-
Call Now
1800-102-2727
Chirality - Introduction, R & S Nomenclature, Assigning R & S Configurations, Cahn-Ingold-Prelog Rules, Practice problems & FAQs
A class 12 student Meenal was studying a topic in chemistry and that topic was chirality. He was facing some difficulties while understanding the topic. The next day when he went to his school, he asked his chemistry teacher related to the topic of chirality.
His teacher explained it to him in a very simple way. He holds Meenal's hand and then superimposes his other hand upside down. They were non-superimposable in nature. Non-superimposable means they will not overlap with one another. No matter how the two hands are orientated, it is impossible for all of the major features of both hands to coincide across all axes, making the left hand a non-superimposable mirror image of the right hand.
Now Meenal feels a bit relaxed with his topic of chirality.

We will study about this topic of chirality in a bit more detail and will learn about the different configurations of molecules in chemistry.
TABLE OF CONTENT
- Definition of Chirality
- R & S nomenclature
- Assigning R & S configuration
- Cahn-Ingold-Prelog Rules
- Major examples with different cases
- Practice Problems
- Frequently asked questions-FAQs
Definition of Chirality:
The word chirality comes from the Greek word kheir, which means "hand." If an object's mirror image cannot be superimposed on the original thing, as our right and left hands cannot, the object is said to be chiral. What we mean by "non-superimposable" must now be a mystery to you. The object and its image are said to not be superimposable when the mirror image of the object is placed over the original object and they do not overlap.
Golf clubs, scissors, shoes, and a corkscrew are instances of "handedness" in chiral things. As a result, golf clubs and scissors are available in both right-handed and left-handed versions. Gloves and shoes, like everything else, come as a set of two, one for each hand. Chirality generally translates to "mirror-image, non-superimposable molecules," and chirality refers to the fact that a molecule's mirror image (which it must have) is not the same as it is.
R & S Nomenclature:
For attributing absolute configuration to chiral compounds, the R-S nomenclature is currently the recommended method. The letters R and S are derived from the Latin words "Rectus" and "Sinister," which mean "right" and "left," respectively. The molecules that rotate the plane of polarized light to the right are known as 'R-isomers,' whereas those that rotate the plane of polarized light to the left are known as 'S-isomers.'
Assigning R & S configurations:
To determine the absolute configuration, we must first identify the carbon(s) that are related to four separate groups (atoms). These are referred to as chirality centres (chiral centre, stereogenic centre).
For example, we have two carbons in our molecule, but out of these, the carbon with four distinct groups is chiral carbon. The carbon attached to bromine in this molecule to which we will assign the absolute configuration is the chiral centre of the molecule.
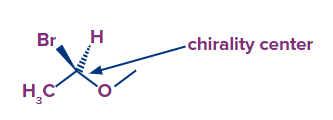
To do so, you must follow the Cahn-Ingold-Prelog system's procedures and guidelines.
Cahn-Ingold-Prelog Rules:
CIP rule lays down the rules for prioritizing atoms/ groups attached to a chiral centre. The atoms or groups are assigned in decreasing order of priority by following the following rules-
Step 1: The atom (directly attached to the chiral atom) with a higher atomic number will be prioritized.
- Based on the higher atomic number of the atom attached to the chiral centre we can decide this priority. The higher the atomic number, the higher the priority and assign say number 1 to it.
Example:
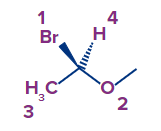
In the given compound, directly attached atoms to chiral carbon are bromine, oxygen, carbon(of methyl group) and hydrogen, whose atomic numbers are 35, 8, 6 and 1 respectively. As per the rule, the order of priority of the atom/group is bromine (1) first followed by oxygen (2), carbon (3) and of lowest priority is Hydrogen (4), the following priority is set based on the atomic numbers of the directly attached atom.
Step 2: In the case of isotopes, the heavier isotope will be assigned a higher priority.
- As in isotopes, atoms with higher atomic weight are given priority.
Step 3: If a group of atoms is attached and the first atom is common in two or more groups, then go for the second or third atom and so on till the first point of difference.
For example:
- Consider a chiral centre attached to H, methyl, aldehyde and carboxylic acid.
