-
Call Now
1800-102-2727
Electrophile and Nucleophile: What is Electrophile and Nucleophile?, Types, Characteristics, Difference Between Electrophile and Nucleophile
What if you win a lottery? Now you have a lot of money. So, if any poor comes to your door, obviously you will help that poor and willingly give some money to that poor.

Similarly, in chemistry, we have electron-rich species that are willing to donate electrons and electron-deficient species that are poor and in need of electrons. Such electron-rich species are known as a nucleophile and electron-deficient species are known as electrophiles.
Table of content
- Introduction
- Electrophile(E+)
- Characteristics of Electrophile
- Types of Electrophile
- Nucleophile (Nu-)
- Characteristics of Nucleophile
- Types of Nucleophile
- Difference between Electrophile and Nucleophile
- Practice Problems
- Frequently Asked Questions - FAQs
Introduction
An attacking reagent is a species that reacts with a substrate molecule or intermediate and produces a product. There are two types of reagents:
1. electrophiles
2. Nucleophiles
Electrophiles and nucleophiles are chemical species that accept or donate electrons respectively in order to establish a new chemical bond. Meanwhile, the terms electrophile and nucleophile are used to characterize the reactions that occur between electron donors and acceptors. These are the most essential organic chemistry principles. They were first used in 1933, and they superseded older terminology like cations and anions.
Electrophile (E+)
The word "electrophile" comes from the word "electro," which is derived from the word "electron," and "phile," which means "love."
An electrophile is a molecule, ion, or atom that is lacking in electrons in some way. In other terms, an electrophile is a chemical species that assaults the molecule's negative or loves electrons. They are usually positively charged or neutral species with vacant orbitals (electron-deficient molecules). Electrophiles are capable of accepting a few electrons.
Characteristics of Electrophile
- They are electron-deficient and have a strong attraction to electrons.
- They have either a positive charge or a neutral species.
- They attack places with a lot of electrons, like carbon-carbon double bonds.
- The density influences electron transport, which normally occurs from a high-density to a low-density location.
- Electrophilic addition and electrophilic substitution processes should be preferred under these reagents. Because they take electrons, they are also known as Lewis acids.
Types of Electrophile
1. Positively Charged Electrophiles: These are positively charged species carrying a positive charge on the central atom, and they act as a lewis acid.
Example-
2. Neutral Electrophiles: They are electron deficient molecules having incomplete octet.
They involve lewis acids like
3. Free radicals: Free radical species containing unpaired electrons like nitrenes and carbenes also act as electrophiles. Example- CH2(carbene)
Nucleophile (Nu-)
The electron-rich species is a nucleophile in general. A nucleophile is a substance that donates an electron pair in order to create a covalent bond or coordinate bond. A nucleophile is often negatively charged or neutral with a single pair of donatable electrons. Examples include H2O, , and .
The word nucleophile is derived from two words: "Nucleo," which refers to the nucleus, and "philic," which means "to love." Nucleophiles are species that target the substrate's positive side or prefer the nuclei.
According to Lewis' theory of acids and bases, nucleophiles give unshared electron pairs (lone pairs) and act as Lewis bases.
Characteristics of Nucleophiles
- They are made up of electrons and are attracted to the nucleus. They are either negatively charged or neutral(Lewis base).
- They are electron donors.
- Nucleophiles always attack on the electron deficient site.They can be used in nucleophilic addition and substitution processes.
Types of Nucleophile
1. Negatively Charged Nucleophiles: These are electron-rich species having negative charge over the atom/molecule.
Examples of negatively charged nucleophile:
2. Neutral Nucleophiles: These are electron-rich species having lone pairs of electrons.
Examples of neutral nucleophile: etc.
3. Ambident Nucleophiles Some nucleophiles have a pair of electrons on two or more atoms(more than one donor atoms) or canonical forms can be drawn in which two or more atoms bear an unshared pair of electrons. Nucleophiles which have two attacking sites, one negatively charged and one neutral site, are known as ambident nucleophiles.
