-
Call Now
1800-102-2727
Colloids: Definition, Classification, Preparation, Purification, Applications, Practice Problems & FAQs
Let’s perform an experiment. Take two glasses, one filled with milk and the other with sugar solution. Pass a light through each of them while they are both in the dark. What standouts to you?
A beam of light is scattered when it passes through a glass of milk. The Tyndall effect is a phenomenon which is responsible for this light scattering.

Only colloids display this phenomenon. In contrast to the sugar solution, which is a homogeneous solution, milk is a heterogeneous mixture of fat and protein.
In this article, we will understand the Definition, Classification, Preparation, Purification & Applications of Colloids.
Table of Content
- Definition of Colloids
- Classification of Colloids
- Preparation of Colloid Solutions
- Purification of Colloids
- Examples of Colloids
- Applications of Colloids
- Practice Problems
- Frequently Asked Questions - FAQs
Definition of Colloids
Any substance that contains particles that are significantly larger than atoms or regular molecules but too small to be seen without a microscope is referred to as a colloid. More broadly, this term refers to any substance, including thin films and fibres, that has at least one dimension in this broad size range, ranging in size from 1 nm to 1000 nm.
The nature of colloids is heterogeneous. A mixture must have suspended particles in order to be categorised as a colloid. The Tyndall Effect, a phenomenon where light beams incident on colloids are scattered as a result of the interactions between the light and the colloidal particles, is known to occur in colloidal solutions.
In a colloidal mixture, the components are typically evenly distributed. The component that is dispersed in these mixtures is referred to as the dispersed phase, while the component through which it is dispersed is referred to as the continuous phase/dispersing medium.
Classification of Colloids
Classification of colloids is done on the basis of three important factors: Physical state, Dispersion medium, Interparticle interactions and properties of the sol particle.
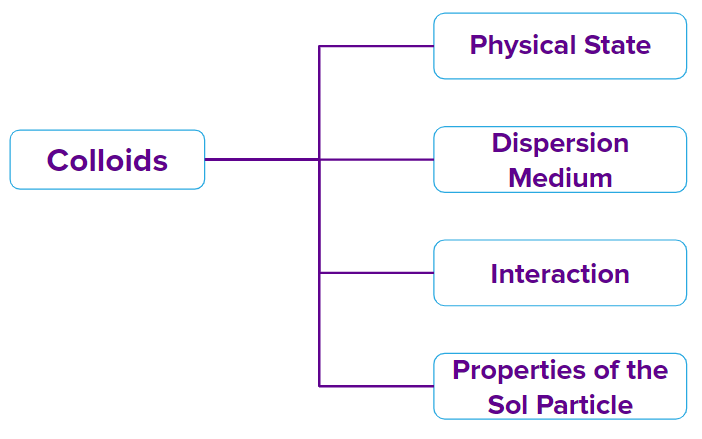
Classification of the colloids on the basis of their physical states:
|
Dispersion Phase |
Dispersion Medium |
Name of Colloidal System |
Examples |
|
Liquid |
Gas |
Liquid Aerosol |
Mist, clouds, fogs |
|
Solid |
Gas |
Aerosol |
Dust, smoke |
|
Gas |
Liquid |
Foam |
Suds, whipped cream |
|
Liquid |
Liquid |
Emulsion |
Cream, milk mayo |
|
Solid |
Liquid |
Sol |
Paints, jellies, sewage |
|
Gas |
Solid |
Solid foam |
Marshmallow |
|
Liquid |
Solid |
Solid emulsion |
Butter, cheese |
|
Solid |
Solid |
Solid sol |
Opals, some alloys |
Classification on the basis of dispersion medium sol are classified as:
Hydrosol:
As the name suggests, water is the dispersion medium for this type of colloids.
Example: Starch
Alcosol:
Alcohol is the dispersion medium for alcosol colloids.
In this type, alcohol acts as a dispersion medium.
Example: Colloidal solution of cellulose nitrate in C2H5OH.
Aerosol:
A colloidal system in which some gas, often air, serves as the dispersion medium and the dispersed phase is made up of either liquid or solid particles.
