-
Call Now
1800-102-2727
Galvanic Cell Working - Principle, Construction, Working Mechanism, Zn-Cu Galvanic Cell & Representation of Galvanic Cell
Have you ever witnessed a live football match in a stadium or on television?
Well, even if you don't know the basic rules and regulations of football. Two teams, 11 players on each team, run after a ball to score goals.

All the players in the team have just one target which is to push the ball forward and score a goal. Before the actual game starts there are a lot of other activities that happen such as tossing, players taking their respective positions and when the referee instructs them to start then only all the players start moving with the ball.
In a galvanic cell, the conversion of chemical energy into electrical energy occurs. Just like the football match, in a galvanic cell, all the components align themselves to carry out the necessary reactions which can lead to the flow of current through the cell.
Table of content
- Galvanic Cell
- Principle of a Galvanic Cell
- Construction of a Galvanic Cell
- Working Mechanism of a Galvanic Cell
- Zn-Cu Galvanic cell
- Representation of Galvanic cell
- Practice Problems
- Frequently Asked Questions - FAQs
Galvanic Cell
A galvanic cell or voltaic cell is an electrochemical device that transforms the chemical energy of spontaneous redox reactions into electrical energy.


Electrons are transferred from one species to another during oxidation-reduction reactions. If the reaction occurs spontaneously, energy is released. As a result, the energy that has been released is put to good use which is converted into electrical energy. To deal with this energy, the reaction must be divided into the two half-reactions of oxidation and reduction. The reactions that are carried out in the half cell drive the electrons from one end to the other end using two different containers and wire. This is how a galvanic cell is constructed.
The cathode is the electrode where the reduction process takes place, and the anode is the electrode where the oxidation process happens. Anode polarity in a galvanic cell is negative, while cathode polarity is positive.
Principle of a Galvanic Cell
The working principle behind electrochemical cells is that when a chemical oxidation-reduction reaction (redox reaction) takes place, electrons moves from one chemical species to another. The electrons in a galvanic cell are often permitted to flow in a circuit outside of the galvanic cell in order to power an external electrical device. So, basically in a galvanic cell, the chemical energy is converted into electrical energy.
Construction of a Galvanic cell
The important components required for a galvanic cell are given below:
- Anode
- Cathode
- Salt bridge
- Half cell
- External circuit
- Load
Anode
An anode in electrochemistry is, in its simplest form, the site of an oxidation reaction. Due to the anode's electrical potential, negative ions or anions usually react there and release electrons. After that, these electrons ascend and enter the driving circuit.
In a galvanic cell, the anode is negatively charged, and the majority of the electrons flow toward the circuit's exterior.
Cathode
In electrochemistry, the cathode is referred to as the electrode where reduction takes place. In an electrochemical cell, this is a common component. Due to the cathode’s electrical potential, positive ions or cations usually react there and accept electrons which are generally released from anode.
In a galvanic cell, the cathode is positively charged.
Salt bridge
Salt bridge contains concentrated solution of an inert electrolyte, such as KCI, KNO3, K2SO4, etc., or a solidified version of these electrolytes in agar-agar and gelatine. Salt bridge is an inverted U-shaped tube.

If the electrodes are not internally connected, the cell initially produces electrical energy, but over time the electrodes become polarised, which causes an accumulation of ions with opposing charges and the electron flow ceases or the cell stops functioning. The two electrolytes are also connected in order to complete the electrical circuit through the salt bridge.
As cations (K+) and anions (Cl-) migrate from the salt bridge electrolyte (let's say KCl) towards the cathode and anode, respectively, they polarise some of the oppositely charged ions in the solution towards themselves. The cell thus continues to function and does not stop.
There will eventually be no electricity flow if there is no salt bridge because the anode half-cell (left beaker) will have an excess positive charge and the cathode half-cell will have an excess negative charge (right beaker).
Electroneutrality is preserved after the addition of a salt bridge because the salt bridge supplies the counter ions and allows the flow of charge (the bulb will light up again).
Important factors while choosing a salt bridge:
- Chosen salt bridge should be made up of mobile cations and anions.
- The ions coming from the salt bridge should not interact with the solution's ions.
- The ions coming from the salt bridge must not undergo oxidation or reduction at the electrodes.
