-
Call Now
1800-102-2727
Difference Between Atom and Ion – Atoms, Ions, Comparison of Properties of Atoms and Ions, Practice Problems and FAQ
Every matter surrounding us is composed of atoms. They are basically the smallest unit or building blocks of anything that has or had an existence on earth!
But what really are atoms?
Atoms are the single units that constitute elements we know of from the modern periodic table. Only roughly 100+ different elements and, thus, only 100 different types of atoms account for the diversity of chemical activity. Atoms are identical to the 26 English alphabets. They combine in a variety of ways to create the enormous number of words that make up our language. Just like each English alphabet is formed by drawing strokes of lines, curves and patterns, atoms are formed of subatomic particles– neutrons, protons and electrons.

Ions are obtained when atoms interact with each other.
However, how do the atoms interact with one another? What guidelines define the possible combinations between them?
It all is decided by the subatomic particle ‘electrons’ present in the atomic orbits. The tendency of atoms to accept or donate or share electrons is the determining force here. Ions are atoms which are either devoid of one or more of their electrons or consisting of additional electrons. A specific atom's derivative is an ion. Atoms don't have a net electrical charge, which is the primary distinction between them and ions.
Let us understand in detail about atoms, ions, and their differences in this article.
TABLE OF CONTENTS
- Atoms
- Ions
- Difference between Atoms and Ions
- Practice Problems
- Frequently Asked Questions – FAQ
Atoms
The smallest particle of matter that still has the properties of the bulk element is an atom. It is made up of a varying number of fundamental particles namely, neutrons, electrons, and protons. Atoms cannot be further broken down into smaller parts without loss of original characteristics. The Greek term "atomos," from which the English word "atom" was derived, signifies "cut" and "not" respectively. Together it meant cutting was impossible. A long time ago, atom research was first initiated, and it is still ongoing now. Every substance that we can see is made up of atoms, according to the basic definition of an atom.
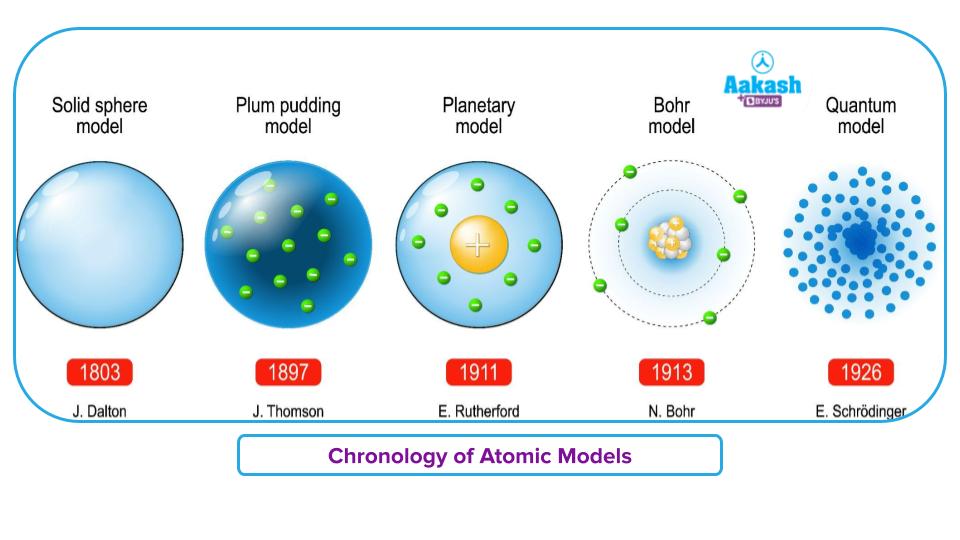
Theories of Atoms
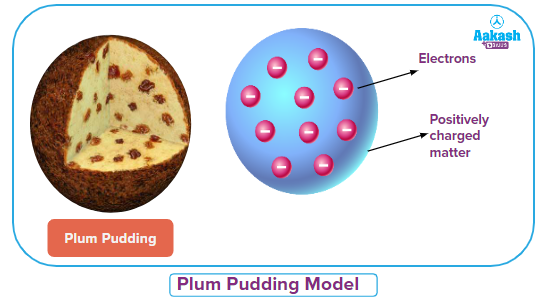
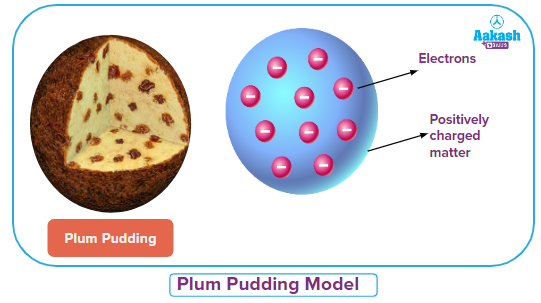
English chemist John Dalton proposed for the first time the term ‘atom’ and defined the characteristics of an atom in 1803. The theory was called Dalton's atomic theory. The atoms are thought to be the fundamental units of matter. But it took a while for this understanding of atoms to emerge. In 1815, William Prout proposed that elements are composed of hydrogen atoms. This theory was rejected because the atomic weight of some elements could not be compared to that of hydrogen. J.J. Thompson carried out numerous experiments in 1889, and on the basis of his findings, he proposed the "plum pudding model" of an atom. According to this model, an atom is a positively charged sphere and negatively charged particles (electrons) are embedded inside of it like a plum in a pudding. The negative charge balances out the sphere's positive charge. So an atom is devoid of electrical charge.