Example: Smoke, Fog
Classification on the basis of interaction forces between the dispersion medium and dispersed phase:
|
Parameter |
Lyophilic |
Lyophobic |
|
Definition |
Lyophilic colloids are solvent-loving colloids. As they can interact with the solvent. |
Lyophobic colloids are solvent hating colloids. As they can not interact with the solvent. |
|
Surface tension |
The surface tension of lyophilic colloids is usually lesser than that of their respective medium. |
The surface tension of lyophobic colloids is usually the same as of their respective medium. |
|
Viscosity |
The viscosity of lyophilic colloids is usually higher than that of their respective medium. |
The viscosity of lyophobic colloids is usually the same as of their respective medium. |
|
Reversibility |
These sols can be reversed. The same kind of sol is created when the dispersion medium is evaporated and the solvent is added once more. |
When the dispersion medium is evaporated once and the solvent is added a second time, no new sol is formed. Hence, these colloids are irreversible. |
|
Stability |
Stronger interaction between the dispersed phase and the dispersion medium makes the system more stable. |
Lyophobic sols are unstable because there is little interaction between the dispersed phase and the dispersion medium. |
|
Visibility |
An electron microscope clearly shows the particles. |
Ultra Microscopes can be used to see the particles. |
|
Addition of electrolyte |
The formation of sol requires a small amount of electrolyte. |
Coagulation happens when the electrolyte is added to the sol. |
|
Hydration |
Significant hydration occurs. |
There is no hydration of the sol. |
|
Examples |
Include starch, gum, and gelatin. |
Include metallic sols like gold and silver. |
Classification based on the properties of the sol particle:
|
Multi Molecular Colloids |
Macromolecular Colloids |
Associate Colloids |
|
Formed when numerous atoms or molecules come together. |
Formed by large size molecules. |
Formed by aggregation of a large number of ions in a concentrated solution. |
|
Lyophilic in nature |
Lyophilic in nature. |
Both lyophilic and lyophobic in nature. |
|
Molecular mass is intermediate |
High molecular mass |
High molecular mass |
|
Van der Waals forces, a type of physical force, hold the particles in these colloids together. |
Held by stronger van der Waal's forces due to the long chain. |
Van Der Waals force increases with an increase in the concentration. |
Preparation of Colloid Solutions
Stable colloids is also known as lyophilic sols, in which strong forces of interaction exist between the dispersed phase and the dispersion medium. These are made using the following techniques.:
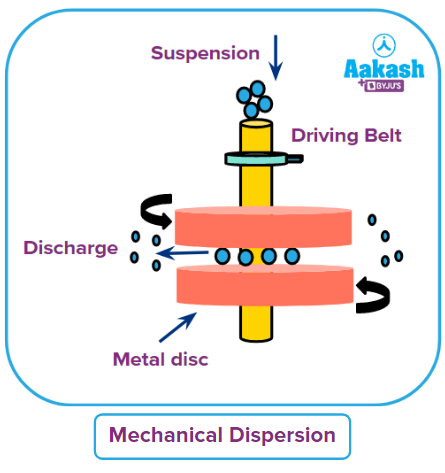
Mechanical dispersion:
This method is used to initially grind the material to gritty particles. The dispersion medium is then combined with it to create a suspension. The suspension is subsequently processed using a colloidal mill. It is made up of two fast-spinning metallic discs that are almost touching and revolving in opposing directions.
The mill's disc spacing has been tuned in such a way that the coarse suspension undergoes intense shearing, producing particles of colloidal size. This technique can be used to create colloidal solutions of black ink, paint, varnish, dyes, etc.
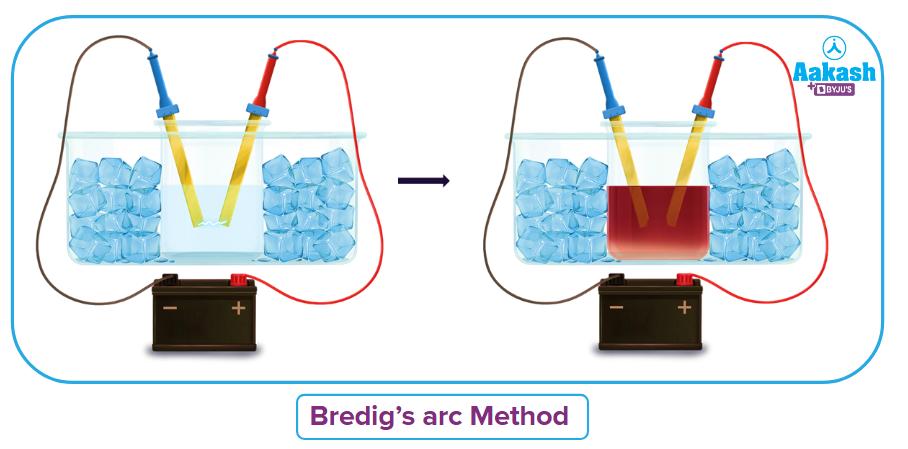
Using the Bredig's arc method:
Sols of metals such as Pt, Ag, Au and Cu are created using this technique.
