-
Call Now
1800-102-2727
Arrhenius Rate Constant and Temperature- Arrhenius Equation, Arrhenius Rate Constant and Pre-Exponential Factor, Graphical Representation, Effect of Temperature, Applications of Arrhenius Equation, Practice Problems, FAQs
When you bring fresh milk packets home from the market, your mom always cleans them and immediately places them in the freezer box of the refrigerator. Only when needed, does she takes them, defroze them and boil them for use. Why not leave them in open like onions?
What about if she forgets to put them in the refrigerator?
If not cooled, milk turns sour. Why is this the case?
Definitely, the temperature has something to do with the speed of souring milk. Lower temperatures in the refrigerator slow down the conversion.
Clearly, the average temperature in the refrigerator is lower than the temperature at room temperature. In fact, increasing the temperature prompts a chemical reaction. Is it true that temperature has a similar effect on some other chemical reactions? Svante August Arrhenius provided the answers to all these questions. The Arrhenius equation is a mathematical relationship that explains the temperature dependence of the rate of a chemical reaction. Let's have a tour of this article to learn more about the Arrhenius equation.
Table of Content
- Arrhenius Equation
- Arrhenius Rate Constant and Pre-Exponential Factor
- Relation Between Arrhenius Rate Constant and Temperature
- Application of Arrhenius's Equation
- Practice Problems
- Frequently Asked Questions-FAQs
Arrhenius Equation
The rate constant is a proportionality constant that describes the relationship between the molar concentration of reactants and the rate of a reaction. The rate constant is represented by the symbol ‘k’
Rate of a reaction = k[Reactant Concentration]x
Arrhenius related the proportionality constant of the rate equation to the activation energy of the reaction. The Arrhenius Equation is a formula for calculating the temperature dependence of reaction rate. It has important applications in deciding the rate at which a reaction occurs and also plays a significant role in determining the activation energy.
Mathematically,
Here,
‘k’ represent the rate constant of the reaction.
‘A’ represents the Arrhenius factor, frequency factor, or pre-exponential factor.
‘T’ represents the temperature on the absolute kelvin scale.
‘Ea’ represent the activation energy of the reaction.
‘R’ represent the universal gas constant.
The above equation is known as the Arrhenius rate equation.
According to the Arrhenius equation, the rate constant of a reaction increases exponentially with increasing temperature, and the rate of a reaction is directly proportional to the rate constant. As a result, as the temperature rises, so does the rate of the reaction.
Let us take 'ln' on both sides of the equation(i) we get,
…….(ii)
Equation(ii) is a modified form of the Arrhenius equation that can be used for a variety of purposes.
When there is insufficient energy, reactions do not progress, transition states cannot be formed, and products cannot be formed. To convert reagents into products, a small amount of energy known as activation energy is required.
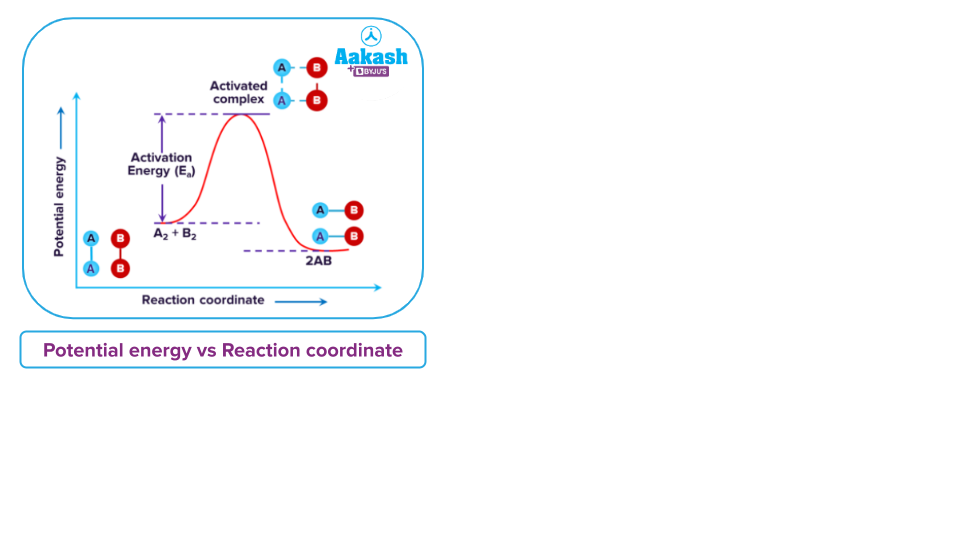
When reactants are converted into activated complexes, activation energy is required. From here the activated complex decomposes, to form the product. In the case of the products having lower energy than the reactants, energy is released to exhibit exothermicity.
Arrhenius Rate Constant and Pre-Exponential Factor
The pre-exponential factor, usually called as frequency factor,n the Arrhenius equation, is denoted by the symbol 'A'. This represents the frequency of collisions between reactant molecules with correct orientations that will result in product formation.
