-
Call Now
1800-102-2727
Ethers Chemical Properties: Introduction of Ethers, Chemical Properties of Ethers, Uses of Ethers, Practice Problems & Frequently Asked Questions(FAQs)
Ethers are the most useful solvents. For example, what kind of substance is used to sedate patients during surgery in earlier days? An ether-soaked cloth is used to sedate patients before surgery. Ethers are flammable liquids that release vapours that patients can breathe in or take intravenously. Before surgery, they cause patients to lose consciousness. For instance, analgesics and muscle relaxants aid Diethyl ether, a potent anaesthetic, work more effectively.
Diethyl ether is a solvent that is frequently used in laboratories. It only partially dissolves in water. Because of its low solubility and high volatility, it is the perfect non-polar solvent for liquid-liquid extraction.
Fuel, gum, oil, resin, and other materials can be dissolved using the solvent ether.
Due to their nearly inert nature and high dissolving power, they are also used as a reaction medium in some reactions, including the Wurtz reaction and the production of Grignard reagent and their reactions. Additionally, it helps keep things cool.
Then how much do we know about these ethers?
Table of Contents:
- Introduction of Ethers
- Chemical Properties of Ethers
- Uses of Ethers
- Practice Problems
- Frequently Asked Questions(FAQs)
Introduction of Ethers:
Ethers are organic substances made up of two alkyl or aryl groups, which may or may not be the same, and an oxygen atom in between them. Either R-O-R, R-O-Ar, or Ar-O-Ar can be used to represent the general formula for ethers. In the above formula, R denotes an alkyl group using the organic nomenclature, and Ar denotes an aryl group. These substances can be separated into two groups or types. The compound's attached substituent groups determine this classification. Therefore, we can classify them as:
- Symmetrical Ethers or Simple Ethers: On either side of the -O- functional group, these ethers are attached with the same alkyl or aryl groups.
Example: C2H5-O-C2H5 (diethyl ether), CH3-O-CH3 (dimethyl ether)
- Unsymmetrical Ethers or Mixed Ethers: On either side of the -O- functional group, these ethers are attached with the different alkyl or aryl groups.
Example: C2H5-O-CH3 (ethyl methyl ether), CH3-O-C6H5 (methyl phenyl ether).
Physical Properties of Ethers:
Ethers are colourless, odourless liquids that are very flammable and volatile. Alkyl or Aryl groups are hydrophobic and form H-bonds, which limit their solubility in water. Hydrogen bonding is absent within ethers. They have much lower boiling points than corresponding alcohols as a result. When inhaled, diethyl ether vapours cause unconsciousness, which is why they are used as an anaesthetic.
The reactivity of ethers is lower than that of compound functional groups. Active metals, powerful bases, and reducing and oxidising agents have no effect on them. The presence of an alkyl group, a lone pair of electrons on the oxygen atom, and the breaking of the C-O bond are what give ethers their chemical characteristics.
Chemical properties of Ethers:
Reactions of Aliphatic Ethers:
1. Reaction with Halogen acids:
Conc. HBr or HI cause ethers to react because they are sufficiently acidic to protonate the ethers.
Bromide and iodide are good nucleophiles for the substitution reaction. The order of reactivity is
HI > HBr >HCl
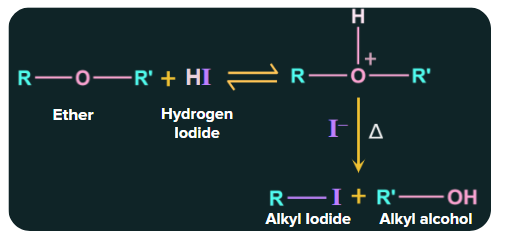
Here the larger alkyl group forms a bond with iodide and the smaller alkyl group forms a bond with alcohol and this reaction is Unimolecular nucleophilic substitution reaction of Ethers. Let see this with one example
2. Combustion:
Ethers combine explosively with air to produce carbon dioxide and water and are very flammable themselves.
3. Hydrolysis:
Alcohol is produced when ether is hydrolyzed in acid.
4. Reaction with Phosphorus Pentachloride (PCl5):
Alkyl chloride is produced as a result of the cleavage of the ether's C-O bonds during heating with phosphorus pentachloride (PCl5).
5. Peroxide Formation:
Because ethereal oxygen has a lone pair of electrons, it reacts with oxygen to form ether peroxide when it comes into contact with atmospheric oxygen in the presence of sunlight. Because ether peroxide is extremely unstable and violently explodes when heated, serious accidents frequently occur. Therefore, it is dangerous to boil a sample of ether that has been kept for a long time.