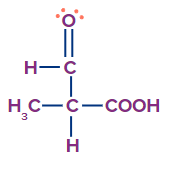
In the methyl group, there are three hydrogens, in aldehyde one hydrogen and one oxygen, while in carboxylic acid, two oxygens are next to carbon. So, the priority of the groups will be carboxylic acid, followed by aldehyde, methyl and lastly hydrogen.
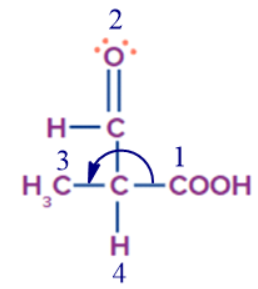
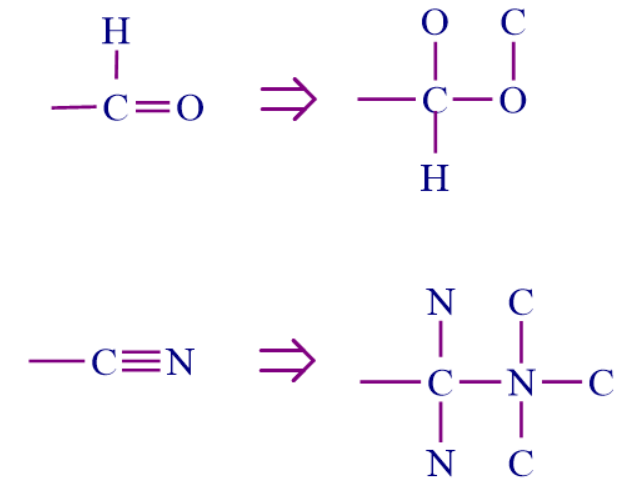
Step 4: Groups containing double or triple bonds between atoms are considered as if both atoms were duplicated or triplicated.
Example:
Step 5: Finding the direction of flow from high priority to low priority - right or left rotation, By keeping the lowest priority at the back. If the 4th (least priority) group is in a horizontal position, then reverse the derived configuration.
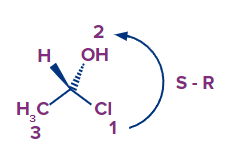
Here the least priority group is on wedge position, then we will reverse the absolute configuration, If it is R then will get to S and vice-versa.
Major examples with different cases:
Below are a few general examples of absolute configuration , let’s discuss them.
Example 1:
- Draw an arrow from priority one to priority two, and finally to priority three:
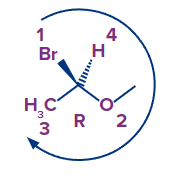
- The absolute configuration is R if the arrow goes clockwise, as it does in the above case.
- In contrast, if the arrow is turned counterclockwise, the absolute configuration is S.
Example 2:
- The priorities in the following molecule are Cl > N > C > H, and the arrow points counterclockwise, indicating a S absolute configuration:
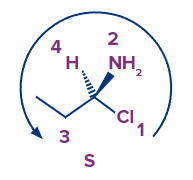
- So, remember: R is for clockwise, and S is for counterclockwise.
Example 3:
Let's look at what the enantiomer's absolute configuration might be:
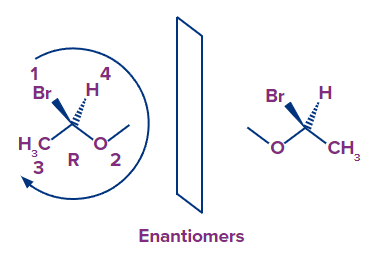
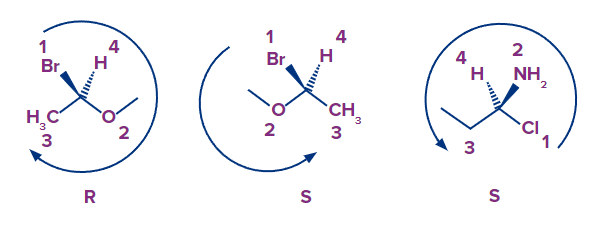
- Because all of the groups that surround carbon are the same, the priorities remain the same. We can see that the arrow goes counterclockwise this time in the mirror image, starting with bromine and moving on to oxygen and ultimately carbon. The absolute configuration is S if the arrow is turned counterclockwise.
