-
Call Now
1800-102-2727
Carboxylic Acid Derivatives - Structure, Preparation, Physical and Chemical Properties, Practice problems and FAQ
What comes to your mind at the very first glimpse of the picture below?

Or to put it in another way, how would you decode this image in your own words? You would probably say, as perceived, that this is a colourful array of emojis portraying different and diverse expressions. Let me tell you an open secret now! There is something common in them all. That common link brings a state of parity that connects them all in one thread!
Can you guess what is the common thing?
For a while in your brain, remove the eyebrows and lips from these spherical faces. You can probably use your fingers to hide these two features of each face and check!
What can you perceive now?
If I am not wrong, probably a pair of oval eyes, that is exactly the same in all of them!
This is the common link among them all. One spherical human-faced emoji in this image, took multiple forms by changing expressions or colours! But what remained unchanged was the basic structure of its face- “The eyes’’ and also the most basic part- ‘The sphere’ of each face!
This is so strange but true! Even us, humans, are subjected to changes in our physical forms as we grow, in one lifetime. But ultimately, all our forms are derived from the basic person that we were, since inception!
So is the case with carboxylic acid derivatives. They are basically compounds derived from their principle group i.e., a carboxylic acid. Hence, they have some groups in common with the carboxylic acid besides their own unique features. Let us learn about carboxylic acid derivatives in detail.
TABLE OF CONTENTS
- Carboxylic Acid Derivatives - Introduction
- Structure and Nomenclature
- Reactivity of Acid Derivatives
- Nucleophilic Acyl Substitution of Carboxylic Acid Derivatives
- Chemical Reactions of Carboxylic Acid Derivatives
- Practice Problems
- Frequently Asked Questions - FAQ
Carboxylic Acid Derivatives - Introduction
Carboxylic acids are a class of organic compounds that include the functional group –COOH and have a wide range of applications, ranging from natural products to synthesised pharmaceuticals and hormones. Carboxylic acids have a hydroxyl group (-OH) attached to an acyl group (R-C=O), and functional derivatives are made by substituting the hydroxyl group with substituents like -NH2, -X, -C ≡ N,–OOCR .
Different carboxylic acid derivatives are formed by replacing the –OH group in the structure of carboxylic acid. Carboxylic acids have the general formula R-COOH, where R is an alkyl group. Typical –OH group replacements include the formation of the following acid derivatives:
- Acyl halides are formed when the –OH group is replaced with a halide group (-X).
- Amides are formed by replacing the –OH group with an amino group (-NH2).
- Esters are formed when the –OH group is replaced with a –OR group.
- Nitriles (R-C ≡ N) are also an acid derivative as they produce carboxylic acid on hydrolysis.
- Acid anhydrides and other compounds are formed when the –OH group is replaced with a –OOCR group.
We’ll discuss the preparation, properties and reactivity of the derivatives of carboxylic acids, also known as Class II carbonyl compounds (Class I carbonyl compounds are ‘Aldehydes and ketones’), as well as their involvement in nucleophilic acyl substitution processes in depth.
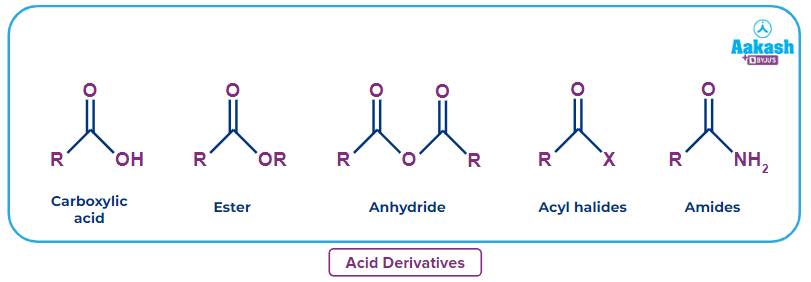
Structure and Nomenclature
Some examples of acid derivatives along with structures are given below.
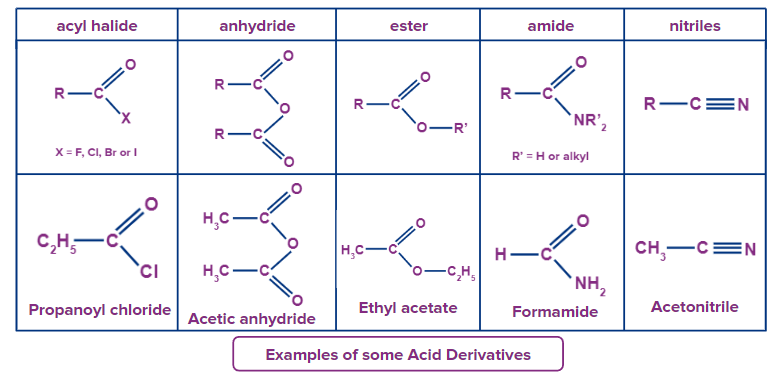
Esters
The alkyl group is called first, then the acyl group is given a derived name, and the ‘oic’ or 'ic’ suffix in the acid name is substituted with ‘ate’.
Example: CH3(CH2)2CO2C2H5 - Ethyl butanoate (or ethyl butyrate).
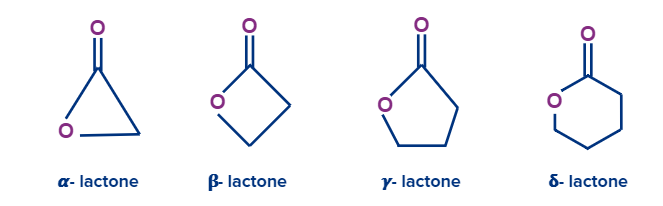
Lactones are the cyclic esters. The position of the alkyl oxygen in relation to the carboxyl carbonyl group is indicated by a Greek letter.
Acid Halides
The acyl group is named first, then the halogen is given its own name.
Example:CH3CH2COCl - Propanoyl chloride (or propionyl chloride).
Anhydrides
The name of the acid(s) in question is used first, followed by the phrase "anhydride."
Example: CH3(CH2)2CO2O - Butanoic anhydride, (CH3COOCOCH2CH3) - Ethanoic propanoic anhydride (or acetic propionic anhydride).
Amides
The related acid's name is used first, with the oic acid or ic acid suffix being replaced by amide (only for primary amides).
Example: CH3CONH2 - Ethanamide (or acetamide).
Alkyl substituents on the nitrogen atom are present in secondary and tertiary amides. These are identified by the term(s) "N-alkyl" at the start of the name.
Examples: CH3(CH2)2CONHC2H5 - N-ethylbutanamide, (CH3)2NCOH - N,N-dimethylmethanamide (or N,N-dimethylformamide).
Lactams are the cyclic amides. The position of the nitrogen on the alkyl chain in relation to the carboxyl carbonyl group is indicated by a Greek letter.