Half cell
A device that uses a single electrode submerged in an electrolytic solution to create a clear potential difference. In the case of a galvanic cell, each of the electrodes of the galvanic cell is called a half cell.
External Circuit
In a galvanic cell, an external circuit facilitates the movement of electrons between electrodes.
Load
An electrical load is a part of a circuit's electrical equipment that uses (active) electric power,
Working Mechanism of Galvanic Cell
When an electrode in a galvanic cell is exposed to the electrolyte at the electrode-electrolyte interface, the metal electrode's atoms tend to produce ions in the electrolyte solution while leaving the electrode's electrons behind. The metal electrode becomes negatively charged as a result. Metal ions in the electrolyte solution, however, also tend to deposit on a metal electrode. Consequently, the electrode becomes positively charged.
Under equilibrium conditions, charge separation is seen, and the electrode can be either positively or negatively charged depending on the tendencies of two opposing reactions. As a result, there is a potential difference between the electrode and electrolyte. The term "electrode potential" refers to this potential difference.
Out of two electrodes, the anode is the electrode where oxidation occurs, and the cathode is the electrode where reduction occurs. The cathode has a positive potential with respect to the solution, whereas the anode has a negative potential.
As a result, a potential difference forms between the galvanic cell's two electrodes. The cell potential is the name for this potential difference. Cell potential, or the electromotive force of the galvanic cell, is what remains when no current is drawn from the galvanic cell. Due to the potential difference, electrons move from the negative electrode to the positive electrode when the switch is turned on.
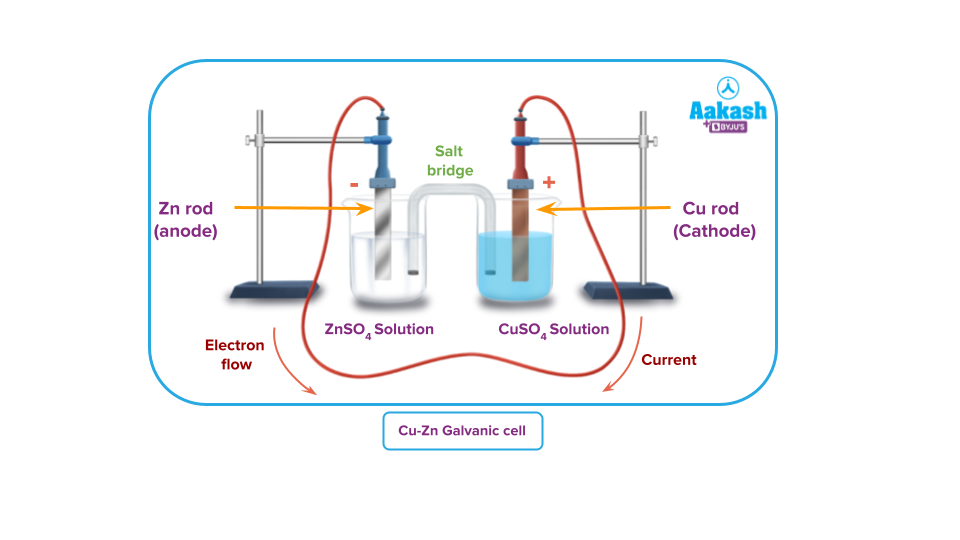
Zn-Cu Galvanic Cell
Let’s understand how the potential difference is being developed in a galvanic cell due to the redox reaction by taking an example of Zn-Cu galvanic cell.
Let’s take two beakers containing Zinc and Copper rods dipped inside their respective salt solutions. The two rods are connected with an electrical wire. The solutions in the beakers are connected by what is called a salt bridge wetted with potassium nitrate solution to complete the electrical circuit.
The redox reaction can be represented as:
Above redox reaction may be split into two half-reactions as shown below:
(oxidation half-reaction)
(reduction half-reaction)
The first reaction is the oxidation half-reaction and the second reaction is reduction half-reaction. The overall reaction is the result of combining the two half-reactions. As a result, if a redox reaction is allowed to proceed so that the oxidation half-reaction occurs in one beaker and the reduction half-reaction in another, the electrons released by the former will be taken by the latter, and a current will flow. This means that the cell is composed of two half-reactions or two half-cells.