Reactions of Aromatic Ethers:
The most important aromatic ether is anisole. Methoxybenzene also referred to as anisole, is a chemical substance with the formula C6H5OCH3. Large number of its derivatives are present in both natural and artificial fragrances. It is a colourless liquid with an aroma resembling that of anise seeds. As a precursor to other synthetic compounds, the substance is primarily synthesised. It belongs to the ether family. The structure of anisole is shown below

Electrophilic Substitution Reaction:
Anisole reacts more quickly than benzene, which in turn reacts more quickly than nitrobenzene, in an electrophilic aromatic substitution reaction. These three sites receive preferential electrophilic substitution because the methoxy group is an ortho/para directing group. Anisole has a higher nucleophilicity than benzene, which is due to the methoxy group's influence on the ring's electron density. Despite the oxygen's electronegativity, the methoxy group has a significant impact on the ring's pi cloud as a mesomeric electron donor rather than an inductive electron-withdrawing group.
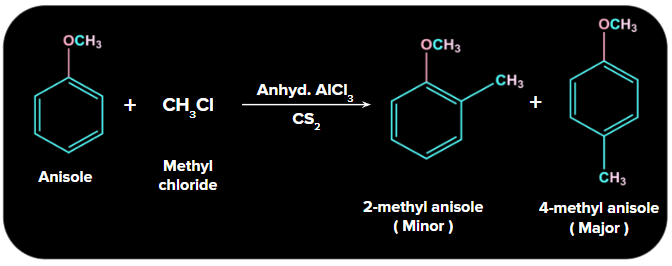
1. Friedel Crafts Alkylation:
In the Friedel-Crafts alkylation reaction, an alkyl group is added to anisole at the ortho and para positions via reactions with an alkyl halide in the presence of anhydrous aluminium chloride (a Lewis acid) as a catalyst.
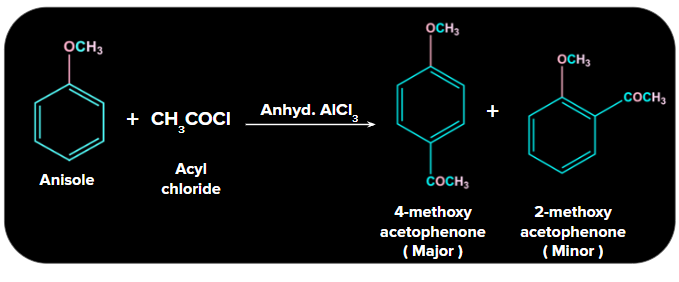
2. Friedel Crafts Acylation:
In the Friedel-Crafts reaction, the acyl group is added to anisole at the ortho and para positions via reactions with an acyl halide in the presence of anhydrous aluminium chloride (a Lewis acid) as a catalyst.
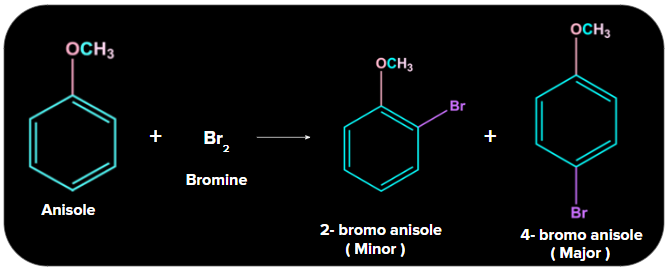
3. Halogenation:
Bromination of anisole with bromine yields a mixture of ortho and para Bromo anisole. It exemplifies the electrophilic aromatic substitution reaction. The bromine atom replaces the H atom on the benzene ring. The methyl group is an ortho para directing group.
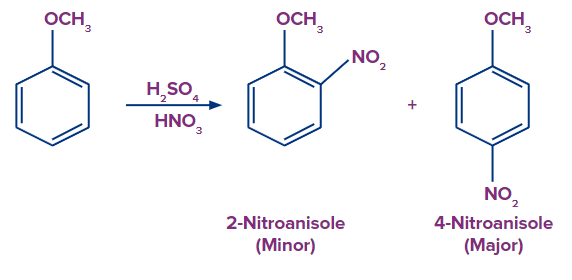
4. Nitration Reaction:
Ortho and para nitro anisole are formed when concentrated sulphuric and nitric acids react with anisole.
Uses of Ethers:
- Ethers are frequently used in the fragrance and scent industry due to their olfactory and organoleptic qualities. They enhance the flavour of a variety of foods and medicines, including desserts, fruit jams, yoghurt, ice cream, chewing gum, fruit-based foods and beverages, and some pharmaceutical preparations. desserts, jams, ice cream, yoghurt, chewing gum, fruit-based foods and beverages, as well as some pharmaceutical preparations.
- Some new ether compounds, like diethyl ether, are especially helpful in the production of fragrances or fragrant goods like soaps, detergents, and common household items like essential oils, medications etc.