Bredig's arc method involves striking an electric arc between metal electrodes submerged in water that contains a stabilising chemical. By enclosing the dispersion medium with a freezing mixture, the temperature is maintained. Electric arc is generated between the electrodes. The metals are vaporized by the arc's intense heat and then instantly condense in the liquid to form a colloidal solution. The prepared colloidal solution is stabilized by a little addition of KOH to it.
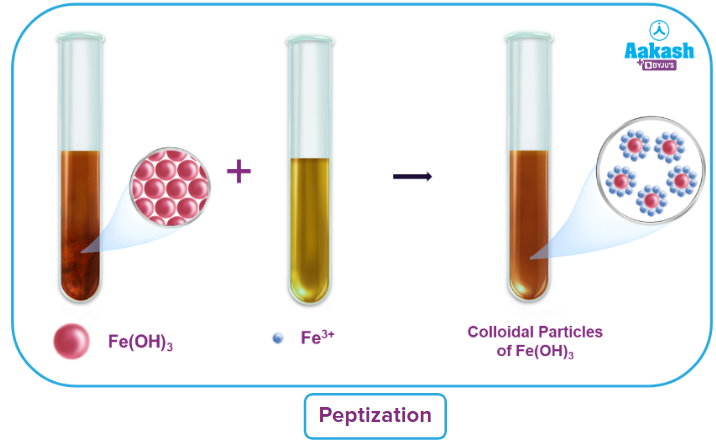
Through peptization:
Peptization refers to the process of converting precipitate into colloidal solution by incorporating an appropriate electrolyte.
Peptizing agents are the electrolytes employed in this process.
The appropriate ions from the additional electrolyte are absorbed by the precipitate particles when an electrolyte is added to a freshly prepared precipitate. A colloidal solution is created when the charged particles repel one another.
As an illustration, a small amount of FeCl3 solution added to a precipitate of iron (III) hydroxide results in the formation of a colloidal solution that is reddish brown in colour.
Fe(OH)3 with Fe3+ (from FeCl3) results in Fe(OH)3Fe3+.
A colloidal solution of silver chloride can be created by peptising a precipitate of silver chloride with a diluted solution of silver nitrate.
AgCl with Ag+(from AgNO3) results in the formation of a colloidal solution of AgCl Ag+.
Condensation method:
Small solute particles are condensed to create a dispersed phase particle using this technique. These methods are used to create the gold or colloidal silica.
Chemical methods:
There are several chemical methods by which colloidal solutions can be prepared.
By oxidation:
Colloidal sulphur can be obtained by passing oxygen gas through a solution of hydrogen sulphide. In this method, any oxidising agent like HNO3, H2SO4 can also be used.
(Sulphur sol)
By double decomposition:
This technique produces an arsenic sulphide solution. This procedure involves passing hydrogen sulphide through a cold water solution containing arsenic oxide.
Arsenic sulphide (sol)
By reduction:
A number of metals such as gold, silver, and platinum are obtained in a colloidal state by reacting the aqueous solution of the salts of these metals with the suitable reducing agents such as hydrogen peroxide, phenylhydrazine, stannous chloride, formaldehyde, etc.
(gold sol)
(gold sol)
The purple of Cassius is the name given to the gold sol created in the reduction of gold chloride solution.
By hydrolysis:
Boiling a diluted solution of their salts causes many salt solutions to hydrolyze quickly. For instance, boiling solutions of the equivalent chloride yields ferric and aluminium hydroxide sols.
colloidal sol
Solvent hydrolysis of sodium silicate yields silicic acid.
By excessive cooling:
In the process, an ice colloidal sol is produced. Ice is consumed with an organic solvent like ether or chloroform. By freezing a water solution in the solvent, ice can be made. The water molecules have separated from the solution and have combined to form particles of colloidal size.
By exchange of solvent:
This method allows for the manufacture of colloidal sol of some compounds, such as sulphur and phosphorus, which are soluble in alcohol but insoluble in water. A sulfur colloidal solution is produced when enough sulfate-containing alcoholic solution is poured into water.
By change of physical state:
Solutions of substances like mercury and sulphur are prepared by passing the vapours of mercury & sulphur through cold water containing a suitable stabiliser such as ammonium salt or citrate.
