-
Call Now
1800-102-2727
Fractional Oxidation States- Fractional Oxidation Numbers, Some Examples, Practice Problems and FAQs
Let us say that your Principal has assigned you one task.
You must compute the average number of students in four different classrooms. Class A has 25 students, Class B has 18 students, Class C has 15 students, and Class D has 20 students.
Average Students per Class =

The number of students cannot, of course, be fractional, but this is the average number of students in each class.
The same is true for fractional oxidation numbers. The number of electrons can never be expressed as a fraction, but fraction oxidation stands for the average oxidation of two or more elements present in a compound.
TABLE OF CONTENT
- Fractional oxidation numbers
- Some Examples
- Practice Problems
- Frequently Asked Questions-FAQs
Fractional oxidation numbers:
For some compounds, the general rules give a fractional oxidation number to some atoms. For example, the oxidation number of Mn in Mn3O4 , comes out to be + 8/3 This is strange because electrons are never shared or transferred in fractions. Right?
Actually, this fractional oxidation number is the average oxidation number of the element in the compound.
Some Examples:
- Mn3O4 (Manganese Oxide):
For example, Mn3O4 is composed of a mixture of MnO and MnO2, with the formula 2MnO.MnO2.
As a result, the oxidation number of Mn in MnO = x
Oxidation number of Mn in MnO2 = y
Fractional or Average Oxidation number of Mn in Mn3O4 (2MnO.MnO2) = [2(+2)+1(+4)]3=+83
- Fe3O4 (Ferric Oxide):
For example, Fe3O4 is composed of a mixture of FeO and Fe2O3, with the formula FeO.Fe2O3.
As a result, the oxidation number of Fe in FeO = x
Oxidation number of Fe in Fe2O3 = y
Fractional or Average Oxidation number of Fe in Fe3O4 () =
Thus, in a compound, the average oxidation number represents the average of atoms in various bonding situations.
To calculate oxidation numbers in some compounds, knowledge of chemical bonds or structures is required.
Let us illustrate this concept more with an example!
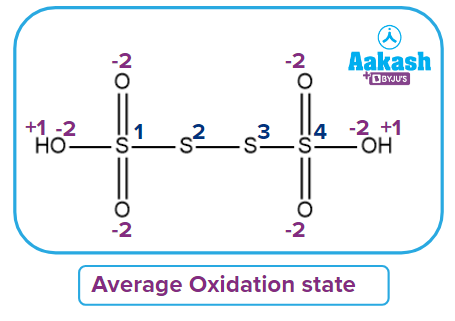
- H2S4O6 (Tetrathionic acid):
Four sulfur atoms are present in the compound's given structure of H2S4O6. As a result, the fractional oxidation of sulfur can be calculated as:
Oxidation number of the First sulfur
Oxidation number of the second sulfur
Because the second sulfur is surrounded by the first and third sulfur, and there is a bond with the same atom, their contribution is assumed to be zero.
Oxidation number of third sulfur
Because the third sulfur is surrounded by the second and fourth sulfur, and there is a bond with the same atom, their contribution is zero.
Fourth sulfur oxidation number
Fractional or Average Oxidation number of S =
- Ferriferrocyanide, :
Oxidation number of Fe outside the coordination sphere = +3
Charge present on one coordination sphere = - 4
The total charge on 4 Fe present outside the coordination sphere become +12 and total charge present on coordination spheres are -12. Hence, they make a neutral compound.
Oxidation number of Fe inside the coordination sphere = y
The iron oxidation number in this molecule is the average of all these atoms,
Fractional or Average Oxidation number of Fe =
Related Topic Link:
Paradox of Fractional Oxidation Number | CHEMISTRY | JEE | Shishir Mittal Sir | Concept of the Day
Practice Problems:
Q1. Calculate fractional oxidation number for Red lead, Pb in Pb3O4.
Answer: (C)
Solution: Pb3O4 exist as PbO and PbO2, with the chemical formula 2PbO.PbO2.
Oxidation number of Pb in PbO = x
Oxidation number of Pb in PbO2 = y
Fractional or Average Oxidation number of Pb in Pb3O4 (2PbO.PbO2) =
Hence, option (C) is the correct answer.
Q2. Calculate fractional oxidation number for Br in Tribromine Octaoxide [Br3O8].
Answer: (D)
Solution: Three bromine atoms are present in the given compound [Br3O8].