Example-, has two sites of attack i.e. O, N as O has two and N has one lone pair of electrons respectively, -C has two sites of attack i.e. C, N etc.
Difference between Electrophile and Nucleophile
|
Electrophile |
Nucleophile |
|---|---|
|
It accepts the electron pair. |
It donates the electron pair. |
|
They may be positively charged or neutral (having empty orbital). |
They may be negatively charged or neutral (having a lone pair of electrons). |
|
Attacks the electron rich center. |
Attacks the electron-deficient centre. |
|
These are also known as Lewis acid because of their tendency to accept a pair of electrons. |
These are also known as Lewis base because of their tendency to donate a pair of electrons. |
|
It forms a bond with the nucleophile on which it attacks. |
Its covalency increases after it attacks the electrophile. |
Practice problems
Q1. Among the following X is the number of electrophiles and Y is the number of nucleophiles. Report your answer as XY.
A). 32
B). 23
C). 33
D). 31
Answer: C
Solution: All the positively charged species are electrophile, example- , , and negatively charged species are nucleophile, example- . Also neutral species like ⠆ carrying a lone pair of electrons belongs to the category of nucleophiles.
Q2. Which of the following is an electrophile?
A). ![]()
B). AlCl3
C). BH3
D). All of the above
Answer: D
Solution: Generally the positively charged species carries a positive charge on the central atom. like ![]() acts as an electrophile. Electron deficient molecules having incomplete octet of the central atoms belong to the category of electrophiles. They involve lewis acids like
acts as an electrophile. Electron deficient molecules having incomplete octet of the central atoms belong to the category of electrophiles. They involve lewis acids like
Q3.
In the above given specie, electron donating atoms are:
A).
B). O
C). -R
D). Both A and B
Answer: D
Solution: Given species is an ambident nucleophile. Such nucleophiles have more than one electron-donating atom. Given specie undergo resonance as follows:
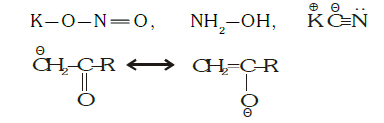
Therefore both and O act as a nucleophile at a time.
Q4. The decreasing order of nucleophilicity among the nucleophiles is:
A). I > II> III
B). III> I > II
C). I > III >II
D). II > III > I
Answer: C
Solution: As we go from left to right in a period electronegativity increases therefore tendency to donate electrons also decreases. So is a better nucleophile than . But as we go down the group, the size of the atom increases, so its effective nuclear charge also decreases, because of which its tendency to hold valence shell electrons also decreases. Hence, order of nucleophilicity of given nucleophiles is:
Option C, is the correct answer.
Frequently Asked Questions-FAQs
Q1. Do all species carrying a positive charge are electron deficient?
Answer: No, all species carrying a positive charge are not electron deficient. Example-Hydronium ion, Although the oxygen atom has a positive charge, it does not qualify as an electrophile as it has completely filled octet.
Ammonium ion, . The ammonium ion, on the other hand, does not have any unoccupied orbitals to attract electrons. As a result, ammonium ions are not considered electrophiles.
Q2. What is an electrophilic substitution reaction?
Answer: If a substitution reaction is brought about by an electrophile, then it is known as an electrophile substitution reaction.
The general reaction represents the electrophilic substitution reaction.
Where E+ is Electrophile and lg is leaving group.Electrophilic substitution processes such as Friedel Craft's acylation and alkylation are well-known.
Q3. Are alkenes nucleophiles or electrophiles?
Answer: Alkenes consist of electron rich C=C pi bond. Electrons are loosely held in a Pi bond. Therefore it is nucleophile.
Q4. Are alkanes nucleophiles or electrophiles?
Answer: Alkanes consist of strong C-C , C-H bond. They are non-reactive, stable compounds.
Therefore in nature, alkanes are neither nucleophilic nor electrophilic.