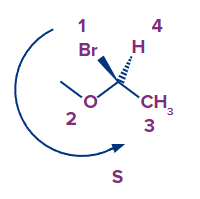
- It is important to note that enantiomers' chirality centres are all inverted (every R is S, every S is R in the enantiomer).
- The arrow's direction is unaffected by the lowest priority. However, it is critical, and it is a must, that when assigning the R and S configurations the priority with the lowest must face away from the spectator.To put it another way, the lowest priority must be a dashed line to assign the R and S based on the arrow's orientation, as we did earlie:
Example 4:
R & S configuration when the lowest priority is on the wedge.
- Molecule's absolute arrangement, where the hydrogen is a wedge line heading towards us.
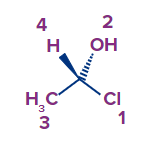
There are two methods through which we can decide the configuration
- Method 1: Turn the molecule 180 degrees so that the hydroxyl now faces you and the hydrogen faces away. This enables the molecule to be drawn as needed — with the lowest priority pointing backwards, as it should be for determining the R and S configuration:
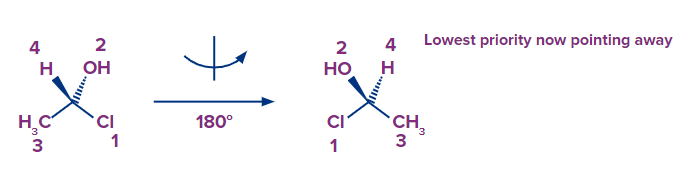
Next, assign the following priorities: chlorine is number one, oxygen is number two, carbon is number three, and H is number four.
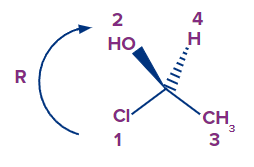
Because the arrow points clockwise, the absolute configuration is R.
The difficulty with this method is that it is impracticable to redraw the entire molecule and change every single chirality centre every time you work with a larger structure.
Consider biotin, which has all of these hydrogens facing forward. Redrawing this structure and replacing all of the hydrogens while keeping the rest of the molecule as it should be is not the greatest solution.
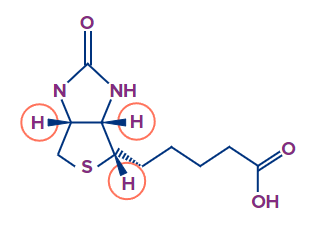
This is why we have the second strategy, which is the one that most people take.
- Method 2: You leave the molecule as is, with the hydrogen pointing in your direction. Continue by assigning priorities and drawing the arrow as you normally would.
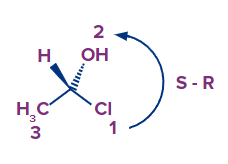
The only thing left to do is alter the outcome from R to S or S to R at the end.
The arrow goes counterclockwise in this scenario, but because the hydrogen is pointing in our direction, we modify the outcome from S to R.
Of course, because this is the same molecule drawn differently, either way should get the same result.
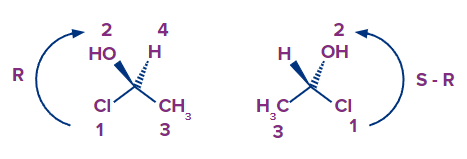
Example 5:
Absolute configuration when no wedge or dash group is involved.
There's a third option for group 4's location, which is when it's neither pointing away nor towards you. This implies we can't readily detect whether the lowest priority is pointing towards or away from us, and subsequently switch it at the end, as we could when group 4 was a wedge line.
Consider the below examples, what would this molecule's arrangement be?

For this, there is a simple yet quite effective approach that makes things much easier. Keep this in mind:
- Any two groups on a chiral centre can be swapped to reverse the absolute configuration (R to S, S to R):
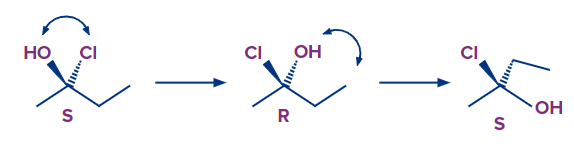
It's important to note that these are separate molecules. We're not talking about spinning about an axis or a single bond, where the absolute configuration(s) must remain constant. By changing the groups, we're essentially converting to a different molecule, making it easier to figure out the R and S configuration.

