-
Call Now
1800-102-2727
Cleavage of O-H Bond: Esterification, Acylation, Reaction With Acid Anhydride, Grignard Reagent, Acidity of Alcohol and Phenol
Do you know about notorious alcohol?
It’s dangerous for health and it is nothing but cholesterol, which is also an alcohol and increased level of cholesterol causes heart attack.
Phenol is also used in bio-molecule research and extraction. In molecular biology, phenol is used to extract nucleic acids from tissue samples for further investigation.
In the cosmetic industry, phenol is used to make sunscreens, skin lightening creams, and hair coloring solutions.
Table of content:
- Esterification
- Acylation
- Reaction with acid anhydride
- Reaction with grignard reagent
- Acidity of alcohols
- Acidity of phenol
- Practice problems
- Frequently asked questions(FAQs)
Esterification:
The process of mixing a carboxylic acid (RCOOH) with an alcohol (ROH) in the presence of an acid to produce an ester (RCOOR) and water is known as esterification.
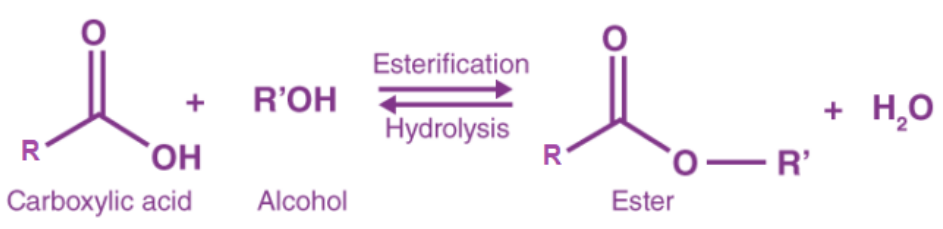
Mechanism:
This process involves five steps.
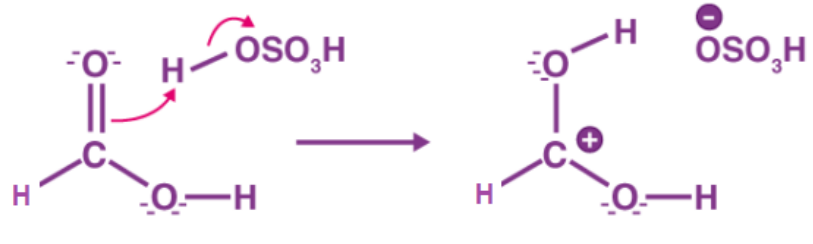
Step 1: Cation formation
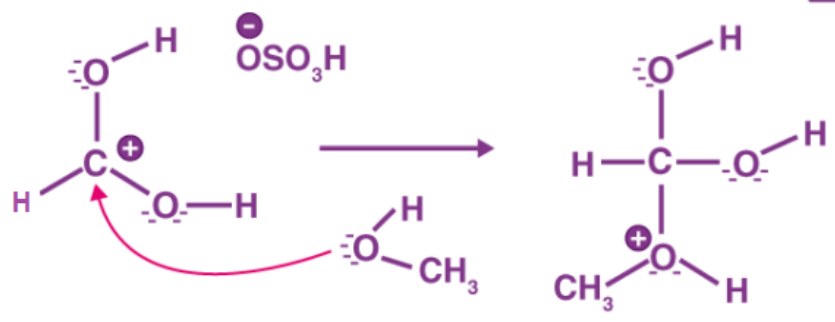
Step 2: To a carbocation, methanol can serve as a nucleophile.
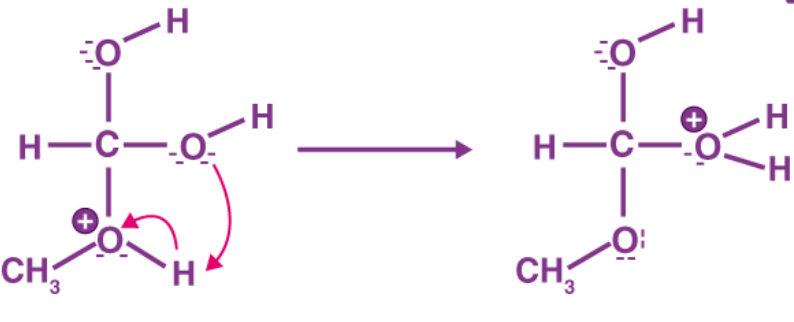
Step 3: To generate a good leaving group, a proton is transferred to one of the hydroxyl groups.
