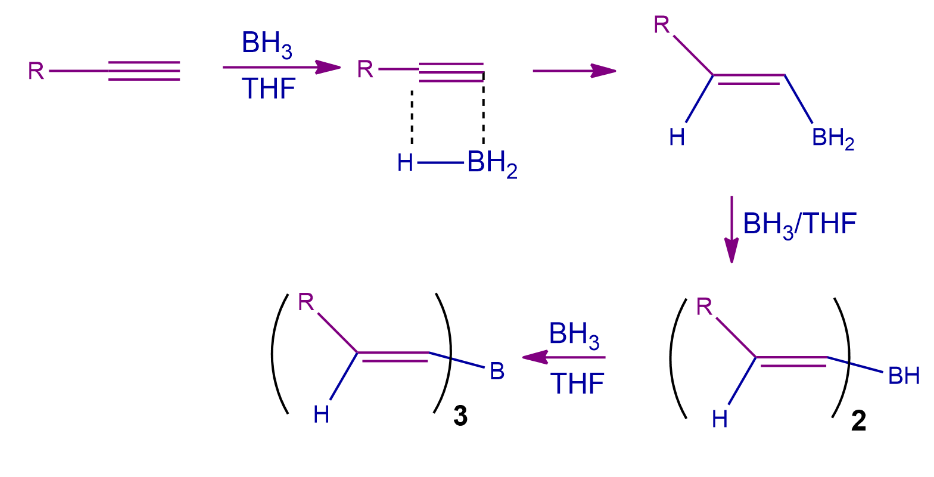
Step-1: Hydroboration Process
The alkynes can undergo hydroboration in an anti-Markovnikov manner. The boron atom prioritizes attacking the least substituted carbon since it is also the least hindered. Substituted boron, being a bulky reagent, is helpful to stop the reaction in the alkenyl group. If borane is utilized alone, both of the alkyne's bonds will be hydroborated. Trialkyl borate is produced as a result of three repetitions of this process.
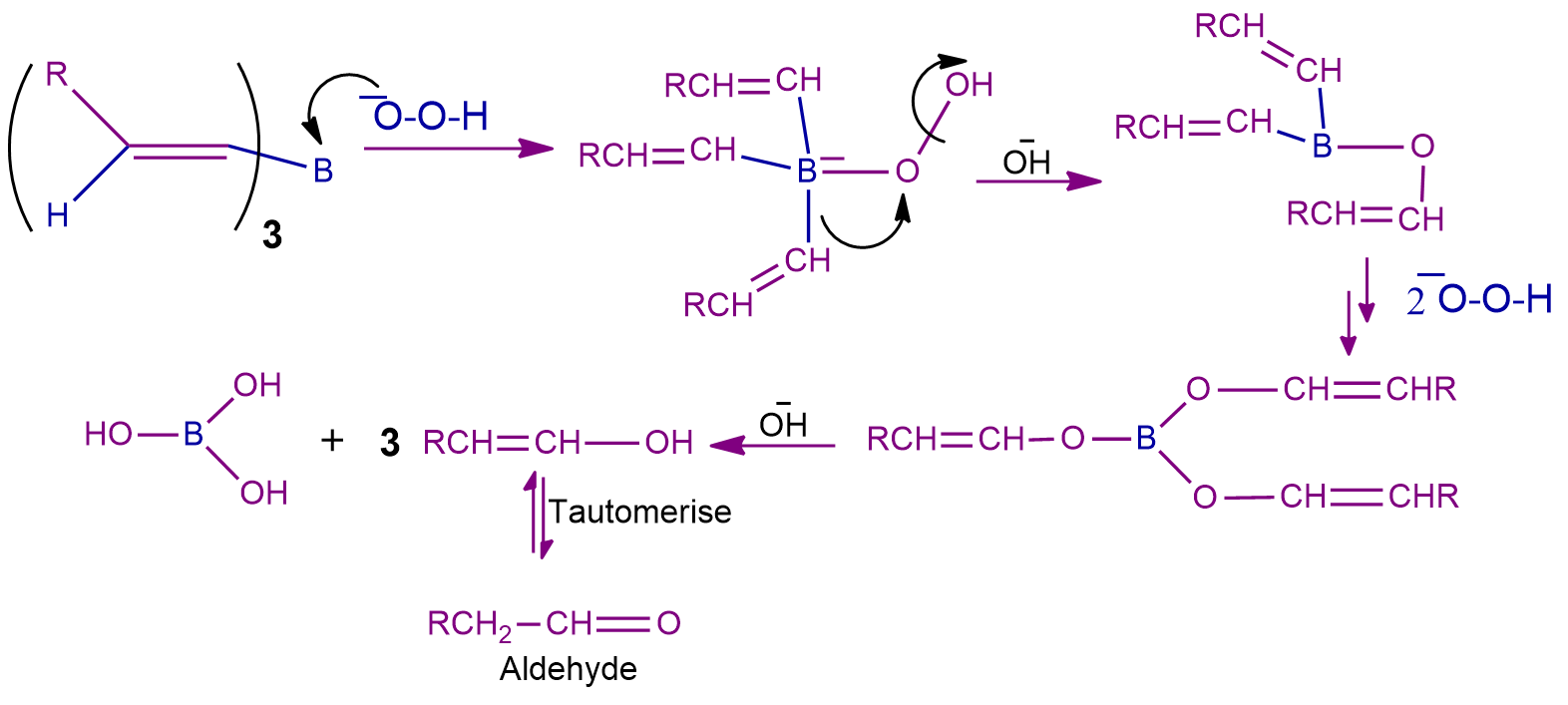
Step-2: The Oxidation Process
