-
Call Now
1800-102-2727
Biomolecules – Carbohydrates, Proteins, Nucleic Acids, Vitamins, Lipids, Practice Problems and FAQ
Do you know what is amazing about living organisms? Think about it!
The most amazing thing about living organisms is that they are composed of non-living atoms and molecules. A living organism grows, sustains and reproduces.
Have you ever wondered why and how these things happen? Also, how do they do it efficiently?
Living things are composed of numerous macromolecules, including lipids, proteins, carbohydrates, nucleic acids, and vitamins. We must include both proteins and carbohydrates in our diets. These biomolecules, which interact with one another to form the molecular logic of life processes, are the real food soldiers in the struggle for life's existence.
We will learn more about biomolecules, their classification, and their applications on this concept page.
TABLE OF CONTENTS
- Biomolecules – Introduction
- Biomolecules – Classification
- Carbohydrates
- Proteins
- Nucleic Acids
- Vitamins
- Lipids
- Practice Problems
- Frequently Asked Questions – FAQ
Biomolecules – Introduction
Biomolecules are common to living systems. They constitute carbohydrates, proteins, nucleic acids, vitamins and lipids. Some compounds are used for storage and transport like adenosine triphosphate (ATP), and some are polymers. Every biological organism's main building blocks are biomolecules. They work together to promote several biological processes that are essential for life. They come in a variety of shapes and sizes. Carbon, hydrogen, oxygen and nitrogen make up the majority of biomolecules.
Biomolecules – Classification
Biomolecules can be classified into:
- Carbohydrates
- Proteins
- Nucleic Acids
- Vitamins
- Lipids
We will learn about each of them in detail.
Carbohydrates
Plants are the main producers of carbohydrates. Early observations revealed that the broad word "carbohydrates" refers to a class of molecules that frequently have the formula They appeared to be carbon hydrates. For instance, the generic formula can be used to describe the molecular structure of glucose ().
However, not every substance that fits into this formula may be categorised as a carbohydrate. One substance that conforms to this general formula, , but is not a carbohydrate is acetic acid (CH3COOH). Although it is a carbohydrate, rhamnose, or , does not fit in this equation.
Carbohydrates are polyhydroxy aldehydes or ketones, or substances that create polyhydroxy aldehydes or ketones when they are hydrolysed. In layman's terms, we refer to carbohydrates as sugars or sweet-tasting things. Saccharides (Greek: sakcharon = sugar) are the collective name for them. Monosaccharides (1 unit), oligosaccharides (2-10 units), and polysaccharides are classified according to the number of constituent sugar units recovered after hydrolysis (more than 10 units).
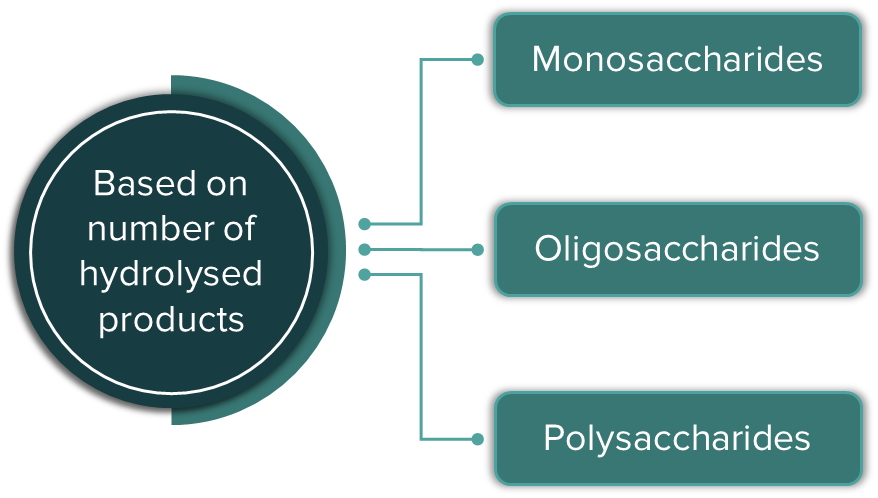
Monosaccharides: The simplest carbohydrates that cannot be hydrolysed into simpler carbohydrates.
Examples: Glucose, fructose, ribose, etc.

Oligosaccharides: Carbohydrates that hydrolyse to produce 2–10 molecules of monosaccharides are called oligosaccharides.
Examples: Sucrose, maltose, etc.

Oligosaccharides can be further classified into disaccharides, trisaccharides, tetrasaccharides, etc., based on the number of hydrolysed products formed.
Carbohydrates that undergo hydrolysis to produce only 2 molecules of monosaccharides are called disaccharides.