Because the group with the lowest priority is in the drawing plane, we can exchange it with the one pointing away from us (Br). We flip the outcome after calculating the R and S since swapping entails changing the absolute configuration and we need to switch back.
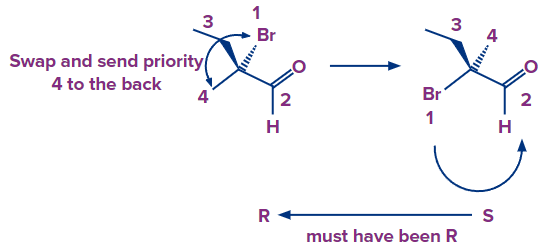
The arrow points counterclockwise, suggesting S configuration, whereas the original molecule had R configuration.
Example 6:
- Alternatively, draw the Newman projection of the molecule from the angle that places group 4 in the back (pointing away from the spectator) and apply the rule.
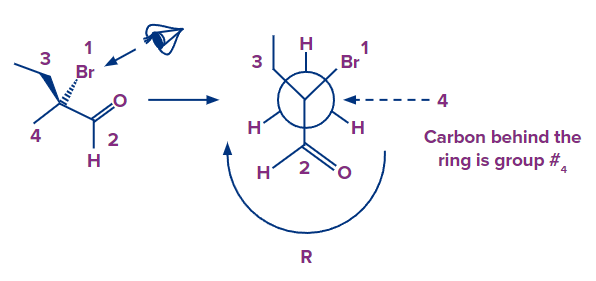
The lowest priority group is pointing, hence the arrow is pointing clockwise, indicating a R arrangement.
When dealing with stereochemistry in Newman projections, these two articles will come in handy:
Newman projections with R and S configuration
Bond-Line, Newman Projection, and Fischer Projection Conversion
Example 7:
R and S when Atoms (groups) are the same
When two or more atoms connecting to the chiral centre are the same, it is not always possible to assign priorities immediately.
Let's return to the 2-chlorobutane scenario, starting with the wedge chlorine:
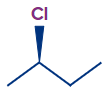
- The first priority is chlorine, followed by two carbons and a hydrogen, which has the lowest priority. We must decide the second priority by comparing two carbon atoms, and there is a tie because they have the same atomic number (clearly).
- So, what exactly do you do? Look at the atoms that are connected to the ones you're comparing
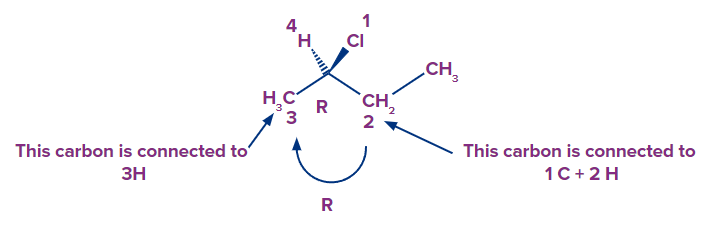
- The carbon on the left (CH3) is linked to three hydrogens, whereas the carbon on the right (CH2) is linked to two hydrogens and one carbon. The CH2 receives second priority as a result of the additional carbon, while the CH3 receives third priority. Because the arrow points clockwise, this is (R)-2-chlorobutane.
- If these atoms were also identical, we'd have to go away from the chiral centre and repeat the procedure until we reached the first point of difference.
- It's like comparing layers, the first layer is the atoms related to the chiral centre, and you only move on to the second layer if there's a tie.
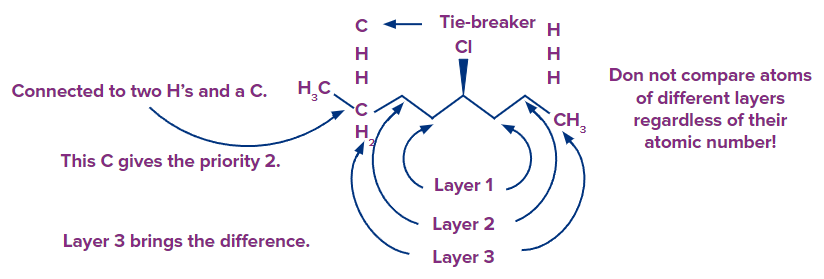
- Any atom in the second layer should never be compared to an atom in the first layer, regardless of its atomic number. Layer 1 is a tie in the following molecule, therefore we move on to layer 2, which gives priority to the carbon attached to the chiral centre on the left because it is connected to oxygen.