Now that the trialkyl borane has been created, the second stage of the hydroboration process can start. In this stage, the nucleophilic hydroperoxide ion attacks the boron atom. The alcohol is now created by treating this trialkyl borate with water which is tautomerism to give the final product aldehyde or ketone. The next image shows the mechanism's steps.
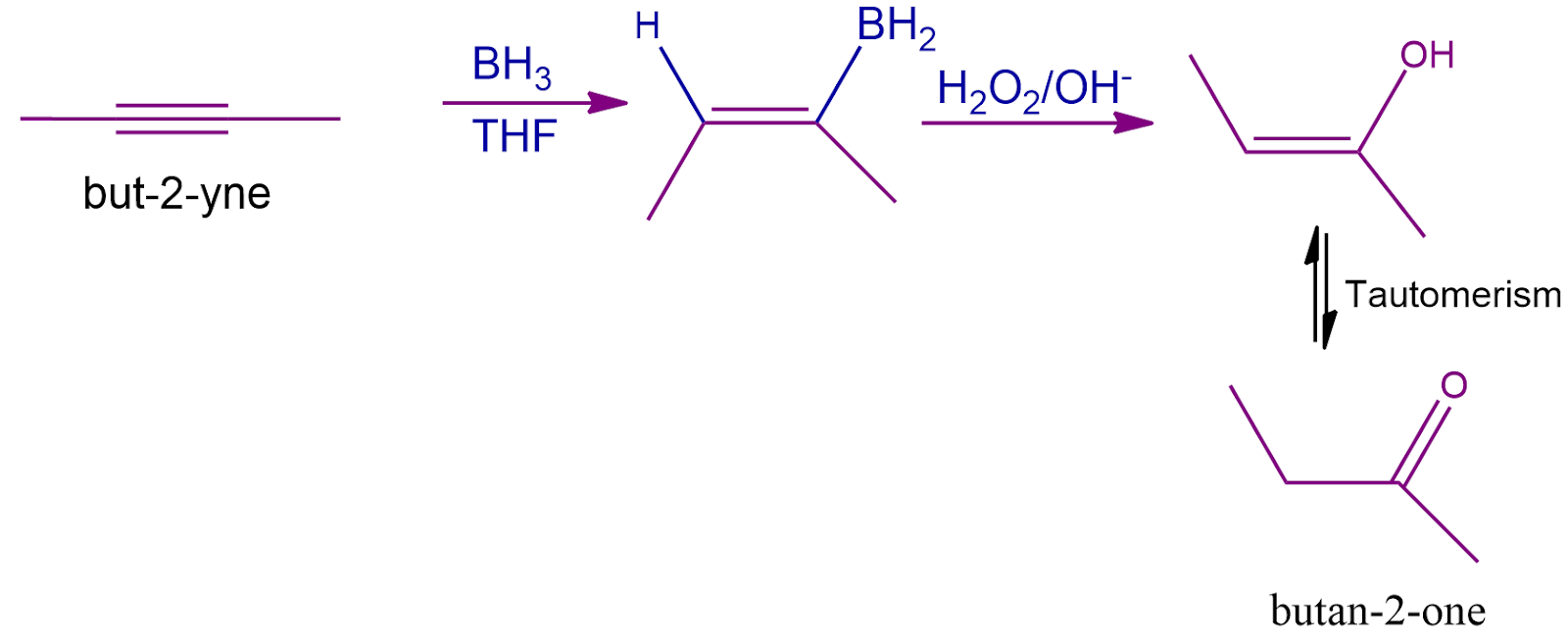
Non-Terminal Alkyne (But-2-yne):
Related Video Link: Oxidation of Alkenes | CHEMISTRY | JEE | Concept of the Day | Nitika Ma'am
Practice Problems:
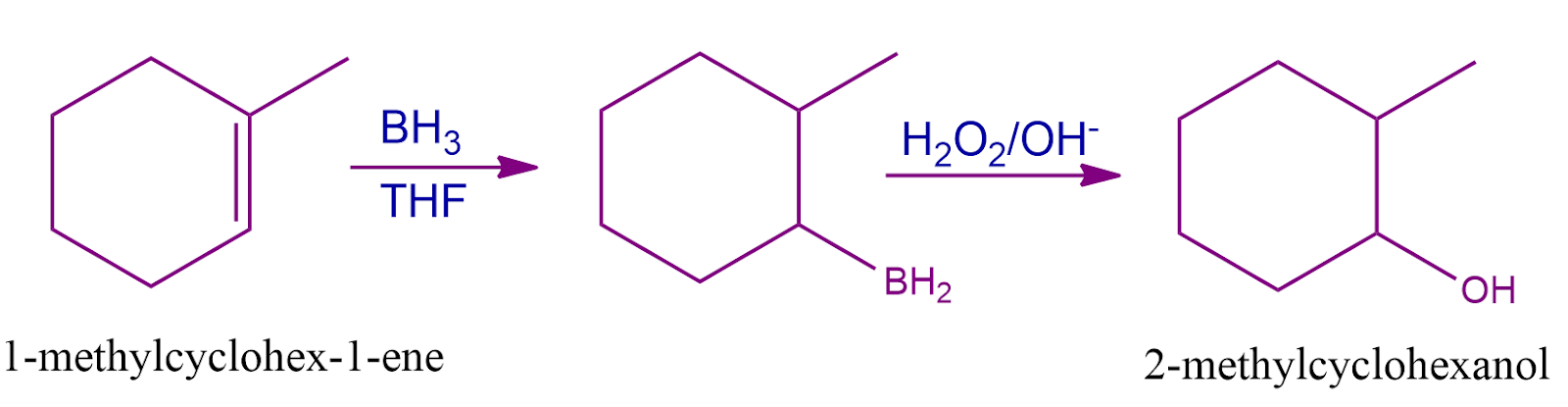
Q1. Predict the final product when 1-methylcyclohexene undergoes a hydroboration-oxidation reaction.
- 2-Methylcyclohexan-1-one
- 2-Methylcyclohexanol
- 2-Methylcyclohexan-2-one
- 1-Methylcyclohexanol
Answer: (B)
Solution: Alkenes convert into alcohol by the hydroboration oxidation reaction which is a two-stage process that includes a hydroboration step and an oxidation step using an anti-Markovnikov Rule. The Anti Markovnikov rule states that the predicted result of the reaction of 1- methylcyclohexene with BH3 followed by H2O2/OH- is 2-methylcyclohexanol. Therefore, the correct answer is option (B).