Representation of Galvanic Cell
As shown below for the Daniell cell (Zn-Cu Galvanic Cell), a galvanic cell is represented in this way.
- According to the convention, the electrode where oxidation occurs (the oxidation half cell) is written on the left, and the electrode where reduction occurs is written on the right.
- The symbols for the metal (or gas) are written first in the oxidation half cell on the left side, followed by the symbols for the ions in equilibrium with their concentrations in brackets. The concentration is written in brackets after the symbol of the cathode metal in the reduction half cell, which is written in the reverse order of the ion in equilibrium (or the gas).
- The metal electrode and the ion in equilibrium for each half of the cell are separated by solitary vertical lines.
- The presence of a connecting salt bridge linking the two electrolytes is inferred by the double vertical line dividing the two half cells.
Metal | Metal ion (conc.) || Metal Ion (conc.) | Metal
Anode (Oxidation) Salt bridge Cathode (Reduction)
Recommended video: Galvanic cell and its working
Practice Problems
Q.1 Which of the following is not a component of a galvanic cell?
- Salt bridge
- Anode
- Cathode
- Bulb
Answer: (D)
Solution: The important components of a galvanic cell are salt bridge, anode, cathode, half cell and external circuit. The bulb is not a component of a galvanic cell. Most of the time you will see there’s a bulb in the image of the galvanic cell but it is just to check whether the current is flowing or not.
Q.2 What will be the cell representation of Cu-Ag galvanic cell?
Answer: (C)
Solution: The representation of a galvanic cell has to be done in the following manner:
Metal | Metal ion (conc.) || Metal Ion (conc.) | Metal
Anode (Oxidation) Salt bridge Cathode (Reduction)
Hence, in case of Cu-Ag Galvanic cell, the representation will be:
Q.3 Find out the wrong statement with respect to a salt bridge?
- The chosen salt bridge should be made up of mobile cations and anions.
- The chosen salt bridge should be made up of static charges.
- The ions furnished by the salt bridge must not undergo oxidation or reduction at the electrodes.
- The ions furnished by the salt bridge should not interact with the solution's ions.
Answer: (B)
Solution:
Salt bridge is an important component of a galvanic cell and while choosing a salt bridge certain things need to be followed such as:
- Chosen salt bridge should be made up of mobile cations and anions.
- The ions coming from the salt bridge should not interact with the solution's ions.
- The ions coming from the salt bridge must not undergo oxidation or reduction at the electrodes.
If ions are not mobile then they can't function which is required from the ions furnished by the salt bridge. Hence, option (B) is the incorrect statement.
Q.4 During the construction of a galvanic cell, a metallic wire is connected to both the electrodes which are further connected to a _______
- Ammeter
- Galvanometer
- Both A & B
- None of the above
Answer: (C)
Solution: During the construction of a galvanic cell, metallic wires are used to connect two half-cells externally to a galvanometer or ammeter and a switch.
Frequently Asked Questions - FAQs
Q.1 What types of batteries are made possible by galvanic cells?
Answer: A battery is constructed from a number of galvanic cells that are connected in series to form a single voltage source. For instance, a typical 12 V lead-acid battery has six galvanic cells connected in series, each of which has lead anodes and lead dioxide cathodes that are immersed in sulfuric acid.
Q.2 Why is the galvanic cell's anode negative?
Answer: Anode is the electrode where oxidation happens, the metal electrode loses its cations which makes the electrode effectively negative. These electrons travel towards the cathode and the current flows in the opposite direction.
Q.3 What type of energy source does a galvanic cell use?
Answer: Galvanic cells convert chemical energy into electrical energy. This chemical energy is derived from the spontaneous redox reaction taking place in the galvanic cell. Hence, we can say that the source of energy for a galvanic cell is the energy produced during a spontaneous redox reaction.
Q.4 Can a galvanic cell run indefinitely?
Answer: No, the galvanic cell can’t run forever to produce electrical energy from chemical energy. Galvanic cell is made up of an anode and a cathode. Typically, the anode can naturally oxidise and be consumed even when not used to produce electricity. As a result, the anode will eventually run out and the circuit won't be complete. Hence, we can’t run a galvanic cell forever.