Ernest Rutherford revealed the fundamental makeup of an atom in 1909 using his well-known alpha-ray
scattering experiment. According to Rutherford’s atomic model, an atom has a nucleus in the centre and electrons revolve around it. The negative charge of the electrons balances out the positive charge of the nucleus. However, his theories were unable to provide specifics regarding the stability of an atom.
![]()
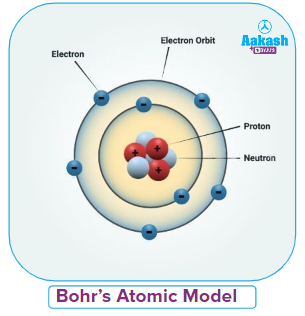
Niels Bohr in 1913, proposed a detailed quantized model of an atom that accounts for the atom's stability. His observations were highly effective. Some notions, like the fine structure of the hydrogen spectral lines and the dispersion of electrons, could not be explained by Bohr's atomic model. Bohr's atomic model was updated by Sommerfeld, who also clarified the electron's route and added to the understanding of the structure of the hydrogen spectral lines.
Properties of Atoms
- Atoms are called the fundamental unit of matter. It is made up of neutrons, electrons, and protons, and it cannot be further broken down into smaller bits.
- Atomic reactions are what cause chemical reactions to occur. A molecule is created when two or more atoms combine.
- The quantity of protons and electrons in a neutral atom is equal. Protons and neutrons make up the atom's nucleus.
- The electrons surround the nucleus and are placed on various levels of orbitals.
- Atoms come together to create molecules.
- The periodic table is set up so that as we move down a group, the atomic radius increases and as we move across the period from left to right, the atomic radius decreases.
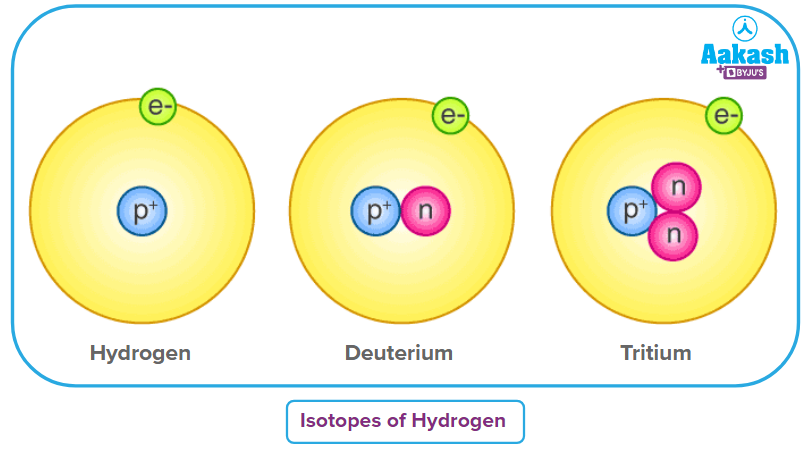
- Nuclear properties: Two atoms that have the same proton number are a part of the same chemical element. Atoms that have the same proton number but a neutron number being different are called isotopes. Example: Isotopes of Hydrogen.
|
Atom |
Mass |
Number of Protons |
Number of Neutrons (Mass -Number of Protons ) |
|
Hydrogen (1H1) |
1 |
1 |
0 |
|
Deuterium (1H2) |
2 |
1 |
1 |
|
Tritium (1H3) |
3 |
1 |
2 |
- Mass: Every atom has a maximal mass that is made up of a mix of neutrons and protons. The word "mass number" refers to the total number of protons and neutrons present. The mass number must always be a positive whole integer and be dimensionless. The unit expressed for the mass of an atom is daltons (Da) or amu or u.
- Shape and Size: Atoms do not possess a well-defined boundary, hence their dimensions are defined by atomic radius. It is measured in terms of its distance where the cluster of electrons expands from its own nucleus. The smallest recorded atom is helium. It has a radius of 32 pm.
- Isobars: Isobars are atoms that belong to distinct elements that have different atomic numbers (Z) and the same mass number (A), so they have the same number of nucleons. Example: 18Ar40 and 19K40.