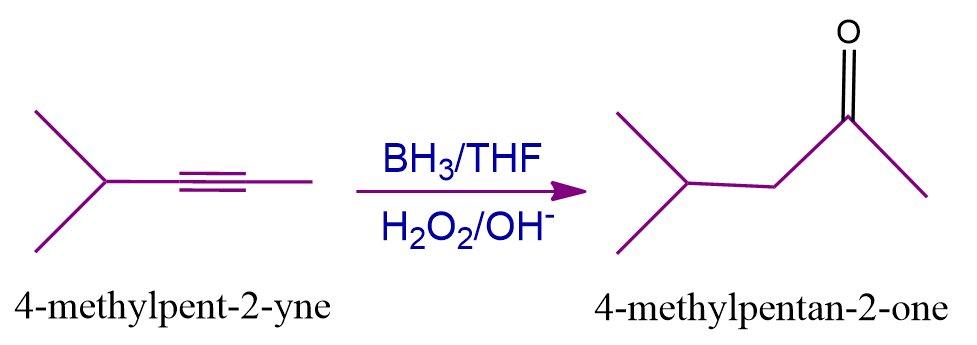
Q2. What should be the nature of final product formed in the hydroboration oxidation reaction when 4-methylpent-2-yne is treated with BH3 followed by H2O2/OH-.
- Aldehyde
- Ketone
- Alcohol
- Carboxylic Acid
Answer: (B)
Solution: Alkynes convert into aldehyde or ketone by the hydroboration oxidation reaction which is a two-stage process that includes a hydroboration step and an oxidation step.
With help of The Anti Markovnikov rule, when 4-methylpent-2-yne is reacted with BH3 followed by H2O2/OH-, the expected product is 4-methylpentan-2-one which is a ketone. Hence, the correct answer is option (B).
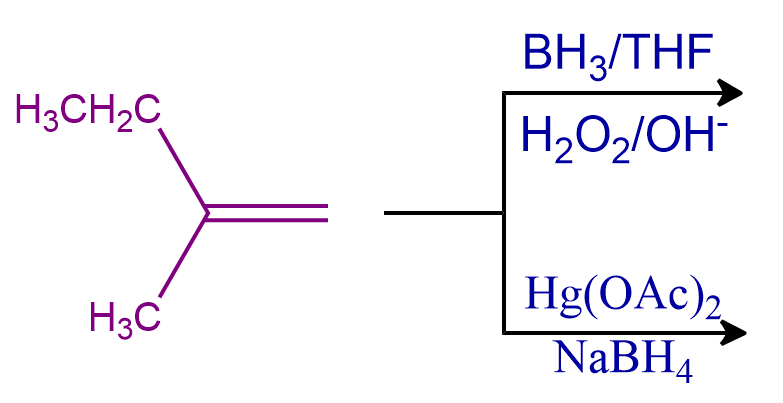
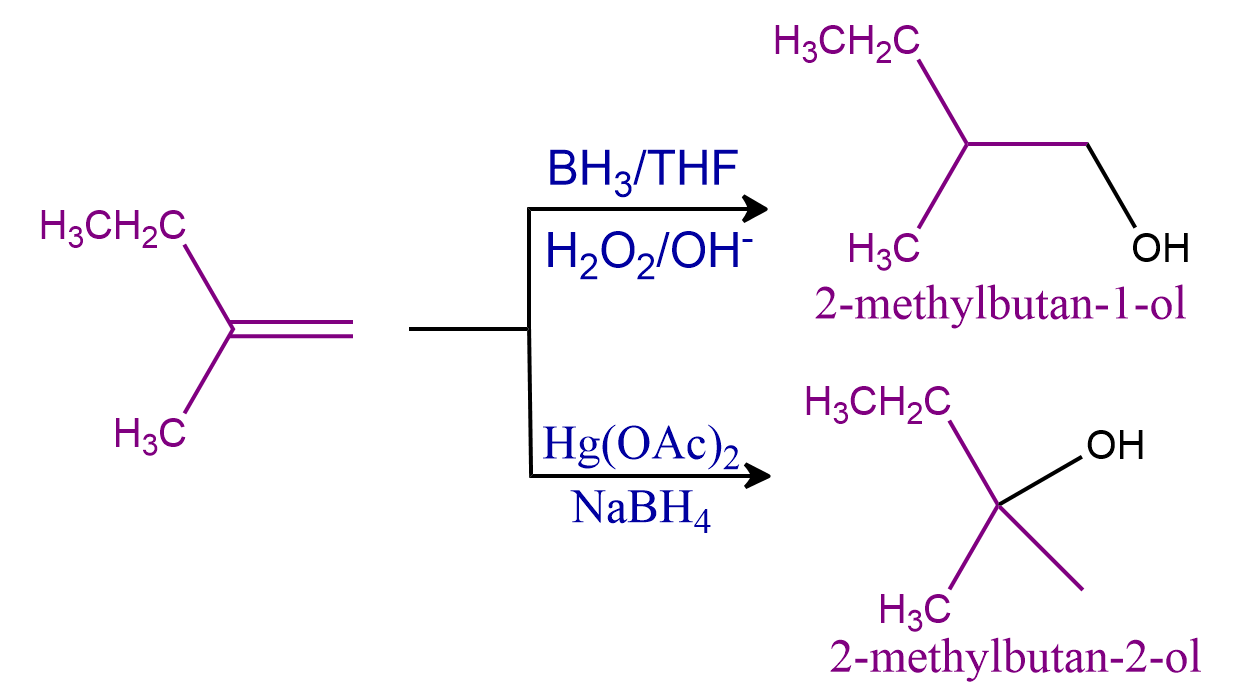
Q3. There is the formation of alcohols using hydroboration oxidation and oxymercuration-demercuration methods. Predict the relation between these alcohols.
- Enantiomers
- Diastereomers
- Mesomers
- Structural Isomers
Answer: (D)
Solution: Using an anti-Markovnikov Rule, alkenes convert into alcohol by the hydroboration oxidation reaction which is a two-stage process that includes a hydroboration step and an oxidation step and followed Antimarknovikoff’s rule. Oxymercuration-demercuration method is a chemical reaction which is a two-stage process that includes an Oxymercuration step and a demercuration step and followed Marknovikoff’s rule.