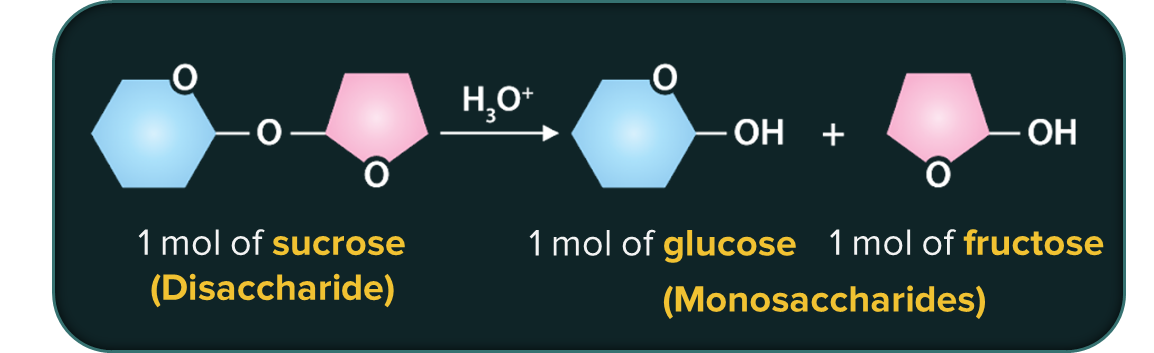
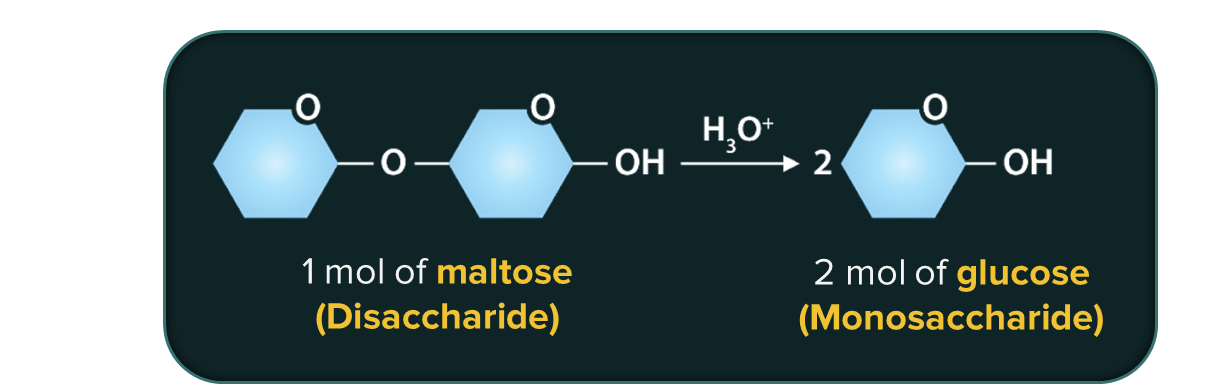
Carbohydrates that undergo hydrolysis to produce only 3 molecules of monosaccharides are called trisaccharides.
Polysaccharides: Carbohydrates that produce a large number of molecules of monosaccharides (>10) on hydrolysis are called polysaccharides.
Examples: Starch, glucose, cellulose, etc.
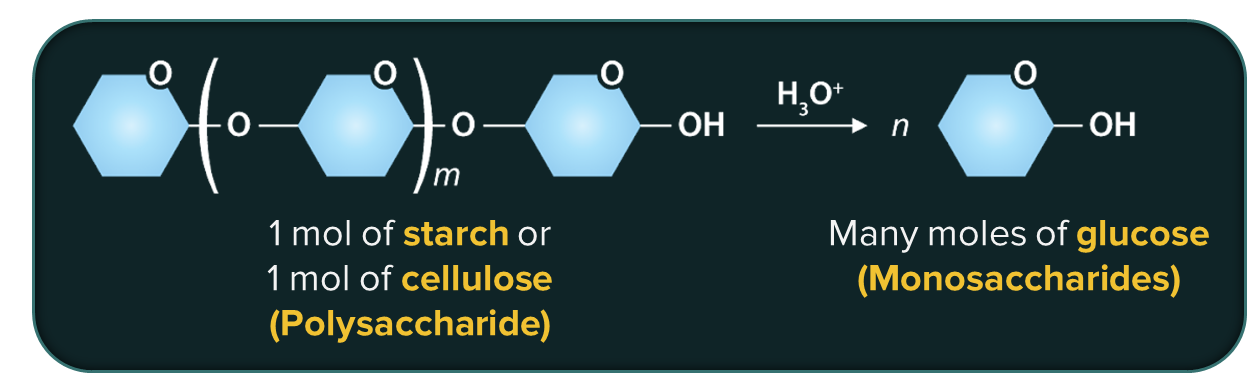

Carbohydrates serve a variety of purposes. They are the most abundant dietary source of energy; they are structurally vital for many living species as a primary structural component. For example, cellulose is an important structural fibre for plants.