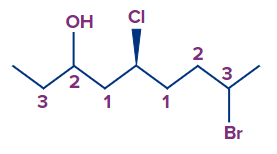
As a result, we don't compare layers 2 and 3, which would have given the carbon with a Br the upper hand because Br has a greater atomic number than oxygen. Because the oxygen is connected to a carbon atom that is closer to the chiral centre, it gives that carbon priority over whatever is connected to the carbon atoms on the next layer:
Example 8:
Double and triple bonds in the R and S configurations
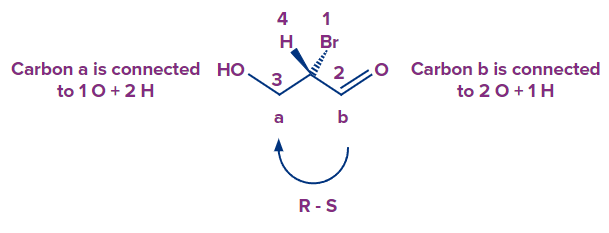
- Bromine is the most important element, followed by hydrogen. One oxygen and two hydrogens are bonded to carbon "a." One oxygen and one hydrogen are bonded to carbon "b." The double bond, on the other hand, causes carbon "b" to be considered as if it were attached to two oxygens. Any other double or triple bond follows the same rule. So, count a double bond as two single bonds, and a triple bond as three single bonds.
- Although the arrow points clockwise, the absolute configuration is S because the hydrogen is facing in our direction.
Practice problems:
Q1. For R, S-Configuration, which of the following is not a priority rule?
A. Priority is determined by the atomic number of the four atoms linked to the chiral centre, with the atom with the lowest atomic number receiving the lowest priority.
B. The atoms attached to each of the first atoms are compared if the two atoms attached to the chiral centre are the same.
C. When a double or triple bond exists, both atoms are regarded as duplicated or triplicated.
D. When the four atoms linked to the chiral centre are all distinct, priority is determined by atomic number, with the atom with the higher atomic number receiving lower precedence.
Answer: (D)
Solution: When the four atoms linked to the chiral centre are all distinct, priority is determined by atomic number, with the atom with the higher atomic number receiving higher precedence.
Q2. The arrangement is ___________ if we move our eyes counterclockwise from the ligand with the highest priority to the ligand with the lowest priority.
A. R-Configuration
B. S-Configuration
C. E-Configuration
D. C-Configuration
Answer: (B)
Solution: Our eyes go counterclockwise from the ligand with the highest priority to the ligand with the lowest priority in the S-Configuration.
Q3. According to the selection rules, the decreasing order of preference is __________
Answer: (A)
Solution: Higher atomic number atoms are given precedence. Therefore, the –NH2 group is given top attention then –C6H5 and so on.
Q4. Which of the following groups, according to the Cahn-Ingold-Prelog sequence rules, has the highest priority?
A. -CH2Cl
B. -CHO
C. -C2H5
D. -CH2OH
Answer: (A)
Solution: The Cahn-Ingold-Prelog sequence rules state that the group with the higher atomic number has higher priority.
The priority order is : -CH2Cl > -CHO > -CH2OH > -CH 3.
Frequently asked questions-FAQs:
Q.1. Enantiomers: are they superimposable?
Answer: One enantiomer will be the mirror image of the other enantiomer since enantiomers are stereoisomers that are non-superimposable mirror images.
Q.2. Can enantiomers be rotated?
Answer: The plane of polarization will rotate in opposite directions but with the exact same amount for enantiomers. A sample is deemed optically pure if there is just one enantiomer present. The effects of each enantiomer in a sample of enantiomers cancel out one another, molecule for molecule.
Q.3. Why is R and S configuration necessary?
Answer: An essential nomenclature system for designating enantiomers is the R/S system. According to a system by which each of its substituents is given a priority according to the Cahn–Ingold–Prelog priority rules (CIP), depending on atomic number, this method names each chiral centre R or S.
Q.4. Is the configuration of R and S the exact opposite (mirror image) of one another?
Answer: They are diastereomers because RS and SR are not mirror images of SS and cannot be superimposed on one another.