- Isotones: Isotones are atoms of different elements that share the same number of neutrons. Examples:8O16 and 6C14. Neutrons in oxygen (A-Z) = 16-8 = 8 and Neutrons in carbon (A-Z) = 14-6 = 8.
Ions
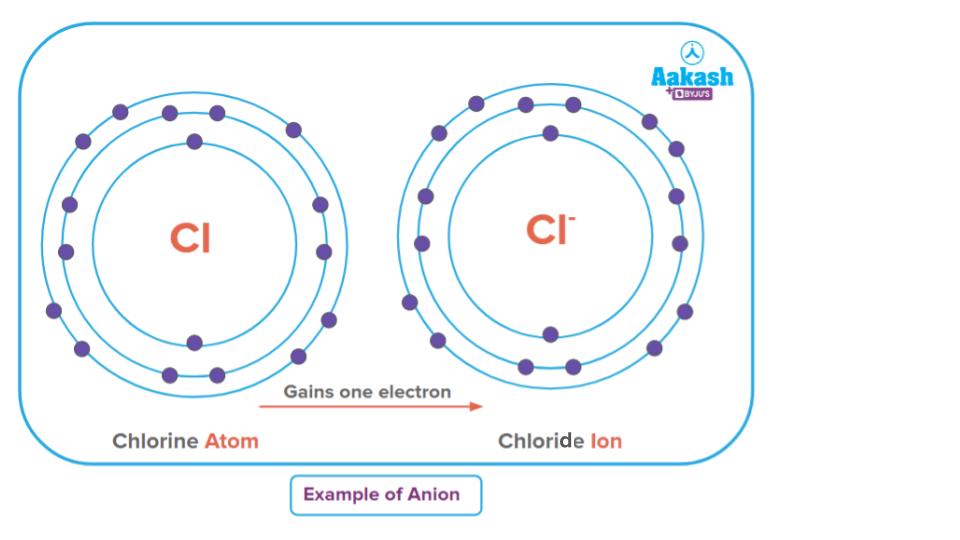
Ions are species with an unequal number of electrons and protons compared to a neutral atom with the same number of protons and electrons. Ions are usually charged particles. The ions can be either positively charged or negatively charged. An atom, gaining an electron from other sources acquires a negative charge and is called an anion. The anion will have more electrons than the number of protons.
Example: Cl-
In a neutral Cl atom, the number of protons and electrons is equal to 17. But in the case of chloride ion, Cl-, the number of electrons is 18, whereas the number of protons is 17. Hence, it is an anion. The number of negative charges on the atom indicates how many electrons the atom has gained as compared to its neutral state.
On the other hand, a neutral atom can lose an electron, by which the number of protons is greater than the number of electrons, and it becomes positively charged. Such positively charged species are called cations.
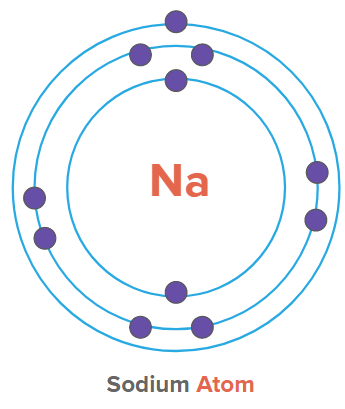
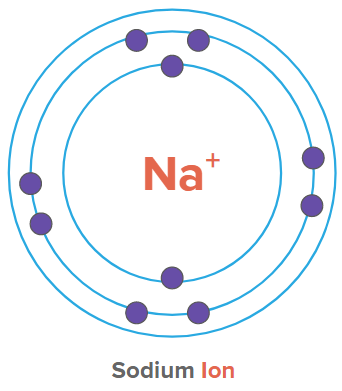
Example: Na+
In a neutral Na atom, the number of protons and electrons is 11. But in the case of Na+, the number of electrons is 10, whereas the number of protons is 11. Hence, it is a cation. The positive charge on the atom indicates how many electrons the atom has lost as compared to its neutral state.

Properties of Ions
- When ions are in their gaseous state, they are highly reactive and hence, unstable. To produce ionic salts or neutral molecules, it quickly reacts with ions that have the opposite charge.
- Ions are released in both the solid and liquid states when salts and solvents interact, creating solvated ions.
- A proton carries an electric charge that is equivalent to the charge of an electron. Two types of ions exist. In general, two types of ions exist. Anions are particles in which there are more electrons than protons. The negative symbol signifies it. Cations are particles in which there are fewer electrons than protons. The positive sign signifies it.
- Monatomic Ions - Monatomic ions are ions that only have one atom. Examples: Na+, Cl-, Br- and K+ ions.
- Polyatomic Ions - Polyatomic ions are ions that have more than one atom in them. Examples: CO32-, Cr2O72-, CH3COO-.