Simple acyclic nitriles are given their names by appending the suffix nitrile to the name of the relevant alkane (same number of carbon atoms). The nitrile carbon is the first in the chain to be numbered. Nitrile is commonly used to replace the oic acid or ic acid ending of the equivalent carboxylic acid. Carbonitrile is the name given to a nitrile substituent, such as one found on a ring.
Example: (CH3)2CHCH2CN - 3-methylbutanenitrile (or isovaleronitrile).
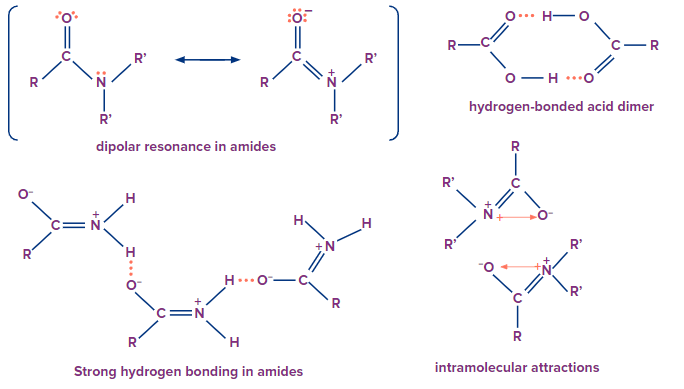
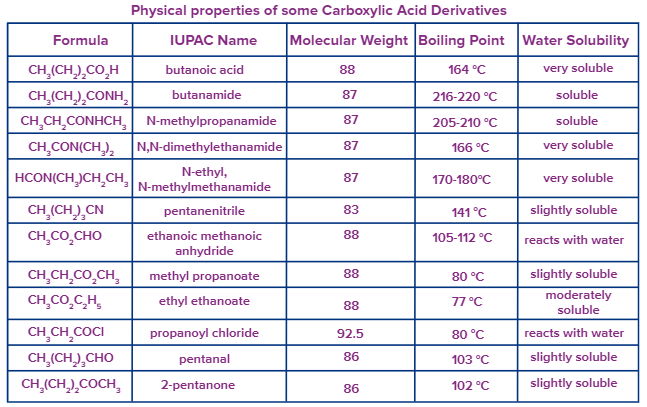
Physical Properties of Acid Derivatives
- For acid derivatives, as the dipolar interactions increase, their boiling points also increase.