Step 4: The oxygen atom of one of the hydroxyl group provides a pair of electrons to a carbon atom, forming a bond by removing water. Because water has a lower concentration than methanol, so it can be released easily.
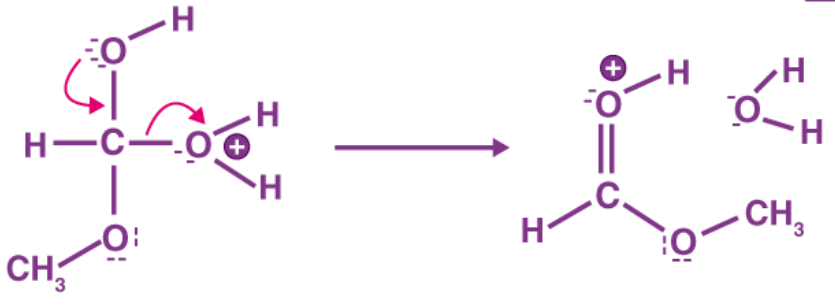
Step 5: Formation of ester
Acylation:
For alcohols and phenols, this approach will work. When it comes to phenols, the reaction can be aided by first converting the phenol to a more reactive state.For phenols, the reaction can be facilitated by first modifying it into a more reactive state.
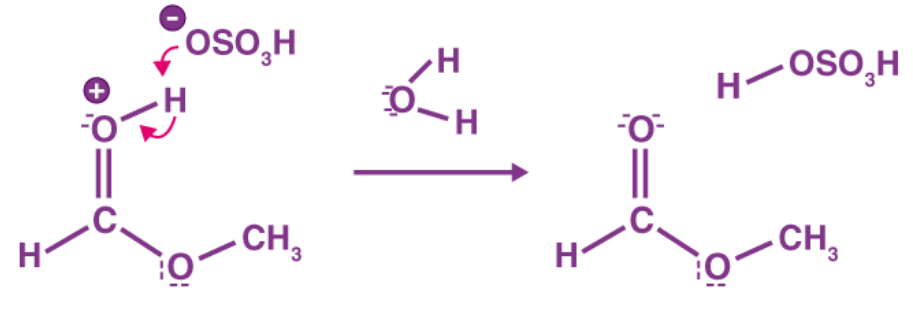
When an acyl chloride is added to an alcohol, a vigorous reaction occurs at room temperature, resulting in an ester and clouds of steaming acidic hydrogen chloride vapors. When liquid ethanoyl chloride is added to ethanol, a burst of hydrogen chloride is created along with the liquid ester ethyl ethanoate.
An ester and clouds of steaming, acidic hydrogen chloride vapour are produced when an acyl chloride is introduced to an alcohol at room temperature. A burst of hydrogen chloride and the liquid ester ethyl ethanoate are produced when liquid ethanoyl chloride is introduced to ethanol.

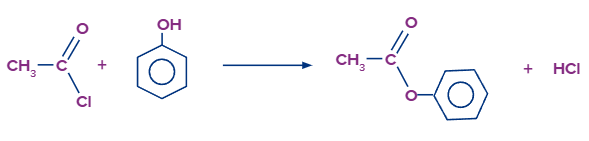
The substance commonly known as "phenol" is the most simplest member of the family of phenol. Phenol is made up of only a -OH group attached to a benzene ring. The reaction between ethanoyl chloride and phenol is similar to, but not as vigorous as the ethanol reaction. While not as intense as the ethanol reaction, the reaction between ethanoyl chloride and phenol is similar.Together with hydrogen chloride gas, phenyl ethanoate is formed.
Improving the reactions between phenols and some less reactive acyl chlorides:
Enhancing the reactions between some less reactive acyl chlorides and phenols:
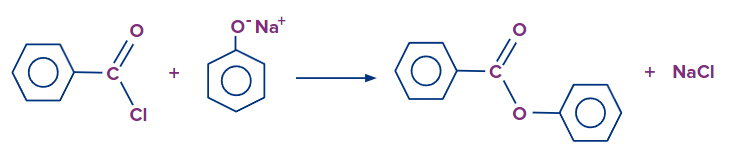
C6H5COCl is the formula for benzoyl chloride. The -COCl group is directly attached to a benzene ring. It is significantly less reactive because of the +M of the benzene ring than simple acyl chlorides such as ethanoyl chloride. By dissolving the phenol in sodium hydroxide solution, it is first converted into the ionic compound sodium phenoxide (sodium phenate). The phenol is first transformed into the ionic substance sodium phenoxide(sodium phenate) by dissolving it in sodium hydroxide solution.