Enantiomers, Diastereomers as mesomers formed when the compounds are stereoisomers which have the same molecular and structural formula but differences in the orientation of atoms or groups. Structural isomers are those which have the same molecular formula but differences in the structural formulas.
In the given reaction, the two products formed are 2-methylbutan-1-ol and 2-methylbutan-2-ol which have the same molecular formula. The structural formula is different because the position of the hydroxyl group is different in both the formed products. Hence, they are structural isomers.
Hence, the correct answer is option (D).
Q4. What should be the nature of the expected product formed when 2-methylbut-2-ene undergoes hydroboration oxidation reaction
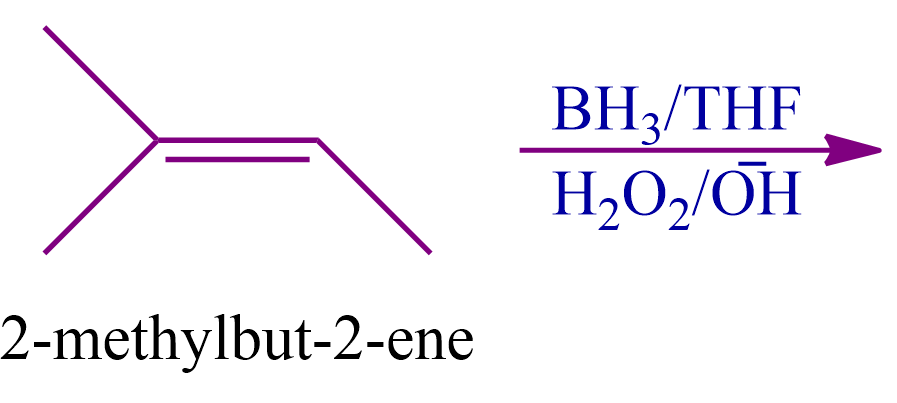
- Optically Active primary alcohol
- Optically Active secondary alcohol
- Optically Inactive primary alcohol
- Optically Inactive secondary alcohol
Answer: (B)
Solution: Alkenes are transformed into alcohol through an organic chemical reaction called the hydroboration oxidation, which makes use of the anti-Markovnikov Rule. This is created via a two-stage process that comprises the steps of hydroboration and oxidation.