- The strong dipolar forces inherent in nitriles, acids, and amides are responsible for their high boiling temperatures.
- The greater the extent of hydrogen bonding, the greater the solubility of a certain acid derivative.
Reactivity of Acid Derivatives
The synthesis of carboxylic acid derivatives is a two-step process, each of which is crucial in anticipating the compound's reactivity. When comparing the reactivities of carboxylic acids and their derivatives, electronic and steric variables are also relevant to consider.
The presence of bulky groups significantly affects the first step of the rate of reaction during the production of an acid derivative. The greater the bulkiness of the alkyl group attached to the carbonyl carbon, the harder it is for the nucleophile to attack the carbonyl carbon. Steric factors are an important consideration when assessing reactivity.
In forecasting reactivities, the polarisation of acyl molecules is also relevant. More polarised acyl compounds react faster than less polarised ones.
Example:
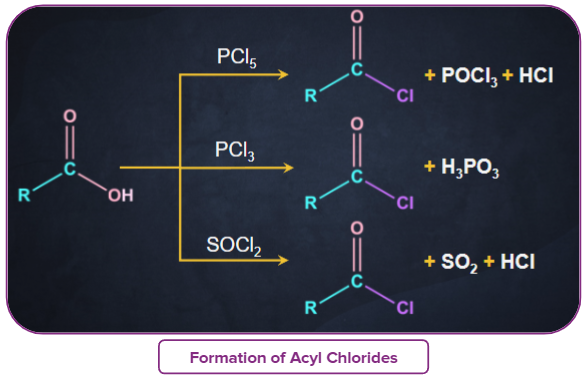
- Acyl chlorides are very reactive acid derivatives, therefore, special reagents like phosphorus halides (PCl5and PCl3) & SOCl2 are used to prepare them.
- Amides are produced when carboxylic acid reacts with ammonia.

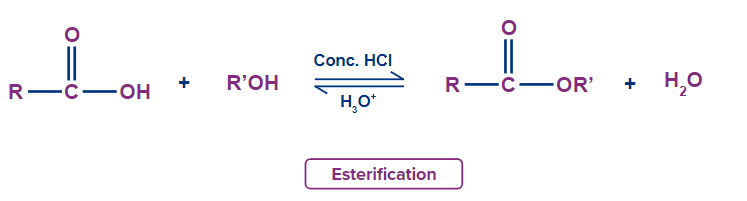
- Esters are produced by the process of esterification involving a carboxylic acid and an alcohol, in the presence of H+ ions.
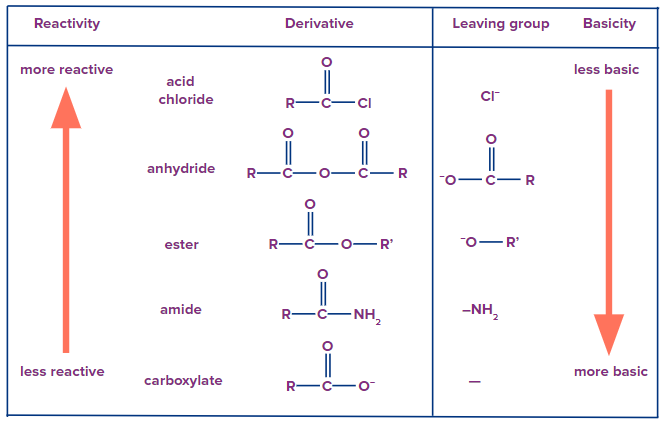
Acid derivatives undergo nucleophilic acyl substitution which is bimolecular. Weaker the base, better will it act as the leaving group, more reactive will be the acid derivative. The reactivity order of acid derivatives towards nucleophilic acyl substitution is given as follows.