The phenoxide ion reacts more quickly with benzoyl chloride than the original phenol, but it still needs to be shaken for about 15 minutes. Compared to the original phenol, the phenoxide ion reacts with benzoyl chloride more quickly, but it still requires around 15 minutes of shaking. Phenyl benzoate solid is formed.
Reaction with acid anhydride:
This reaction can again be used to produce esters from both alcohols and phenols. The reactions are slower than those with acyl chlorides, and the mixture must usually be warmed. In the case of a phenol, the phenol can be first treated with sodium hydroxide solution to produce the more reactive phenoxide ion.
Once more, esters can be made from phenols and alcohols via this procedure. The combination typically needs to be warmed up since the reactions are more slowly than they are with acyl chlorides. To create the more reactive phenoxide ion in the instance of a phenol, the phenol can first be treated with a sodium hydroxide solution.
As an example of a typical alcohol reaction, consider ethanol reacting with ethanoic anhydride: At room temperature, the reaction is slow (or faster on warming). There is no visible appreciable change in the colorless liquids, but a mixture of ethyl ethanoate and ethanoic acid is formed.
![]()
The reaction is faster if the phenol is first transformed to sodium phenoxide by adding sodium hydroxide solution. Phenyl ethanoate is generated again, but this time instead of ethanoic acid the sodium ethanoate is generated.

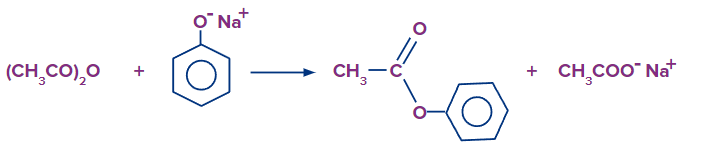
Reaction with grignard reagent:
Alkanes are formed when alcohols react with Grignard reagents. The alkane produced matches to the alkyl group of the Grignard reagents.
R - OH + R' - MgX → R' - H + Mg(OR)X
C2H5OH + CH3MgBr → CH4 + Mg(OC2H5)Br
Acidity of alcohol:
Alcohols, like water, are weak acids and bases. This is due to the polarization of the O–H bond, which makes the hydrogen partially positive. Furthermore, the electron pairs on the oxygen atom of alcohol make it both basic and nucleophilic. Protonation of an alcohol converts hydroxide, a poor leaving group, into water, a good leaving group. The two acid–base equilibria for ethanol are shown below.

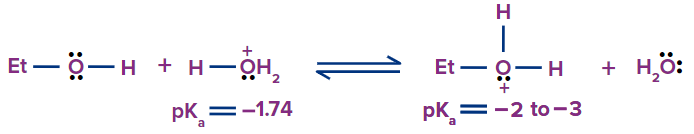
Alcohols are less acidic than water, The only alcohol that is slightly stronger than water (pKa = 15.7) is methanol (pKa = 15.5). Weaker acids include ethanol (pKa = 15.9), tert-butanol (pKa = 18.0), and others. All alcohols, on the other hand, are far stronger acids than terminal alkynes, hydrogen, ammonia, and alkanes.

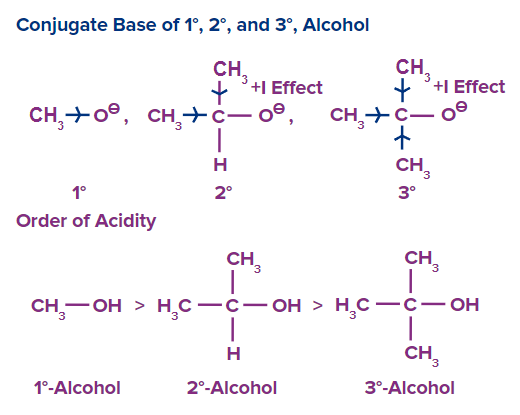
Order of acidity of alcohol:
The acidic strength of alcohol is determined by the strength of the conjugate base produced, i.e. alkoxide ions. It means the stronger the conjugate base of alcohol, the weaker will be the acid. It implies that the acid will be weaker the stronger the alcohol's conjugate base. As a result, a more stable alkoxide ion is a weaker conjugate base, and as a result, the alcohol will be more acidic because the stable alkoxide ion will easily release H+ ion. A weaker conjugate base results from a more stable alkoxide ion, and as a result, the alcohol will be more acidic because the more stable alkoxide ion will more readily release H+ ion.