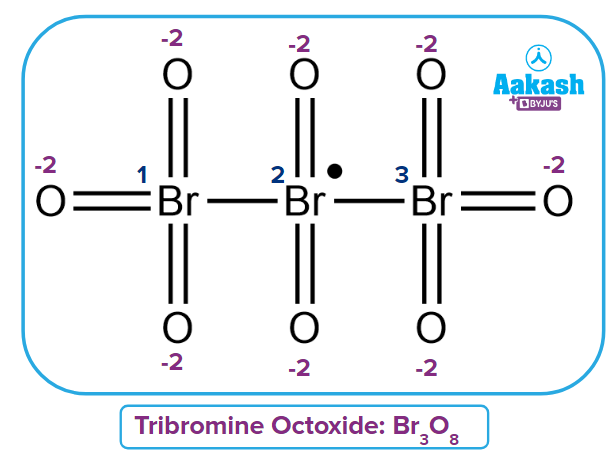
The fractional oxidation of Bromine can be calculated as:
Oxidation number of the First Bromine
Oxidation number of the second Bromine
Because the second bromine is surrounded by the first and third bromine, and there is a bond with the same atom, their contribution is assumed to be zero.
Oxidation number of the third Bromine
Fractional or Average Oxidation number of Br =
Hence, option (D) is the correct answer.
Q3. Calculate fractional oxidation number for C in Carbon suboxide C3O2.
Answer: (A)
Solution: Three carbon atoms are present in the given compound C3O2(O=C=C=C=O).
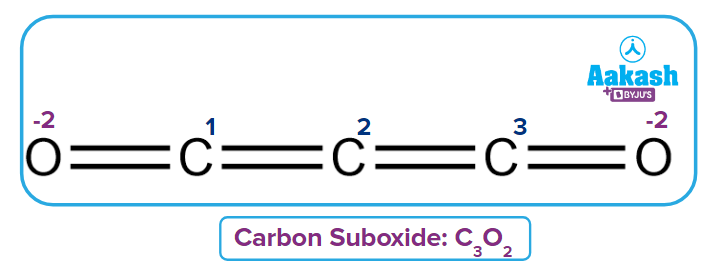
The fractional oxidation of Carbon can be calculated as
Oxidation number of the First Carbon 2
Oxidation number of the second Carbon
Because the second carbon is surrounded by the first and third carbon, and there is a bond with the same atom, their contribution is assumed to be zero.
Oxidation number of the third Carbon
Fractional or Average Oxidation number of C =
Hence, option (A) is the correct answer.
Q4. Calculate fractional oxidation number for C in Octane C8H18.
Answer: (D)
Solution: Three carbon atoms are present in the given compound C8H18.
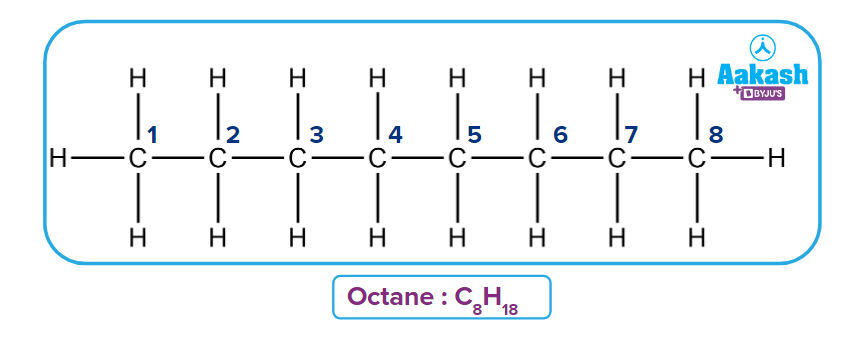
The fractional oxidation of Carbon can be calculated as
Oxidation number of the First Carbon
Oxidation number of the second Carbon
Because the second carbon is surrounded by the first and third carbon, and there is a bond with the same atom, their contribution is assumed to be zero.
Oxidation number for C3, C4, C5,C6 and C7 is same as for C2 because they are on equivalent positions.
Oxidation number of the eighth Carbon
Fractional or Average Oxidation number of C =
Hence, option (D) is the correct answer.
Frequently Asked Questions-FAQs:
Q1. What exactly is the distinction between oxidation state and oxidation number?
Answer: The primary distinction between oxidation number and oxidation state is that oxidation number is the charge of the central atom, whereas oxidation state is the number of electrons that a specific atom can lose, gain, or share with another atom.
The oxidation state can never be in fractions but the oxidation number can be in fractions.
But we often use both the terms oxidation state and oxidation number.
Q2. What practical applications does oxidation have?
Answer: Oxidation reactions involve either electron loss or oxygen addition. Daily life processes such as breathing, combustion, and plant photosynthesis all involve oxidation. Fats and oils are oxidized and rancidified as a result of this process.
Q3. Do you know where the term "oxidation state" came from, and why "reduction state" isn't used instead?
Answer: Antoine Lavoisier invented the phrase "oxidation" to describe a substance's reaction with oxygen, and I'd like to share it with you now. Additional mechanisms for losing electrons that do not require oxygen atoms were added to the concept. So, after a few additional improvements, we begin to streamline things by using the oxidation state term for both reduction and oxidation. Hence, it is just a convention.
Q4. What exactly is the paradox of fractional oxidation state?
Answer: Fractional oxidation state is defined as the average oxidation state of an element when two or more than two atoms are present in different oxidation numbers or present in different bonding situations in a given compound.


