-
Call Now
1800-102-2727
Examples of Condensation Polymers with Monomers- Monomer and the Condensation Polymer, Characteristics of the condensation polymer, Examples, Practice Problems and FAQs
Can you guess why you don't get an electric shock when you unintentionally or intentionally switch on the fan in your home?
Electric switches though help in the conduction of electricity from one source to another still it is generally safe when touched with a naked hand.
But do you know which insulating material is used for making electric switches?
A material that does not conduct electricity is known as an insulator. Nonmetal is an excellent insulator, and the insulator that protects us from getting electric shock is a polymer known as bakelite formed by the step-growth polymerization technique in which the smaller molecules which are also known as monomers react with each other to form larger structural units known as polymers, releasing also small by-product molecules. Polymer formation or polymerization process can take place in many ways. Let us learn in this article about the polymer formed by what is called condensation method..

Table of Content
- Monomer and the Condensation Polymer
- Characteristics Properties of Condensation Polymerisation
- Examples of Condensation Polymers with Monomers
- Practice Problems
- Frequently Asked Questions-FAQs
Monomer and the Condensation Polymer
Polymerization is generally defined as a chemical reaction between many molecules of the same or different substance called monomers that results in the formation of either a long linear polymer chains or three-dimensional networks. Polymerization is typically accomplished through a variety of reaction mechanisms that are influenced by factors such as the functional groups of the reacting compounds and their non-bonding interactions.
Condensation polymers are created through a step-growth polymerization technique in which smaller molecules or monomers react with one another to form larger structural units (called polymers) while releasing a small by-product molecule like water. Condensate is the common name for the by-products.
Two bifunctional monomers condense to form dimers, which then form tetramers, and so on, with the loss of simple molecules such as H2O, CO2 and HCl in the process.
Characteristics Properties of Condensation Polymerisation
- One or two functional groups should be present in the monomer molecules (like alcohol, amine, or carboxylic acid groups).
- The reaction takes place between two functional groups or monomers that are either similar or dissimilar. It can occur between a dimer and an oligomer, one monomer and one dimer, or a polymer chain and another polymer chain.
- Smaller molecules typically join together to form larger molecules through a process known as polymerisation.
- The mixed properties of both molecules or functional groups are considered.
- When both functional groups are difunctional, the condensation product is generally a linear polymer.
- When one of the functional groups is tri- or tetra-functional, the polymer formed is a three-dimensional cross-linked polymer.
- When monomers with one reactive group are combined, the average molecular weight decreases. As a result, the average molecular weight and cross-link density are determined by the functionality of each monomer.
Examples of Condensation Polymer With Monomers
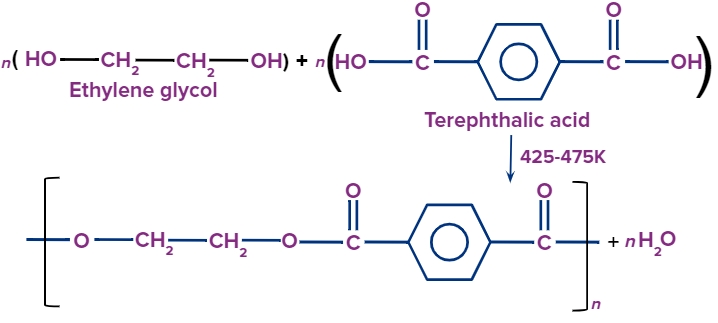
Terylene:
It is a polymer produced by polymerizing ethylene glycol [OH-(CH2)2-OH] and terephthalic acid [HOOC - C6H4 - COOH].
- It is crease-resistant, long-lasting, and contains little moisture. It's resistant to both alkaline and acidic substances, as well as pests like moths.
- It is used in the production of washable fabrics.
- Terrycot and Terrywool are produced by combining polyester and cotton, respectively.
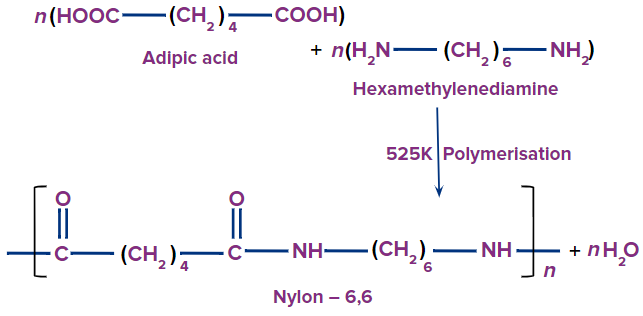
Nylon-6,6:
Nylon-6,6 is produced by the condensation polymerisation of hexamethylenediamine and adipic acid.
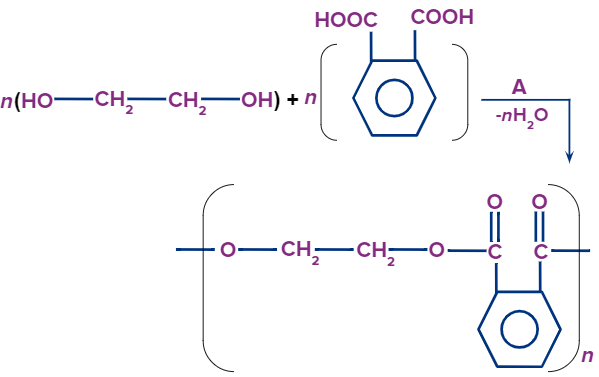
Glyptal:
- Glyptal is produced by the condensation polymerisation of phthalic acid and ethylene glycol.