Primary alcohols (1o) are more acidic than secondary alcohols (2o), which are more acidic than tertiary alcohols (3o), in the case of for simple alkyl alcohols.
We'll look at two factors to better comprehend and evaluate the alkoxide ion's stability:
Steric Factors : The alkoxide ion is bulkier with more substituted alkyl groups, making it more difficult for the solvent to stabilize the alkoxide ion. Since alkoxide ion will be less stable, it will be more reactive as a conjugate base with higher basic strength.Since the alkoxide ion has more substituted alkyl groups and is bulkier, the solvent has a harder time stabilising it. As a conjugate base with a higher basic strength, alkoxide ion will be more reactive due to its decreased stability. As a result, the acid produced by this conjugate base is weak.
Electronic Factors: As the number of Electron Donating Groups linked to a carbon with a hydroxyl group increases, the electron density on the O atom rises, making the alkoxide ion less stable and more reactive, making it a strong conjugate base. As a result, more substituted alcohols have a lower acidity.
Taking into account both electronic and steric factors, primary alkoxide ions are the most stable, whereas tertiary alkoxide ions are the least stable. As a result, primary alcohols are the most acidic in nature, whereas tertiary alcohols are the least acidic.
As a result, the decreasing order of alcohol acidity is:
Primary alcohol (1o) >Secondary alcohol (2o) >Tertiary alcohol (3o)
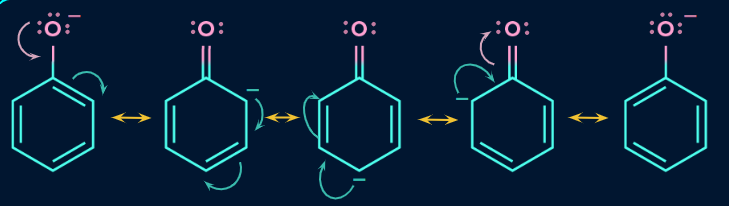
Acidity of phenol:
Alcohols and phenols are both very weak acids, however due to the stability of phenoxide ions, phenols are more acidic than alcohols. The delocalization of electrons in the benzene ring is the major cause for its stability. The negative charge on oxygen is delocalized within the benzene ring, making it extremely stable.
As the phenoxide ion is relatively persistent, phenols rapidly release protons in the presence of a base, making it more acidic. The following are the resonating structures of phenoxide ion:
Acidity of substituted phenol:
Effect of Substitution on acidity of phenol:
By forming stable phenoxide ions by delocalization of the negative charge and inductive effects -I/-M effect, phenol substituted with electron-withdrawing groups (EWG) such as Nitro and Chloro makes it more acidic. Due to the destabilization of the phenoxide ion due to the +I/+M effect, phenol substituted with electron-donating groups (EDG) or electron-releasing groups (ERG) such as methyl or methoxy group makes it less acidic. The order of decreasing acidity of para-substituted phenols with EWG and EDG attached is as follows:
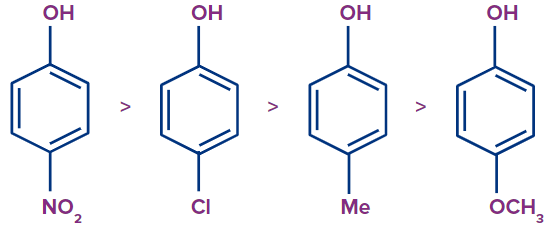
Effect of Substituent Position on Acidity of Phenol:
We can see that a negative charge is concentrated on the ortho and para positions in the resonating structure of phenoxide ion. As a result, it can be deduced that phenol's acidity is increased by electron withdrawing groups in the ortho and para positions. The following is a list of ortho, para, and meta substituted Nitro-phenol in decreasing order of acidity:
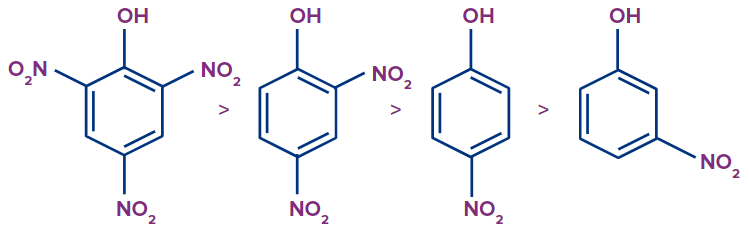
Practice problems:
Q.1. The breaking of ____ bonds occurs during the oxidation of alcohol.