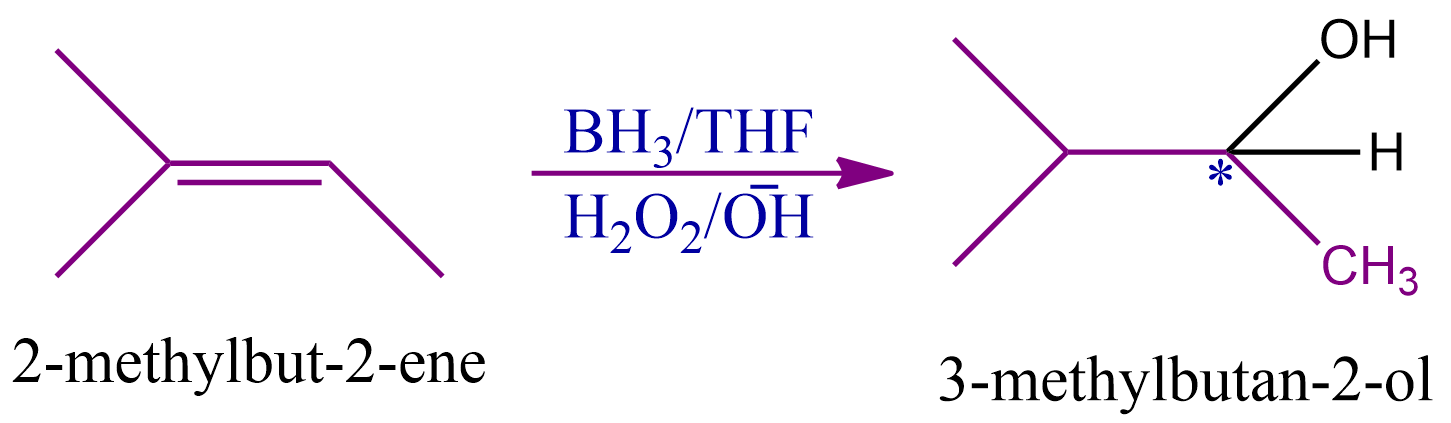
The alcohol formed by this process is secondary alcohol since carbon has one hydrogen, one hydroxyl group, and two distinct alkyl groups. This compound is optically active because it has four distinct groups attached to the carbon, or it is chiral.
Therefore, the correct answer is option (B).
Frequently Asked Questions-FAQs:
1. What functions do aldehydes and ketones serve?
Answer: Currently, aldehydes are used to make resins and polymers. Propanone, the most basic ketone, is frequently referred to as acetone. Acetone is a typical organic solvent that was once a component of most nail polish removers, but alternative solvents have mostly taken its place.
2. What functions does alcohol serve in modern society?
Answer: In order to dissolve numerous organic molecules that are insoluble in water, ethanol is utilized as a solvent. It is a component of paints, cosmetics, detergents, inks, and scents. Due to its ability to inhibit or stop the growth of microorganisms, ethanol possesses antibacterial characteristics.
3. What precisely do you mean by "hydroboration"?
Answer: The process of adding a hydrogen boron link to a carbon-carbon or carbon-nitrogen double bond is known as hydroboration. On a triple carbon bond, it is also feasible to accomplish this. Many organic compounds can be synthesized by hydroboration.
4. What are some uses for alkenes?
Answer: Alkenes have a variety of uses in manufacturing. They serve as the raw ingredients for the synthesis of alcohols, polymers, lacquers, detergents, and fuels. Ethene, propene, and 1,3-butadiene are the three most significant alkenes for the chemical industry.