Proteins
Each living cell is made up of thousands of different proteins. Proteins are another class of essential macromolecules. Proteins are polypeptide chains made up of amino acids organised in a specific order. In some circumstances, the structure of proteins is classed as primary, secondary, tertiary, or quaternary. These structures are based on the degree of complexity of a polypeptide chain's folding. Proteins have structural and dynamic functions. Myosin is a protein that permits muscles to contract and move. The majority of enzymes are proteinaceous.
Fibrous proteins and globular proteins are the two forms of protein molecules in general.
Fibrous Proteins: When polypeptide chains run parallel to each other and have a fibre-like structure, they are called fibrous proteins. In fibrous protein, chains are held together by hydrogen and disulphide bonds. These are insoluble in water.
Examples: Myosin, keratin, etc.
Globular Proteins: When a folded polypeptide chain forms a spheroidal shape, it is called a globular protein. Globular proteins are compact, soluble proteins. Examples: Insulin, albumin, etc.
The structure and shape of proteins can be studied at four different levels namely, primary, secondary, tertiary, and quaternary.
Primary structure: Each polypeptide in a protein has amino acids linked with each other in a specific
sequence known as the primary structure. Any change in this primary structure, i.e., sequence of amino acids, creates a different protein.
Secondary Structure: The shape in which a long polypeptide chain can exist is called secondary structure. Secondary structure can be either 𝛂-helix structure or 𝝱-pleated sheet structure.
Tertiary structure: It represents the overall folding of the polypeptide chains, i.e., further folding of the secondary structure, producing a 3D structure. R-groups determine the tertiary structure. The Quaternary Structure is made up of many tertiary structures folded together.
Quaternary structure: Some of the proteins are composed of two or more polypeptide chains referred to as subunits. The spatial arrangement of these subunits with respect to each other is known as quaternary structure.
Proteins are of high molecular mass and are complex biopolymers of amino acids. The amino acids contain amino as well as a carboxylic acid groups. There are around 20 amino acids in the living system.
Amino acids can be classified into three types based on the functional groups present.
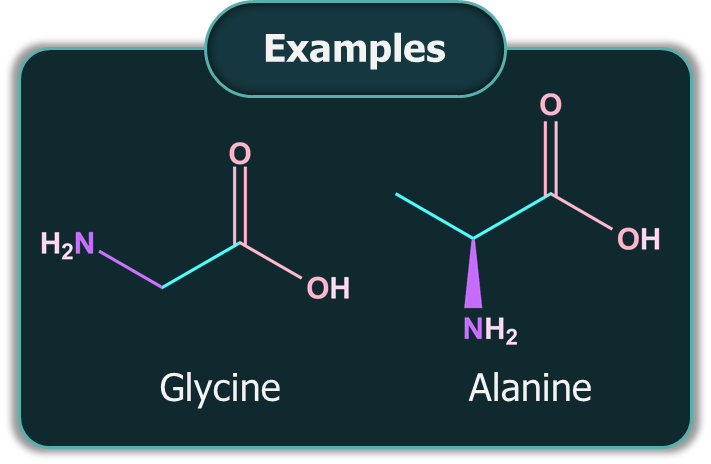
Neutral Amino Acids: If an equal number of and -COOH groups are present in an amino acid, it is a neutral amino acid.
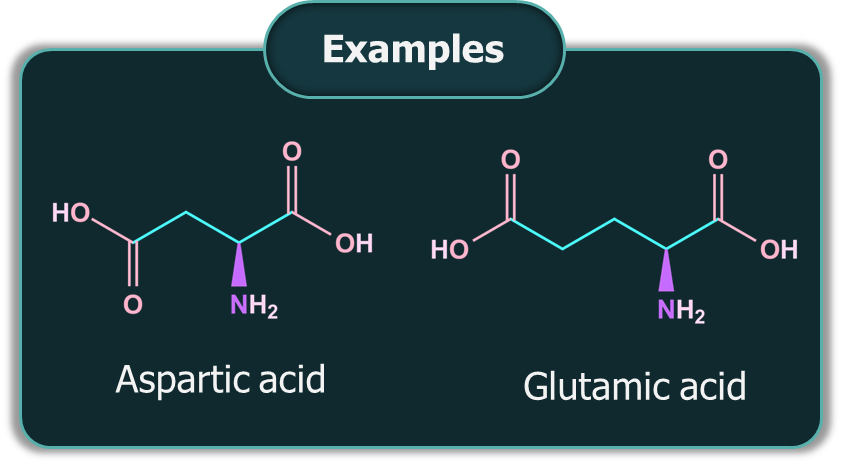
Acidic Amino Acids: If the number of -COOH groups present in an amino acid is greater than the number of groups, it is an acidic amino acid.