The pre-exponential factor is frequently represented by the equation:
A=Z
Where ‘Z’ is the frequency factor (collision frequency)
'' is the steric factor (related to the orientation of molecules).
Pre-exponential factor(A) takes on different values for different reactions, it must be determined experimentally. It is also impacted by the temperature at which the reaction can occur.
Relation Between Arrhenius Rate Constant and Temperature
Let us consider, that a reaction with 'Ea' represent the energy barrier known as activation energy for any given reaction and 'k1' and 'k2' represent the rate constants at temperatures 'T1' and 'T2', respectively.
Substituting, the above variables in the equation(i) we get,
…..(iii)
…..(iv)
Subtracting equation (iv) from the equation (iii) we get,
Converting ln into log by multiplying with 2.303 we get,
This equation is known as Vant Hoff equation.
Graphical representation of a plot of ln k vs 1/T from Arrhenius equation
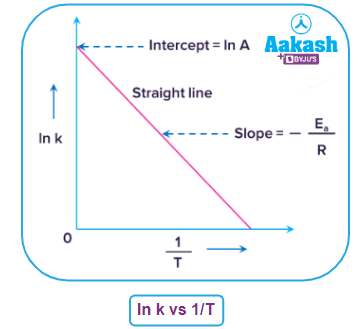
We know that the general expression of a straight line is Y=mX+C....(v)
where 'Y' and 'X' are the coordinate axes, 'm' is the slope of the curve, and 'C' is the intercept on the Y-axis.
Now, according to the Arrhenius equation in its modified form,
…….(ii)
When we compare equations (ii) and (iv), we can conclude that
The above curve is a straight line with a constant and negative slope
The rate constant and temperature in a chemical reaction is directly proportional to each other. The rate of reaction increases exponentially as the temperature rises. As the temperature of the chemical reaction rises, so does the rate constant. The kinetic energy of the reaction rises as the temperature rises. As the temperature rises, the kinetic energy in the number of molecules exceeds the activation energy. The overall reaction rate increases, whereas the activation energy decreases.
Let us consider an example for the decomposition of nitrogen dioxide,
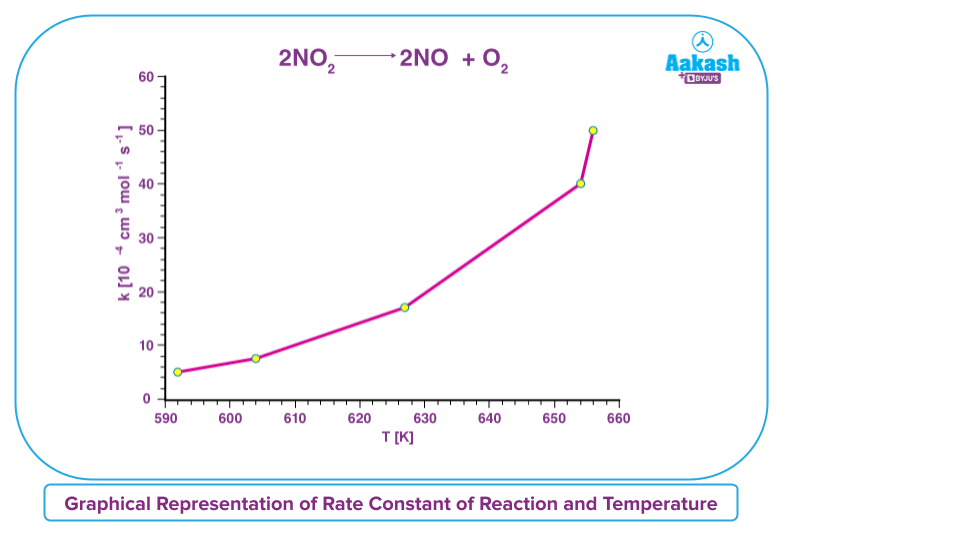
A graph plotting the rate constant of reaction(k) on the Y-axis and the absolute temperature (T) on the X-axis is provided below for the nitrogen dioxide decomposition reaction (given by2NO2(g) 2NO(g) +O2(g) ). It is worth noting that the rate of the reaction increases when the temperature rises.
Applications of Arrhenius Equation
Some real-life applications of the Arrhenius equation include:
- When milk is left out in the open, it sours much faster than when it is kept in the refrigerator.
- Butter goes rancid much faster in the summer than it does in the winter.
- Eggs cook faster at the earth's surface but take much longer in the mountains.
- Cold-blooded animals, such as reptiles, become sluggish in the winter.
- The Arrhenius equation can be used to determine the optimal temperature for obtaining the maximum product.
Related video: https://www.youtube.com/watch?v=xWvnxyWRuB0
Practice Problems
Q1. If the activation energy required for the conversion of reactant A into product B is50 kJ mol-1. Determine the value of the rate constant for the reaction at 300 K. Given the value of pre-exponential factor(A) is 50 M-1 s-1.