Nucleophilic Acyl Substitution of Carboxylic Acid Derivatives
Nucleophilic acyl substitution usually takes place by an addition-elimination mechanism. The incoming nucleophile adds to the carbonyl to form a tetrasubstituted intermediate. A pair of nonbonding electrons on the oxygen reforms the π bond and either the leaving group (lg-) or the nucleophile (Nu-) is eliminated with its bonding electrons.
Hence, it depends on basicity. Weaker base is a better leaving group. Weaker base is preferentially eliminated.
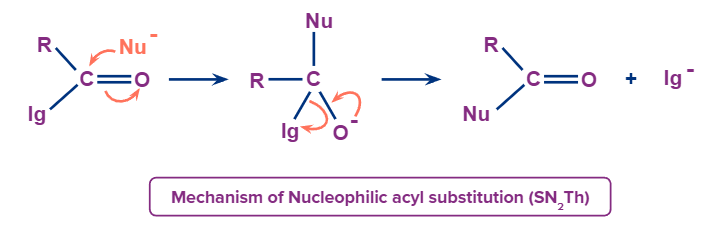

Following this transformation we can convert one acid derivative to another form of acid derivative. A more reactive intermediate can be converted to a less reactive intermediate.
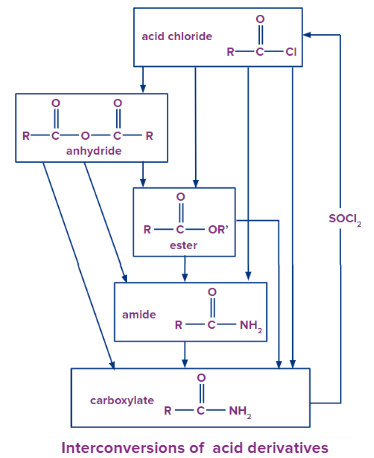
Chemical Reactions of Carboxylic Acid Derivatives
Formation of Acid Anhydride from Acyl Chloride
It is to be noted that an anhydride cannot be converted into an acyl chloride because chloride ion is a weaker base than a carboxylate ion.

Conversion of Acid Anhydride to Ester

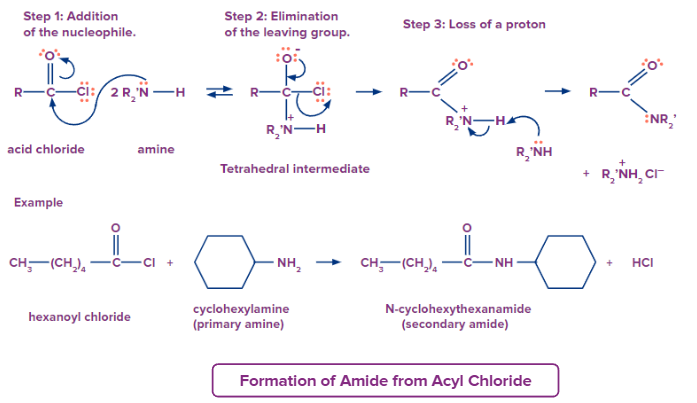
Formation of Amide from Acyl Chloride
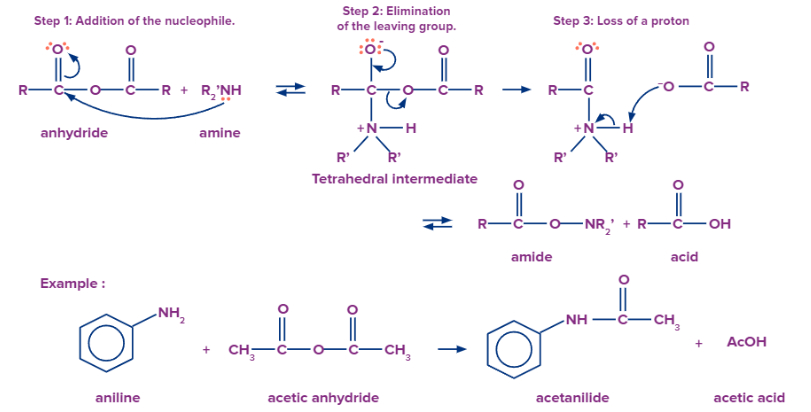
Formation of Amides from Acid Anhydrides
![]()
- Upon hydrolysis acid chlorides convert to carboxylic acids, with Cl- being the leaving group.
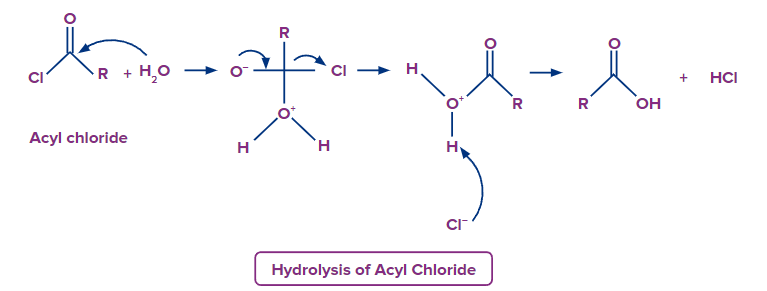
- Reductions of the acid derivatives usually produce either aldehydes or alcohols, i.e., functional groups having a lower oxidation state of the carboxyl carbon. Reduction occurs either through catalytic hydrogenation, diborane reduction or complex metal hydride reactions.
Example:

Practice Problems
Q. 1. Why do aldehydes and ketones not give nucleophilic acyl substitution?
Solution: Acyl substitutions will not be given with ketones or aldehydes. Aldehydes and ketones have R- or H- leaving groups, which are highly basic and thus, weak leaving groups. As a result, they are the least susceptible to acyl substitution.
Q. 2. Which among the following is the correct order portraying the relative reactivity of an acid derivative towards SN2Th?
a. Acyl chloride > Ester > Acid anhydride > Amide
b. Acyl chloride > Acid anhydride > Ester > Amide
c. Ester > Acyl chloride > Amide > Acid anhydride
d. Acid anhydride > Amide > Ester > Acyl chloride
Solution: The ease of nucleophilic substitution depends upon the nature of the leaving group. When the leaving tendency of a group in a compound is high, then the compound is more reactive, towards nucleophilic substitution. The reactivity of the compound may be explained on the basicity of leaving the group. A weaker base is a better leaving group. The order of basicity is
Cl- < RCOO- < RO- < NH2-
Hence, the order of leaving tendency is Cl- > RCOO- > RO- >NH2-. So order of reactivity will be highest for acyl chloride and lowest for amide.
So, option B) is the correct answer.
Q. 3. What happens when acetyl chloride undergoes reduction?
a. Aldehyde is obtained
b. Ketone is obtained
c. Anhydride of acid is obtained
d. No reaction
Solution: Acetyl chloride undergoes reduction in the presence of H2/Pd, BaSO4 to form acetaldehyde. This is also known as the Rosenmund Reaction.

So, option A) is the correct answer.
Q. 4. Which is a better leaving group -Clor -OH ?
Solution: -OH is strongly basic and hence is a weaker leaving group. -Cl on the other hand is bulkier and a weaker base; so a better leaving group.
Frequently Asked Questions - FAQ
1. What are the major four acid derivatives?
Answer: The four major types of acid derivatives obtained by the removal of -OH from the -COOH the group of carboxylic acids are acyl anhydrides, acid halides, amides and esters.
2. What is an acyl halide?
Answer: An acyl halide has the formula RCOCl where -R is an alkyl group. It is derived from carboxylic acid and has a halide ion attached in the place of -OH in RCOOH of the acid.
Example: Acetyl Chloride (CH3COCl)
3. Which is the most reactive acid derivative?
Answer: The substituent's ability to function as a leaving group determines the reactivity. RCOCl is the most reactive acid derivative because -Cl, being an electron withdrawing group, creates greater positive charge on -C=O and is the best leaving group. So, acyl halides are the most reactive.
4. Are nitriles (cyanides) considered acid derivatives?
Answer: Although nitriles do not contain a R-COO group, it is still sometimes referred to as acid derivatives. This is because on hydrolysis of nitriles, carboxylic acid can be obtained.