Basic Amino Acids: If the number of -NH2 groups present in an amino acid is greater than the number of -COOH groups, it is a basic amino acid.
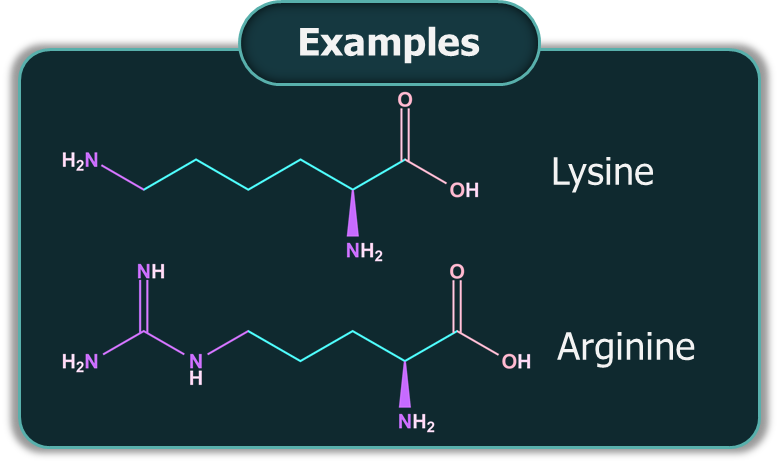
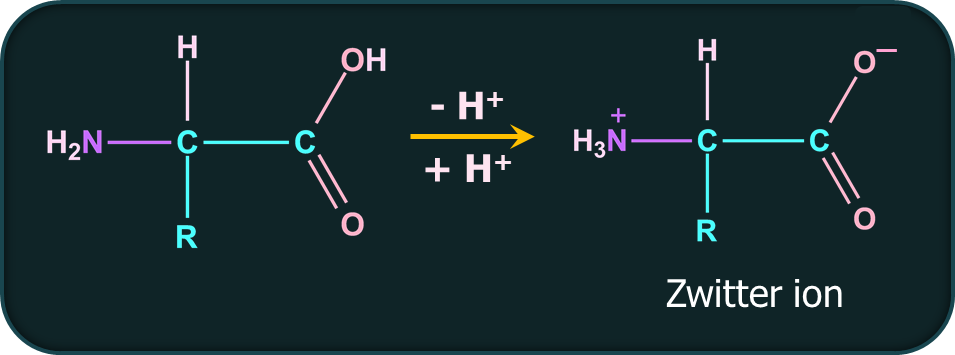
In a neutral amino acid solution, the -COOH loses a proton and the -NH2 of the same molecule picks up the proton. This form of amino acid is called the Zwitter ion form. The resulting ion is dipolar, charged, but overall electrically neutral. Amino acids are amphoteric in nature.
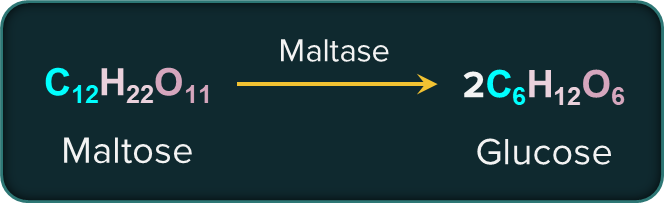
Enzymes: Reactions of cellular metabolism are mediated by remarkable biological catalysts known as enzymes. Almost all enzymes are globular proteins. Enzymes are very specific for a particular reaction and a substrate. The name of enzymes end with -ase.
For example, the enzyme that catalyses the hydrolysis of maltose into glucose is named as maltase.
Nucleic Acids
The nucleus of a living cell is responsible for the transmission of inherent characteristics, also called heredity. The particles in the nucleus of a cell which are responsible for heredity are known as chromosomes. They are made up of proteins and another type of biomolecule known as nucleic acids. There are two types of nucleic acids.