(A) O-H
(B) C-H
(C) O-H and C-H
(D) Hydrogen
Answer: (C)
Solution: Oxidation of alcohols involves the cleavage of both O-H and C-H bonds so as to form a carbon-oxygen double bond. This results in the release of dihydrogen and as a result is also known as dehydrogenation reactions.
Q.2. How alcohols react when the bond between the O and H of the hydroxyl group is broken.
(A) nucleophiles
(B) electrophile
(C) protonated molecules
(D) electron seeking compounds
Answer: (A)
Solution: Depending on whether the O-H bond or the C-O bond is broken, alcohols can react as nucleophiles or electrophiles. In the nucleophile case, carbocations are formed, while in the electrophile case, protonated alcohols are formed.
Q.3. Which of the following compounds is the best proton acceptor?
(A) Ethanol
(B) Phenol
(C) p-Cresol
(D) p-Nitrophenol
Answer: (A)
Solution: Proton acceptor indicates the basic nature of the compound, which can be seen by the availability of the lone pair. Ethanol is the only alcohol among all options in which the lone pair is not delocalised and available for the donation. And it is the least acidic among all hence it is most basic.
Q.4. What role does concentrated H2SO4 play in the reaction of alcohol and carboxylic acid?
(A) Deprotonating agent
(B) Dehydrating agent
(C) Reducing agent
(D) Nucleophile
Answer: (B)
Solution: Concentrated H2SO4 is a protonating and dehydrating agent that acts as a catalyst in the esterification reaction. The reaction is reversible and is pushed forward by the removal of water as soon as it forms.
Frequently asked questions(FAQs):
Q1. Why tertiary alcohols are least reactive in esterification reaction?
Answer: The oxygen atom is to take up the electron pair from the bond in reactions involving O-H bond cleavage. The presence of an alkyl group with a +I (inductive) effect raises the electron density around the oxygen atom. As a result, bond cleavage in the O-H bond becomes difficult. Alternatively, the alkyl group reduces the polarity of the O-H bond, making H+ release is difficult. This has a negative impact on alcohol's reactivity. The greater the number of alkyl groups in the alcohol, the lower its reactivity. Hence tertiary alcohols are least reactive compared to secondary and primary alcohols in the esterification reaction.
Q2. Why is pyridine base used as a catalyst in the acetylation of alcohols?
Answer: Alcohols are reacted with acid chlorides and acid anhydride in the presence of a base, which neutralizes the acid formed during the reaction. This facilitates the forward shift in equilibrium of the reaction.
Q3. Why does phenol not react with carboxylic acids?
Answer: Carbonates do not react with phenol because it is such a mild acid. Phenol, like other alcohols, can form esters, but it does not react directly with carboxylic acids. Only an acid anhydride or an acyl chloride can react with phenol to form an ester.
The carboxylate ion produced is stabilized by resonance due to efficient delocalization of the negative charge. As a result, the carboxylate ion is more stable than phenoxide ions. Carboxylic acids, on the other hand, are more acidic than phenols.
The sodium bicarbonate test easily distinguishes between phenols and carboxylic acids. Because carboxylic acids are stronger acids than phenols, sodium bicarbonate is a weak base that quickly interacts with carboxylic acids but not with phenols.
Phenol is such a weak acid that carbonates do not react with it. While phenol can produce esters like other alcohols, it can not directly interact with carboxylic acids. To create an ester, phenol can only be combined with an acid anhydride or an acyl chloride.
Because the negative charge is effectively delocalized, the resulting carboxylate ion is stabilised by resonance. The carboxylate ion is hence more stable than phenoxide ions. Contrarily, carboxylic acids are more acidic than phenols.
The sodium bicarbonate test makes phenols and carboxylic acids distinct with ease. Sodium bicarbonate is a weak base that quickly reacts with carboxylic acids but not phenols since carboxylic acids are stronger acids than phenols.
Q4. Why are phenols less acidic than carboxylic acids?
Answer: Carboxylic acids are stronger acids than corresponding alcohols and even phenols because they lose their proton to form a stable conjugate base, i.e., carboxylate ion, which is more resonance stabilised than alkoxide or phenoxide ion. Along with stable carboxylate ions, carboxylic acid contains a carbonyl group, which is an electron-withdrawing group, making it more acidic than phenol, which is stabilised by resonating structures of phenoxide ions. In addition, in phenol, only carbon carries the negative charge, whereas in carboxylic acid, the negative charge is shared by two oxygen atoms.