Purification of Colloids
Colloids contains ionic impurities and other categories of impure substances that decrease the quality of colloids used in various applications. Following are the methods to purify the colloids:
Dialysis:
It is based on the principle of diffusion. In the process of dialysis, dissolved substances are removed from a colloidal solution through a suitable semi-permeable membrane. Only those membranes can be used that allow a true solution, containing ions or smaller molecules to pass through it and not the colloidal particles. Therefore, parchment paper, animal membrane, or cellophane sheets are employed for this purpose.
Dialyser is the name of the device used to separate the colloidal solution. The colloidal solution is suspended in a bag of an appropriate membrane in a container with continuously flowing freshwater. The pure colloidal solution is left behind as the molecules and ions diffuse through the membrane (selectively permeable membrane) and into the outside water.
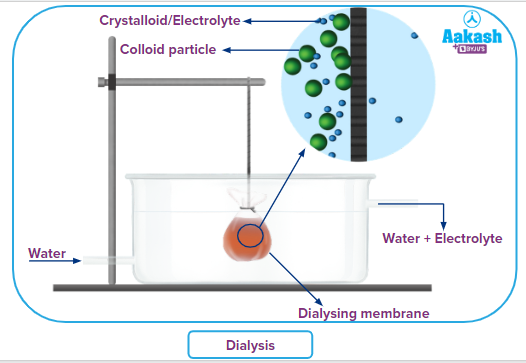
Applications: Dialysis is the most commonly used to remove undesired tiny substances like salts, reducing agents, or dyes from bigger macromolecules like proteins, DNA, or polysaccharides.
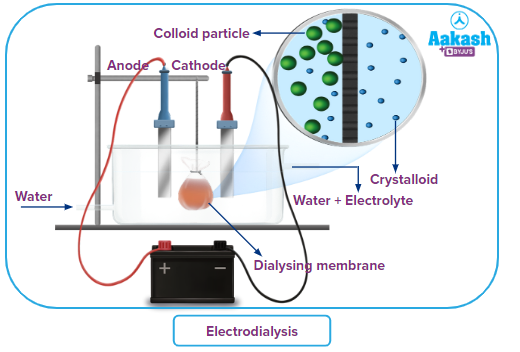
Electrodialysis:
It is founded on the idea that when an electric field is passed through an electrolytic solution, the ions migrate in the direction of the electrodes that have an opposing charge. If the contaminants in the colloidal solution only comprise an electrolyte, electrodialysis is utilised. When compared to straightforward dialysis, this modification speeds up the procedure. While pure water is brought outdoors, the colloidal solution is placed in a bag made of the appropriate membrane. The chamber is equipped with electrodes. Because of their bigger size, colloidal particles stay inside the solution while the ions in the solution migrate to the electrodes that are negatively charged and penetrate the membrane.
Ultrafiltration:
Ultrafiltration is the technique of using specially designed filters to separate colloidal particles from solvent and soluble solutes. Except for the colloidal particles, all substances should pass through the filter utilised for this purpose. Therefore, Ultra-filter paper is employed in this scenario.
For concentrating and purifying medium to high molecular weight components, such as plant and dairy proteins, polysaccharides, and enzymes, ultrafiltration is a selective separation technique. It is utilised to create potable water.
Smaller lactose, water, mineral, and vitamin molecules pass through the barrier, but larger protein and fat molecules—which are crucial for making cheese—are kept and concentrated. Polysaccharides, proteins, and colloidal impurities are eliminated using this method in the refinement of sugar.
Ultra-filter paper is prepared by soaking the filter paper in a collodion solution. Collodion is a 4% solution of nitrocellulose in a mixture of alcohol and ether. After that, formaldehyde makes it rigid, and it is dried.

Ultracentrifugation:
It is based on the principle of sedimentation, according to which denser particles settle down faster than less dense ones due to gravity.
The sample is made to rotate extremely quickly while using the ultracentrifugation method. As a result, larger and denser particles move faster and settle down in the form of pellets at the bottom of the tube. On the other hand, the smaller and less dense particles move slower and remain floating on the surface.
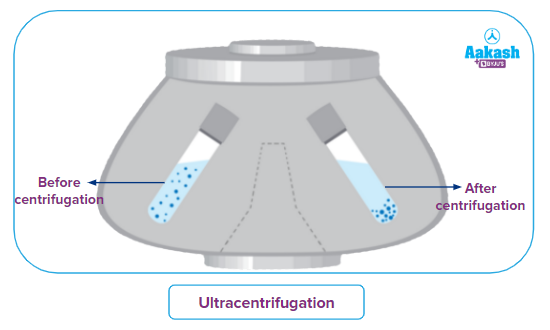
Applications: used to determine the characteristics of biomolecules like nucleic acids and proteins and to quantitatively analyse macromolecules in solution.