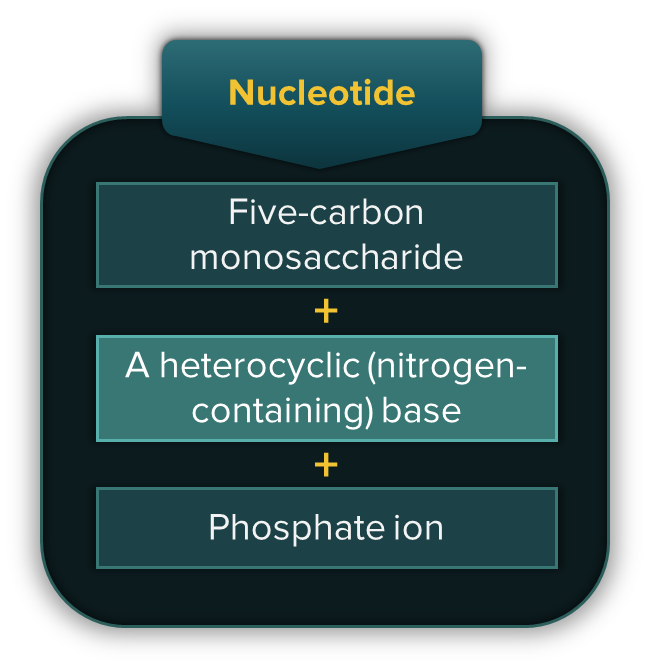
Nucleic acids are made up of monomeric units called nucleotides. A nucleotide consists of a five-carbon monosaccharide, a heterocyclic base (nitrogen-containing) and a phosphate ion.
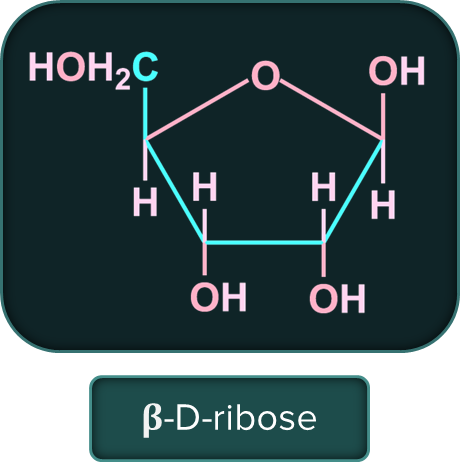
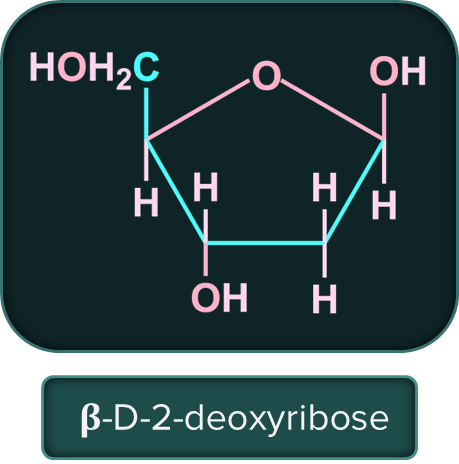
The sugar molecule in DNA (deoxy ribose nuclei acid) is 𝛃-D-2-deoxyribose and in RNA (ribose nucleic acid) is 𝛃-D-ribose.
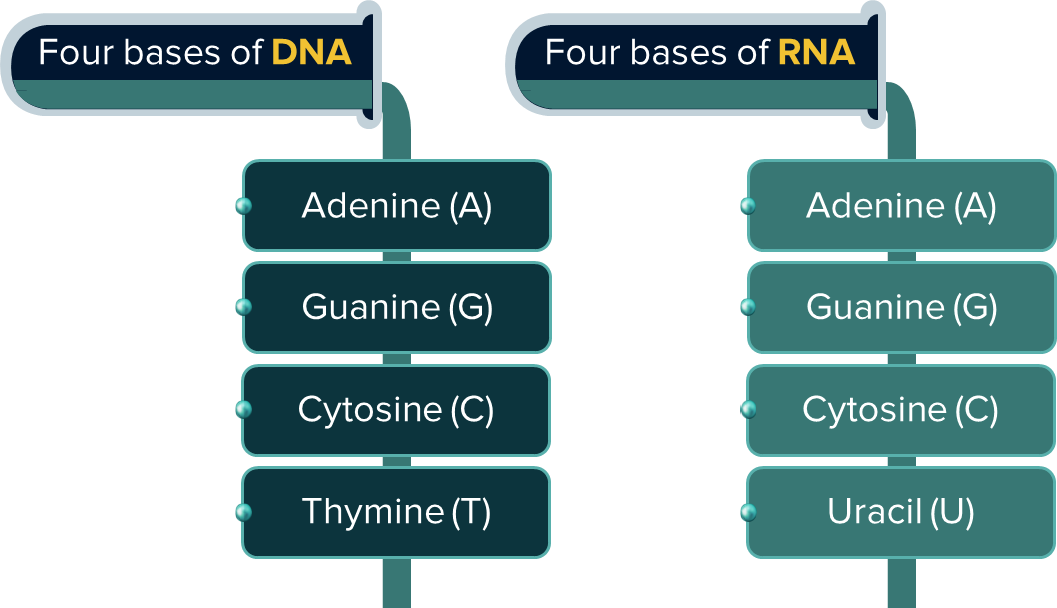
The following are the nitrogenous bases present in DNA and RNA.