- 6.8210-8 M-1 s-1
- 9.110-8 M-1 s-1
- 9.8210-10 M-1 s-1
- 9.8210-8 M-1 s-1
Answer: (D)
Solution:
According to the given question,
Activation energy(Ea) = 50 kJ mol-1
Temperature at which reaction takes place = 300 K
Pre exponential factor(A) = 50 M-1 s-1
Rate constant of the reaction be x M-1 s-1
We know that modified Arrhenius equation is mathematically represented as,
…….(ii)
Putting the values of the known variables in the above equation we get,
ln x = -16.14
Taking antilog on both the side of the reaction we get,
Therefore rate constant for the given reaction will be
Q2. At 500 K, the rate constant of a reaction is 110-8 s-1, but when the temperature is increased to 800 K the rate constant of the reaction becomes 0.510-6 s-1 . Calculate the activation energy in J mol-1 required for the reaction to convert into the product.
- 94.473 J mol-1
- 4,473.3 J mol-1
- 473.3 J mol-1
- 94,473.3 J mol-1
Answer: (D)
Solution: According to the given data,
Let the activation energy of the reaction(Ea)= x J mol-1
Initial temperature at which reaction takes place(T1)=500 K
Final temperature at which reaction takes place(T2)=800 K
Rate constant of reaction(k1) at 500 K temperature = 110-8 s-1
Rate constant of reaction(k2) at 800 K temperature = 0.510-6 s-1
Using the Van’t Hoff equation we know that Arrhenius equation at two different temperatures is represented as,
Putting the values of the variables in the above equation we get,
Using the properties of logarithm and solving for x we get,
Therefore the activation energy for the given reaction will be x=94,473.3 J mol-1
Q3. The Arrhenius equation can be used to express the rate of a reaction as . In this equation, Ea stands for:
- The fraction of molecules that have more energy than the reaction's activation energy.
- The amount of energy above which all colliding molecules will form product
- The minimum energy below which colliding molecules will not form the product.
- At a given temperature, T, the total energy of the reacting molecules.
Answer: (C)
Solution: In the Arrhenius equation represent the energy barrier known as activation energy for any given reaction. When there is insufficient energy, reactions do not progress, transition states cannot be formed, and products cannot be formed. To convert reagents into products, a small amount of energy known as activation energy is required. Therefore option (C) is correct.
Q4. Let’s consider the second order reaction in which A B the Arrhenius equation is represented as . Activation energy for the given reaction in J mol-1 will be
A). 8314 J mol-1
B). 54.6 J mol-1
C). 8.314 J mol-1
D). 4157J mol-1
Answer: (A)
Solution: According to the given question Arrhenius equation is represented as,
We know that modified Arrhenius equation is mathematically represented as,
Here,
‘k’ represents the rate constant of the reaction which depends on the temperature
‘A’ represent the Arrhenius factor, frequency factor, or pre-exponential factor.
‘T’ represents the temperature on the absolute Kelvin scale.
‘Ea’ represent the activation energy of the reaction.
‘R’ represent the universal gas constant.
Comparing the equation vi and ii we get
ln A= 4
Therefore, Activation energy of the given reaction will be 8314 J mol-1
Frequently Asked Questions-FAQs
Q1. What is the basis for the formulation of the Arrhenius equation?
Answer: The Arrhenius equation is derived from the Arrhenius theory, which states that a reaction can occur only when the reactants collide with a specific amount of energy in the proper orientation. The collision causes the formation of an unstable state that lasts only a short time. This is referred to as a transition state. The transition-state complex / activated complex is the unstable complex at the transition state. The activation energy is the minimum amount of energy required to form the transition state. When the transition state is formed, it becomes unstable and results in the formation of the product.
Q2. What is the effect of catalyst on the rate constant of reaction?
Answer: Catalysts alter the reaction mechanism, which alters the reaction rate. In general, a catalyst accelerates a reaction by lowering the activation energy required for it to proceed. When adding negative inhibitors, the rate of reaction decreases as the activation energy increases. But the rate constant of the reaction remains unchanged when the reaction takes place in the presence or the absence of the catalyst.
Q3. What is the unit of pre-exponential factor ‘A’ in the Arrhenius equation?
Answer: The units of pre-exponential factor ‘A’ in the Arrhenius equation are determined by the unit of rate constant of the reaction. The value of 'A' for a second-order reaction, for example, is expressed in L.mol-1s-1 (or, M-1 s-1 since M represent the molarity and its unit is mol.L-1) and that of a first-order reaction is s-1.
Q4. What is the importance of the Arrhenius equation's pre-exponential factor?
Answer: The frequency factor, also known as the Arrhenius factor, is represented by the symbol 'A.' It is concerned with molecule collisions and can be thought of as the frequency of correctly oriented collisions between molecules that have enough energy to cause a chemical reaction.
Q5. What are the factors that affect the rate of reaction?
Answer: The following factors influence the rate of a chemical reaction:
- Reactant concentration
- Temperature
- Light exposure (Radiation)
- The composition of reactants and products