Difference between Atoms and Ions
|
Atom |
Ion |
|
The smallest piece of matter that may exist is the atom. |
It can exist as a single particle or as a collection of particles. |
|
It cannot exist independently in a solution. |
It can exist independently in a solution. |
|
Atoms combine together to generate molecules either by electrovalent, covalent or dative bonds. |
An electrovalent bond is formed between two oppositely charged ions. Same charged ions repel each other and do not come close. |
|
The number of electrons and protons is the same. |
Electrons and protons are unequal in number and hence the ions are unstable. |
|
|
|
|
Atoms are neutral in nature. Example: Hydrogen atom (H-atom). |
Ions are either positively or negatively charged. Examples: Na+, Cl-, etc. |
|
Atoms are unattracted by electric fields as they are neutrally charged. |
Ions are attracted towards the electric field depending on their charge. |
Recommended Videos
Electronic Configuration of Atoms - Atomic Structure Class 11 Chemistry (L12) | NEET 2024 Exam
Subatomic Particles & Atomic models - Atomic Structure Class 11 Chemistry Concepts | JEE Main 2024
Structure of Atom Class 11 One Shot (Full Chapter Revision) | JEE Chemistry One Shot | JEE Main 2022
Structure of Atom Class 11 Chemistry (Ch-2) | JEE Main 2023 Important Topics | JEE 2023 Exam Prep
Practice Problems
1. Which of the following scientists introduced the plum pudding model?
a. Rutherford
b. Bohr
c. J.J Thompson
d. Dalton
Answer: C
Solution: J.J. Thompson carried out numerous experiments in 1889, and on the basis of his findings, he proposed the "plum pudding model" of an atom. According to this model, an atom is a positively charged sphere and negatively charged particles (electrons) are embedded inside of it like a plum in a pudding. The negative charge balances out the sphere's positive charge. So an atom is devoid of electrical charge.
So, option C is the correct answer.
2. Which of the following is not a subatomic particle?
a. Electron
b. Molecule
c. Proton
d. Neutron
Answer: B
Solution: The atom consists of 3 important subatomic particles which are electrons, protons and neutrons. When two or more atoms combine, they form a molecule. Hence, a molecule is not a subatomic particle.
So, option B is the correct answer.
3. Which of the following statements about atoms is untrue?
a. The number of electrons and protons is always equal in a neutral atom.
b. Electrons are negatively charged.
c. Protons are positively charged.
d. Atoms always exist independently.
Answer: D
Solution: An atom is the fundamental unit of matter. It cannot be further divided into smaller parts and comprises neutrons, electrons, and protons. Chemical reactions take place because of the reactions between atoms. Atoms combine with each other to form molecules. In a neutral atom, the number of protons and electrons is equal. Electrons are negatively charged whereas protons are positively charged particles. Since the majority of atoms of elements lack an octet configuration and strive to achieve a noble gas structure, they are not always able to exist independently. Atoms react and form bonds as a result, making it impossible for them to exist alone.
So, option D is the correct answer.
4. In which of the following scenarios an atom will be considered an anion?
a. If the number of electrons is more than the number of protons in an atom.
b. If the number of electrons is less than the number of protons in an atom.
c. If the number of electrons is equal to the number of protons in an atom.
d. If electrons are absent in an atom.
Answer: A
Solution: An atom is considered to be an ion if it is electrically charged. An atom is called an anion if the number of electrons is greater than the number of protons. And an atom is considered a cation when the number of protons is greater than the number of electrons.
So, option A is the correct answer.
Frequently Asked Questions – FAQ
1. Do ions need to be free to conduct electricity?
Answer: Ions in ionic compounds are free to move about, which allows them to conduct electricity when they are liquid or dissolved in water. Because their ions are locked in fixed places and are unable to move, ionic compounds cannot conduct electricity when they are solid. Hence, ions need to be free to conduct electricity.
2. Between an atom and an ion, which one is more stable?
Answer: Atoms can not exist independently. They combine with other atoms to acquire stability. Whereas ions can exist independently as they acquire noble gas configuration. Hence, ions are more stable than an atom.
3. Where can one find negative ions naturally?
Answer: Positive ions are damaging to the body while negative ions are healthy. Pure, fresh air has the highest quantities of negative ions. In nature, negative ions are prevalent, particularly around waterfalls, on the ocean's surface, at the beach, and all over the mountains and forests. Negative ions have a number of advantages, but their ability to rid the air of allergens including pollen, mould spores, bacteria, and viruses is their most significant advantage.
4. What distinguishes one atom from another?
Answer: The distinguishing characteristic of an atom is its number of protons or electrons. It is what distinguishes one element from another. The atomic number of an atom is the sum of all of its protons or electrons. 8 protons and 8 electrons make up an oxygen atom, whereas 6 protons and 6 electrons make up a carbon atom.