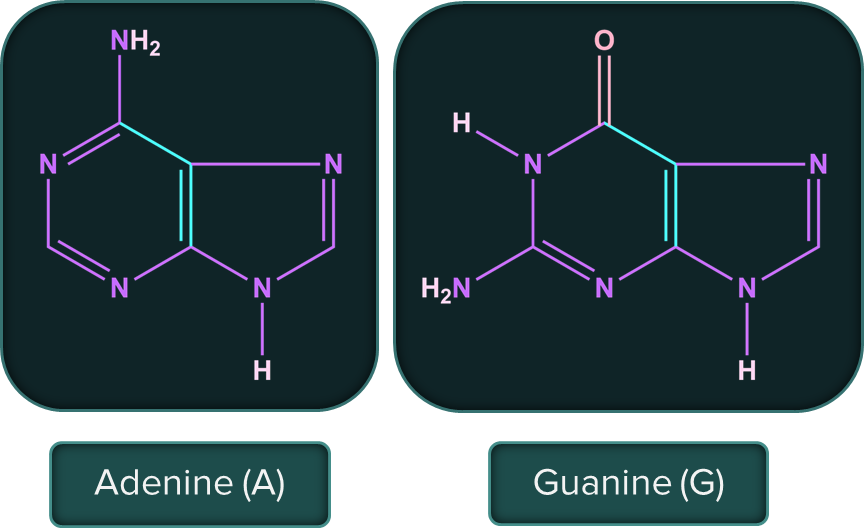
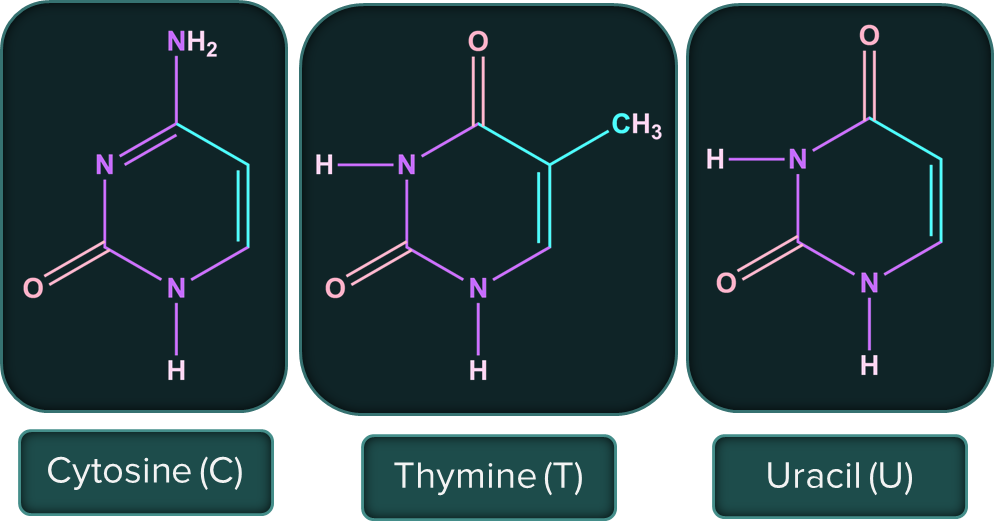
Nucleic acids are long-chain polymers of nucleotides, so they are also known as polynucleotides. The information regarding the sequence of nucleotides in the chain of nucleic acid is known as its primary structure. Nucleic acids have a secondary structure as well.
DNA:
- James Watson and Francis Crick gave a double-stranded helix structure for DNA.
- Two nucleic acid chains are wound about each other and held together by hydrogen bonds between pairs of bases.
- The two strands are complementary to each other because the hydrogen bonds are formed between specific pairs of bases.
- Adenine (A) forms hydrogen bonds with thymine (T). Whereas, cytosine (C) forms hydrogen bonds with guanine (G).
RNA:
- In the secondary structure of RNA, helices are present which are only single-stranded.
- Sometimes, they fold back on themselves to form a double helix structure.
Vitamins
Vitamins are organic compounds required in the diet in small amounts to perform specific biological functions for normal maintenance of optimum growth and health of the organism. Most vitamins cannot be synthesised in our bodies. All the vitamins are generally available in our diet. Vitamins can be classified into two types based on their solubility in water or fat.