Examples of Colloids
Blood:
a pigment for respiration that contains albumin protein in water. Albumin, which serves as the dispersed phase in the pigment component, is present. Water serves as the dispersion medium.
Cloud:
It includes water droplets as a dispersed phase and air as the dispersion medium.
Gold sol:
It is a type of metallic solution in which particles gold are dispersed over water.
Applications of Colloids
- Colloids are extensively used in commercial, home, and medicinal purposes.
- As food items: Syrup, Halwa, Soup belong to a colloidal type of system.
- Medicine: Argyrols, a brand of colloidal silver, works as an antiseptic for eye infections.
- In purification of air by Cottrell precipitator: This process involves coagulation of solution particles. An electrified chamber with a central electrical plate that is equipped with an opposite charge of a dent: a smoke particle when dust passes, the particles are coagulated, and pure air is passed via another outlet is used to pass through dust or smoke.
- Tanning of leather: Animal skins are very soft, when they are immersed in the solution of tannin which has the opposite charge of animal skin, particles are coagulated and the process of making skin firm is called leather tanning.
- Formation of delta: It involves the coagulation of clay particles of the river with an electrolyte of seawater.
- Naturally occurring macromolecules are starch, cellulose proteins, enzyme gelatin etc. Artificial macromolecular and synthetic polymers such as,plastics, polyester, nylon, polishers etc. They usually have lyophobic sols.
Recommended Video: NEET 2020 | Surface Chemistry - Colloids: Colloidal State | Aakash Institute
Practice Problems
Q1. Which of the following cannot be made into a colloidal solution using the Bredig's arc method?
a. Gold
b. Silver
c. Platinum
d. Iron
Answer: (D)
Solution: The Bredig's arc method is appropriate for creating colloidal solutions of metals such as gold, silver, platinum, and others. Under the surface of water containing some stabilising agent, such as a trace of KOH, an arc is created between the metal electrodes. However, Iron does not react with alkalis, which is why Bredig's arc method cannot be used to obtain it.
Q2. Which of the following reducing agents is not used to obtain a gold sol?
a. HCHO
b. SnCl4
c. SnCl2
d. Ph-NH-NH2
Answer: (B)
A gold metal can be obtained in a colloidal state by reacting the aqueous solution of its salts with the suitable reducing agents such as hydrogen peroxide (H2O2), phenylhydrazine (Ph-NH-NH2), stannous chloride (SnCl2), formaldehyde (HCHO) etc.
(gold sol)
SnCl4 can not be used a reducing agent to obtain gold sol.
Q3. A heavy milk cream is an example of:
a. Aerosol
b. Foam
c. Emulsion
d. Solid sol
Answer: (B)
Solution: A milk cream is an example of emulsion as liquid (dispersed phase) is suspended in the liquid (dispersion medium).
Q4. During an experiment, accidentally iodine got mixed up with a biological sample of proteins and polys. The smallest colloid that can be produced during the separation process is:
a. Fats
b. Amino acids
c. Polys
d. None of the above
Answer: (B)
Solution: Given that the solution of iodine got mixed up with the milk sample, we know milk is already a colloidal solution of polymers like amino acids, proteins, fats, vitamins etc., amino acids are the smallest of all these colloidal particles.
Frequently Asked Questions - FAQs
Q1. How is the ultrafiltration method used in the production of potable water?
Answer: Ultrafiltration is used in water purification in which water is forced through an ultrafilter. Water and low-molecular-weight solutes filter through the membrane at one side, while suspended particles and high-molecular-weight solutes remain on the other side of the membrane. Most organic compounds and viruses, as well as a variety of salts, can be removed with ultrafiltration. It removes 90-100% of pathogens and does not require chemicals.
Q2. Why does motorbike exhaust appear blue at times?
Answer: A colloid's dispersed particles reflect light because they are larger in size. The Tyndall effect is the term used to describe how colloidal particles scatter visible light.The intensity of dispersed light is dependent on the density and frequency of the suspended particles. The Tyndall effect, in a similar manner as Rayleigh scattering, scatters blue light more than red light. This is why motorbike exhaust might seem blue at times.
Q3. How can we distinguish multimolecular and macromolecular colloids based on forces?
Answer: Multimolecular colloids have weak Van der Waals interactions between them, while macromolecular particles have strong chemical interactions between the particles.
Q4. What type of colloid is blood?
Answer: Blood is a colloidal suspension. It consists of lymphocytes, red blood cells, and white blood cells suspended in plasma.