- Drinks are given an exotic, fruity-green flavour by 1-methoxy-3-hexanethiol, which also gives them a grapefruit flavour.
- Some ethers are also used to flavour foods like soups and broths, seasonings, snacks, sauces, and ready-to-eat non-vegetarian meals with salty aromas, particularly meat-like aromas.
- A typical ingredient used to give anise seed essential oil fragrance is methyl benzene (anisole).
- As an aprotic solvent, diethyl ether is frequently used in laboratories. In Grignard's reactions, it is primarily used as a solvent.
- Tetrahydrofuran (THF) is a cyclic ether frequently employed as an organic compound solvent. Due to their low boiling point, they are better suited for use as solvents because evaporation makes it simple to remove them from a compound after a chemical reaction.
- Lithium batteries utilise the solvent dimethyl ether (DME), which is quite miscible in water.
- The cyclic ether 1,4-dioxane is widely used as a solvent in labs for the synthesis of chemicals because it is water soluble.
Practice Problems:
Q1. Methanol on reaction with sulphuric acid gives
(A) Dimethyl ether
(B) Diethyl ether
(C) Both A and B
(D) None of the above
Answer: (A)
Solution: Formation of dimethyl ether from methanol and sulphuric acid is an example of the dehydration reaction. The following is the reaction:
Q2. When phenol is combined with sodium hydroxide and methyl iodide, it produces
(A) Anisole
(B) Phenoxy benzene
(C) Benzene
(D) None of the above
Answer: (A)
Solution: Anisole is produced when phenol reacts with sodium hydroxide to form the phenoxide ion, which then interacts with methyl iodide in a process known as the Williamson ether synthesis. Following is the reaction:
Q3. Which of the following functional groups is the least reactive?
(A) Alcohols
(B) Ethers
(C) Aldehydes
(D) Ketones
Answer: (B)
Solution: Ethers are significantly more reactive than alkanes but relatively less reactive than alcohols and carbonyl compounds.
Because there is no polarity or charge separation in the molecule, ethers are less reactive. Unlike carbonyl carbon, there is no transient or partial positive charge. As a result, nucleophiles interact with ethers without consequence (no reaction takes place). As a result, bases, alkali metals, active methylene groups, etc. have no effect on ethers.
Only when oxygen is protonated in an acidic medium are ethers reactive. To remove the positive charge on the ether oxygen, the ether goes through several reactions. The most well-known cleavages are SN1 and SN2
Q4. What isomers are ethers and alcohols?
(A) Functional isomers
(B) Chain isomers
(C) Position isomers
(D) None of the above
Answer: (A)
Solution: Because they have a similar molecular formula but a different structure and come from different homologous series, alcohols and ethers are functional isomers.
Frequently Asked Questions(FAQs)
Q1. Are ethers polar or nonpolar?
Answer: Ethers are polar compounds. The reason for this is that the electronegativity of the oxygen and carbon atoms differs. The oxygen atom draws the shared pair of electrons due to the difference in electronegativity, which causes a partial negative charge on the oxygen atom and a partial positive charge on the carbon atom.
In case of ether, two polar C-O bonds are inclined toward one another at an angle of 111.7 degrees. The two dipoles do not cancel out as a result, leading to a net dipole moment. In other words, the dipole moment in ether is ≠0
The polarity of ethers is higher than that of alkenes but lower than that of alcohols, esters, or amides.
Q2. Why does ether have a lower boiling point than isomeric alcohols?
Answer: Because ethers lack intramolecular hydrogen bonds, their boiling point is lower than that of isomeric alcohols but closer to that of alkanes of comparable mass. Despite both having the same chemical formula( C2H6O) as alcohol and ethers, methoxy methane (CH3OCH3) has a lower boiling point than ethanol (CH3CH2OH).
Q3. Even though they have the same molecular formula, ethers are more volatile than alcohols. Why?
Answer: An ether has a higher volatility than alcohol with the same molecular formula. This is because intermolecular hydrogen bonding occurs in alcohols. In alcohols, the H atom is joined to the electronegative oxygen atom. As a result, the H atom of one alcohol molecule's OH group forms a hydrogen bond with the oxygen atom of the second alcohol molecule's OH group. As a result, intermolecular association occurs. It raises the boiling point of alcohol while decreasing its volatility. Others, such as ethers, do not allow for intermolecular hydrogen bonding because the H atom is not attached to an electronegative oxygen atom.
Q4. Why does diethyl ether act as a base?
Answer: Strong Lewis base diethyl ether reacts with a variety of Lewis acids. The oxygen atom in a diethyl ether molecule behaves as a base because it has two lone pairs of electrons available for coordination.