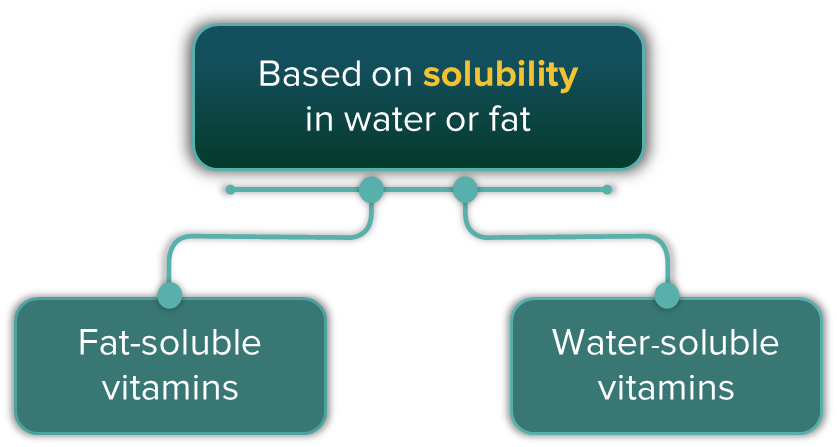
Fat-Soluble Vitamins: These are soluble in fat and oil but insoluble in water. Examples include vitamins A, D, E and K.
Water-Soluble Vitamins: These are insoluble in fat and oil but soluble in water. Examples include B- group vitamins and vitamin C.
The following table gives the important vitamins, their sources and their deficiency diseases.
|
Vitamin |
Sources |
Deficiency Diseases |
|
Vitamin A |
Fish liver oil, carrots, butter, and milk. |
Xerophthalmia (hardening of eye cornea), night blindness. |
|
Vitamin B1 (Thiamine) |
Yeast, milk, green vegetables, and cereals. |
Beriberi (loss of appetite and retarded growth). |
|
Vitamine B2 (Riboflavin) |
Milk, egg white, liver, and kidney |
Cheilosis (fissuring at corners of mouth and lips), digestive disorders, and burning sensation of the skin. |
|
Vitamine B6 (Pyridoxine) |
Yeast, milk, egg yolk, cereals, and grams. |
Convulsions |
|
Vitamin B12 |
Milk, fish, egg, and curd. |
Pernicious anaemia (RBC deficiency in haemoglobin). |
|
Vitamin C (Ascorbic acid) |
Citrus fruits, amla, and green leafy vegetables. |
Scurvy (bleeding gums). |
|
Vitamin D |
Fish, egg yolk, and exposure to sunlight. |
Rickets (bone deformities in children) and osteo-malacia (soft bones and joint pain in adults). |
|
Vitamin E |
Vegetable oils like wheat germ oil, and sunflower oil. |
Increased fragility of RBCs and muscular weakness. |
|
Vitamin K |
Green leafy vegetables |
Increased blood clotting time. |
Lipids
Lipids are chemical molecules that are linked to fatty acids, are insoluble in water but soluble in organic solvents, and are utilised by living cells. They include lipids such as phospholipids, mono, di, triglycerides, fat-soluble vitamins, sterols, waxes, and others. Lipids are not polymeric substances like proteins, nucleic acids, or carbohydrates. The main source of energy and a crucial component of the cellular structure are lipids.
Fats and oils belong to the chemical family of lipids, which is organic. These energetic molecules play a number of different roles in the human body. Here are a few essential characteristics of lipids.
- Adipose tissue in the body stores lipids, which are nonpolar oily or fatty molecules.
- A wide range of compounds known as lipids are essentially composed of hydrocarbon chains.
- Lipids are organic substances with a lot of energy that provide energy for a number of biological functions.
- A family of compounds called lipids is differentiated by its members' solubility in nonpolar solvents and insolubility in water.
- Because they make up the cell membrane, a mechanical barrier that isolates a cell from its environment, lipids play a crucial role in biological processes.
Recommended Videos
Biomolecules Class 12 Chemistry One Shot | Chapter 14 Chemistry Class 12 | NEET 2022 Chemistry Exam
Biomolecules Class 12 Chemistry One-Shot (Full Chapter Revision) Concepts & Imp Ques | JEE Main 2022
JEE Main 2022 - Top 10 Most Important & Expected Questions of Biomolecules Class 12 Chemistry
JEE Biomolecules and Polymers | Target JEE | Solved Questions | JEE Chemistry
Practice Problems
Q1. Which among the following is a nitrogenous base that is not found in DNA?
A. Uracil
B. Thymine
C. Guanine
D. Cytosine
Answer: A
Solution: Adenine, guanine, cytosine, and thymine are the four primary nitrogenous bases found in DNA. Thymine is replaced by uracil in RNA.
So, option A is the correct answer.
Q2. Which of the given statements is correct regarding lipids?
A. Adipose tissue in the body stores lipids, which are nonpolar oily or fatty molecules.
B. A wide range of compounds known as lipids is essentially composed of hydrocarbon chains.
C. Lipids are organic substances with a lot of energy that provide energy for a number of biological functions.
D. All of the above.
Answer: D
Solution: Some key features of lipids are listed below.