- Polyethene phthalate is a thermoplastic that is used in paints and lacquers as an adhesive.
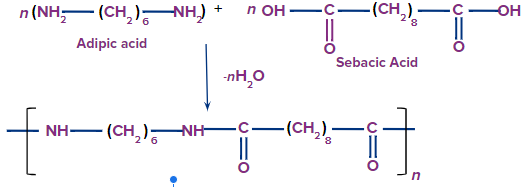
Nylon-6,10:
Nylon-6,10 is produced by the condensation polymerisation of hexamethylenediamine and sebacic acid.
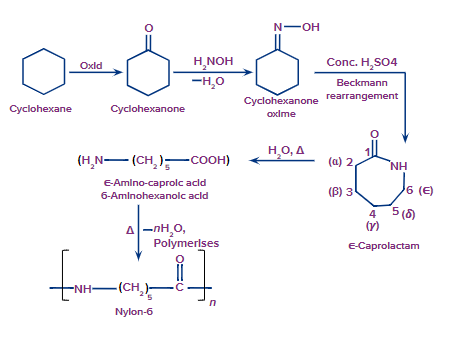
Nylon-6:
Nylon-6 is produced by heating caprolactam at temperatures ranging from 533 K to 543 K. Nylon-6 is also known as Perlon-L.
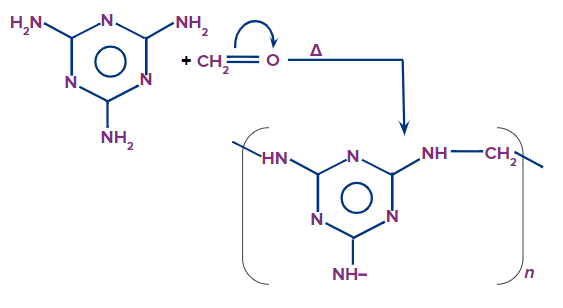
Melmac:
- Melmac is produced by polymerising melamine and formaldehyde.
- It is used to make unbreakable plastic crockeries such as plates and cups.
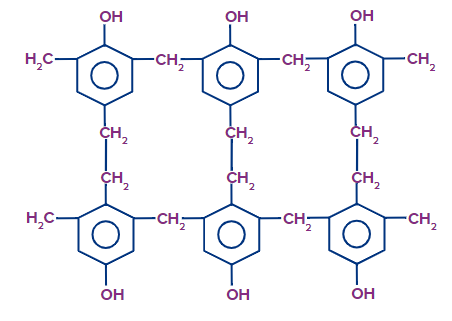
Bakelite:
It is a condensation polymer constituted from the monomers of phenol and formaldehyde.
- Polyoxybenzyl methylene glycol anhydride is another name for bakelite..
- It is one of the first synthetic polymers.
- It is also a type of thermosetting plastic.
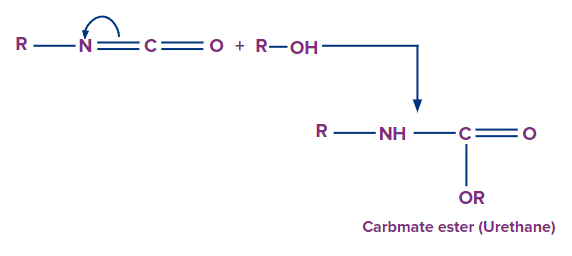
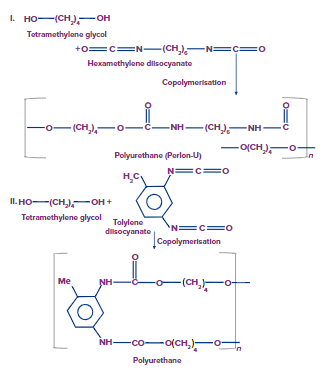
Polyurethanes:
- Isocyanates react with alcohols to form urethanes, which are carbonate esters. Polyurethanes are formed when diols react with diisocyanates.
- Polyurethanes are used to create polyurethane foams (PUF).
- A small amount of water is added during the PUF manufacturing process, which reacts with some of the diisocyanates to produce carbamic acid.
- Carbamic acid rapidly loses CO2 to form an aromatic diamine.
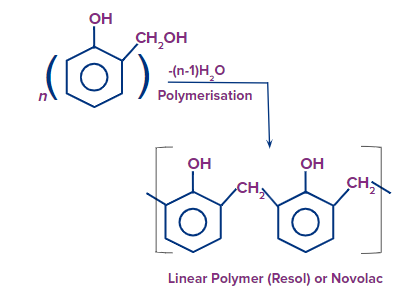
Novolac or Resol:
- Novolac (or) Resol is made by polymerizing phenol or substituted phenol with formaldehyde.