- Adipose tissue in the body stores lipids, which are nonpolar oily or fatty molecules.
- A wide range of compounds known as lipids are essentially composed of hydrocarbon chains.
- Lipids are organic substances with a lot of energy that provide energy for a number of biological functions.
- A family of compounds called lipids is differentiated by its members' solubility in nonpolar solvents and insolubility in water.
- Because they make up the cell membrane, a mechanical barrier that isolates a cell from its environment, lipids play a crucial role in biological processes.
The statements given in options A, B and C are correct.
So, option D is the correct answer.
Q3. When two monosaccharides form a bond and are joined, a disaccharide is created. What is this bond formed known as?
A. Peptide
B. Phosphodiester
C. Ionic
D. Glycosidic
Answer: D
Solution: When two monosaccharide units combine, they lose a water molecule and form an oxide bond. The glycosidic linkage is a covalent binding between two monosaccharide molecules created by an oxygen atom.
So. option D is the correct answer.
Q4. Sucrose is a sugar that when hydrolysed produces a combination of
A. dextrorotatory glucose and laevorotatory fructose
B. dextrorotatory fructose and laevorotatory glucose
C. Fructose and maltose
D. None of the above
Answer: A
Solution: When hydrolysed, sucrose, a dextrorotatory sugar, yields a mixture of dextrorotatory glucose and laevorotatory fructose. Because fructose has a greater specific rotation than glucose, the resulting mixture is laevorotatory.
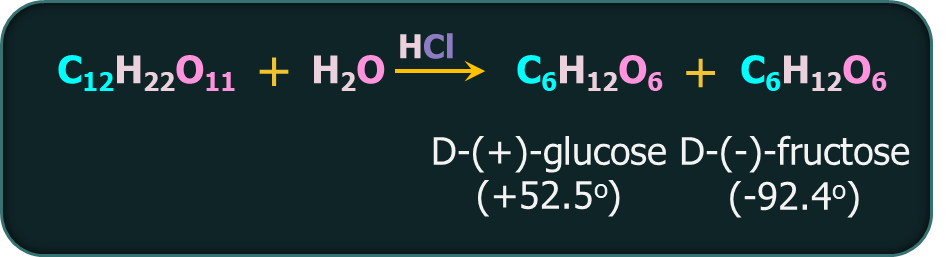
So, option A is the correct answer.
Frequently Asked Questions – FAQ
Q1. What are the different types of lipids?
Answer: Fats, phospholipids, steroids, and waxes are the major lipid categories. Lipids as a class of chemicals are water insoluble but soluble in other organic solvents. Acetone and ether are two examples of such solvents. The most common lipid categories include waxes, steroids, phospholipids, and fats.
Q2. What are the different types of nucleic acids found in RNA?
Answer: The nitrogenous bases adenine, guanine, cytosine, and uracil are all found in RNA. Deoxyribonucleic acid (better known as DNA) and ribonucleic acid (better known as RNA) are two examples of nucleic acids. Long strands of nucleotides are bound together by covalent bonds in these molecules.
Q3. What exactly are proteins and what are their functions?
Answer: Large, intricate molecules known as proteins perform numerous crucial functions in the human body. They perform the majority of their job in cells and are crucial for the development, maintenance, and control of the body's tissues and organs. Proteins are composed of hundreds or thousands of amino acids, which are linked together in long chains. Twenty different kinds of mixable amino acids make up a protein. Each protein's three-dimensional structure and purpose are determined by its amino acid sequence.
Q4. What are carbohydrates' primary functions?
Answer: The four main activities of carbohydrates in the body are to supply energy, store energy, make macromolecules, and free up protein and fat for other purposes. The majority of glucose energy is converted to glycogen in the muscle and liver.
Q5. What are the most common carbohydrate sources?
Answer: Bread, vegetables and fruits, as well as dairy, are high in carbohydrates. Carbohydrates are sugars, starches, and fibres found in fruits, cereals, vegetables, and milk products.
Q6. What function do biomolecules have in disease diagnosis?
Answer: All living processes and activities require biomolecules. This involves illness progression. As a result, reliable biomolecule identification is critical in illness diagnosis and therapy.
Q7. What are necessary fatty acids, and what do they do?
Answer: Essential fatty acids are fatty acids that must be consumed since the body cannot produce them and need them for good health and biological processes. These do not include fats used just for fuel. Because the body cannot generate them on its own, they must be obtained through your diet.
Q8. What are the benefits of lipids for living things?
Answer: Lipids are organic compounds that are high in energy and supply energy for several living processes. Lipids are a class of compounds distinguished by their insolubility in water and solubility in nonpolar solvents.