- It is a thermoplastic that is used in the production of adhesives. This polymer is formed when the phenol to formaldehyde ratio (P/F ratio) is greater than one.
Practice Problems
Q1. The monomer required for the formation of bakelite is_________.
- Phthalic acid and ethylene glycol
- Phenol and formaldehyde.
- Caprolactam
- Ethylene glycol and terephthalic acid
Answer:(B)
Solution: Glyptal is produced by the condensation polymerisation of phthalic acid and ethylene glycol. Nylon-6 is produced by heating caprolactam at temperatures ranging from 533 K to 543 K. Nylon-6 is also known as Perlon-L. Terylene is a polymer produced by polymerizing ethylene glycol and terephthalic acid. Bakelite is a condensation polymer constituted from the monomers of phenol and formaldehyde. Polyoxybenzyl methylene glycol anhydride is another name for bakelite. Therefore, option (B) is correct.
Q2. Which of the following option is correct with respect to the condensation polymer?
- When monomers with one reactive group are combined, the average molecular weight decreases in condensation polymerisation.
- Two bifunctional monomers condense to form dimers, which then form tetramers, and so on, with the loss of simple molecules such as H2O, CO2 and HCl in the process.
- Condensation polymers are formed through a step-growth polymerization technique
- All of the above
Answer:(D)
Solution: Condensation polymers are formed through a step-growth polymerization technique in which monomers with one reactive group are combined, the average molecular weight decreases in condensation polymerisation. Two bifunctional monomers condense to form dimers, which then form tetramers, and so on, with the loss of simple molecules such as H2O, CO2 and HCl in the process.
Q3. The monomer required for the formation of terylene is_________.
- Phthalic acid and ethylene glycol
- melamine and formaldehyde
- Caprolactam
- Ethylene glycol and terephthalic acid
Answer: (D)
Solution: Glyptal is produced by the condensation polymerisation of phthalic acid and ethylene glycol. Melmac is produced by polymerising melamine and formaldehyde. Nylon-6 is produced by heating caprolactam at temperatures ranging from 533 K to 543 K. Nylon-6 is also known as Perlon-L. Terylene is a polymer produced by polymerizing ethylene glycol [OH-(CH2)2-OH] and terephthalic acid [HOOC - C6H4 - COOH]. Therefore, option(D) is correct.
Q4. List some important use of the polymers formed by the condensation polymerisation method.
Answer: Some important use of the condensation polymers are:
- Among the synthetic polyamides are urea-formaldehyde resins. They are used in the production of moulded items, as well as textile and paper adhesives and coatings.
- Polyisoprene and polychloroprene are both essential in the production of rubber. These rubbers are used in the production of tyres and tubes, hoses, balls, and conveyor belts, among other things.
- Mixed organic-inorganic compounds are a type of polymer that is used to make nonstick utensils and nonstick sprays in the rubber industry. The most well-known members of this polymer family are silicones.
- Rubber is another well-known isoprene polymer. It is used to seal wood, seal kitchen work surfaces, waterproof it, and so on.
- Melmac is used to make unbreakable plastic crockeries such as plates and cups.
- Terrycot and Terrywool are produced by combining polyester and cotton, respectively
Frequently Asked Question-FAQs
Q1. What is the difference between condensation polymerisation and addition polymerisation?
Answer: In addition polymerisation, molecules of the same or different monomers add on to one another, resulting in the formation of a macromolecule. The repeating unit of the polymer has the same molecular formula as the parent monomers. For examples, polyethene, polyacrylonitrile (PAN), buna-S, buna-N, and so on. Whereas in condensation polymerisation, monomers combine with the loss of simple molecules such as water, carbon dioxide, ammonia, and so on to form a macromolecule. The repeating unit of the polymer has a different molecular formula than the parent monomers.
Dacron, Nylon-6, Nylon-6,6 etc are some examples of condensation polymers.
Q2. What is step growth polymerisation?
Answer: Step-growth polymerisation is a polymerization method in which bi-functional or multifunctional monomers react to produce dimers, trimers, longer oligomers, and finally long-chain polymers in stepwise manner. Many are found naturally and some synthetic polymers, such as polyesters, polyamides, and polyurethanes, are generated by step-growth polymerisation.
Q3. Why bakelite is also classified as thermosetting plastics?
Answer: Thermosetting polymers are low molecular weight polymers that, when heated in a mold cavity, change chemical composition to form a hard, infusible, and insoluble mass. The hardening is caused by extensive cross-linking of the polymers. Bakelite is an example of a thermosetting polymer because when bakelite is formed from the cross-linking of phenol and formaldehyde and once the polymer is formed it becomes hard and cannot be remolded.
Q4. What is the difference between thermosetting plastics and thermoplastics?
Answer: These are low molecular weight polymers that, when heated in a mold cavity, change the chemical composition to form a hard, infusible, and insoluble mass. The hardening is caused by extensive cross-linking of the polymers.
Intermolecular forces exist between elastomers and fibers in thermoplastics. These are linear polymers that are hard at room temperature but soften and become viscous when heated. They become rigid again after cooling. The heating and cooling processes can be repeated as many times as necessary without affecting the chemical composition or mechanical properties.


