-
Call Now
1800-102-2727
Aromatisation – Definition, Mechanistic Pathways of Aromatization, Aromatization of n-Alkanes, Practice Problems and FAQ
Enticing aromas have the power to attract everyone! Similarly, ‘aromatic compounds’, in the world of chemistry, have immense power to influence a significant section of the chemical kingdom. Some of them are extremely essential for a healthy life, and some are indispensable members of the chemical industries that impact us positively in a multitude of ways.

All living things have a biochemistry of aromatic compounds, which is crucial. Certain aromatic amino acids are essential for protein biosynthesis. Additionally, the five nucleotides that make up the genetic code in DNA and RNA—adenine, thymine, cytosine, guanine, and uracil—are all aromatic purines or pyrimidines. The molecule heme in haemoglobin has a 22 -electrons strong aromatic system. Similarly, chlorophyll has an aromatic system.
The four most important aromatic hydrocarbons for the chemical industry are ortho-xylene, para-xylene, benzene, and toluene. They are used to make a variety of significant chemicals and polymers, including nylon, aniline, polyester, phenol, and styrene.
Each year, about 35 million tonnes are manufactured worldwide. Hence it is essential for chemistry enthusiasts and students to understand the process of aromatization, which forms the basis of transforming a straight-chain alkane molecule to its corresponding aromatic hydrocarbon.
Let’s get a detailed know-how of the process.
TABLE OF CONTENTS
- Aromatisation
- Mechanistic Pathways of Aromatisation
- Uses of Aromatisation Reactions
- Practice Problems
- Frequently Asked Questions – FAQ
Aromatisation
Aromatic hydrocarbons are hydrocarbons that have delocalized pi electrons and sigma bonds between their carbon atoms in a ring. Benzene is one such example. They are referred to as aromatic because of their characteristic pleasant fragrance. For a compound to be aromatic, it has to follow the given conditions.
- The compound should be cyclic.
- The compound should have a planar structure.
- sp2 hybridised atoms that can create a delocalized system of electrons should be present in aromatic rings.
- It should follow Huckel’s rule of aromaticity, i.e. it should have (4n+2) -electrons delocalised within the ring, where n= 0,1,2,3... .
An aromatic system is created from a single non-aromatic precursor by a chemical reaction known as aromatisation reaction. Paraffins (acyclic hydrocarbons) undergo dehydrocyclisation to become aromatic compounds. As demonstrated by the transformation of cyclohexane into benzene, aromatization is typically accomplished through the dehydrogenation of pre-existing cyclic molecules or at times, by cyclisation of straight chain alkanes (above 6 carbon atoms).
Mechanistic Pathways of Aromatisation
There are several pathways or mechanisms that are responsible for aromatisation. Let’s see them in detail.
Cyclization
At a temperature of 773 K, n-alkanes undergo oxidative dehydrogenation in the presence of vanadium or chromium oxides and alumina in order to aromatize. In this reaction, each terminal carbon atom of n-hexane loses one hydrogen atom in order to create cyclohexane. This reaction is cyclization. n-Alkanes (C≥6) on heating to 773 K at 10-20 atm in presence of oxides of chromium, molybdenum or vanadium supported over alumina get aromatized to benzene and its homologues.
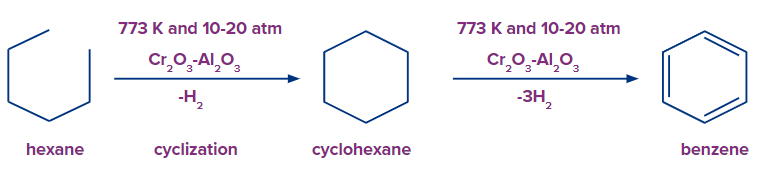
Oxidative or Catalytic Dehydrogenation
The conceptually simplest process for aromatization for the cyclohexane, cyclohexene, and cyclohexadiene family of compounds is dehydrogenation. As unsaturation increases, the activation barrier reduces. Cyclohexadiene is hence particularly susceptible to aromatization. This can be brought about in the presence of platinum catalysts.
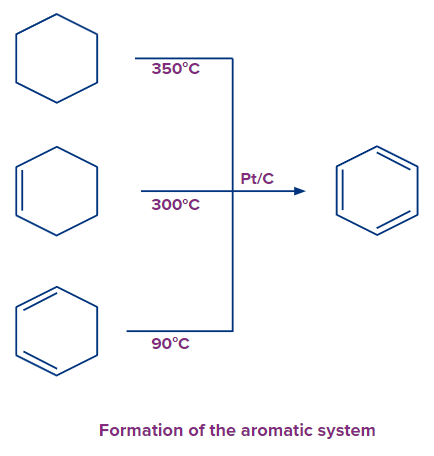
Dehydration
The dehydration of oximes in acidic conditions can also lead to aromatization. In a process known as the Semmler-Wolff reaction or Wolff aromatization, the oxime of cyclohexenone is converted into aniline.
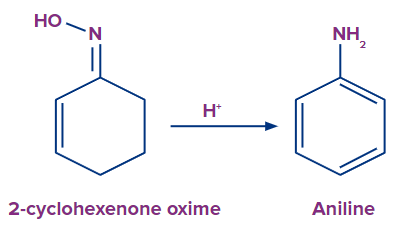
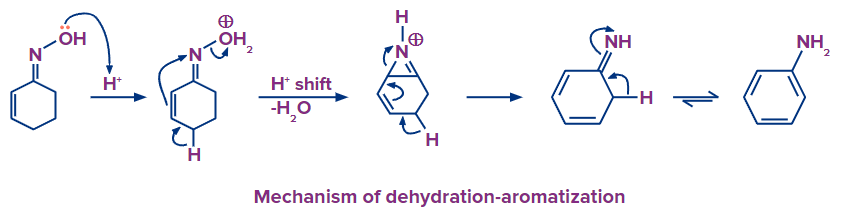
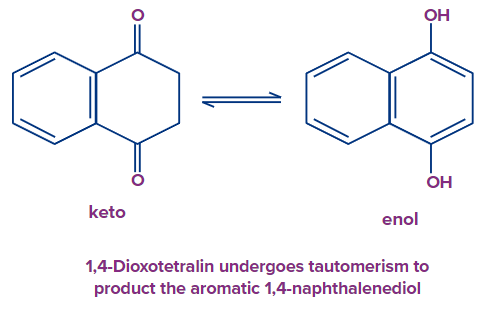
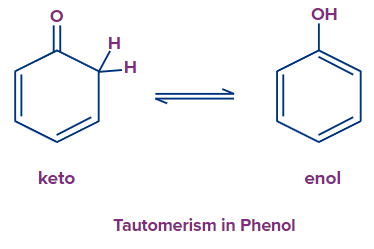
Tautomerization
Aromaticity is the most potent factor that favours an enol tautomer formation. For example, phenol is an aromatic tautomer produced by cyclohexadienones.
Abstraction of Proton and Hydride
Traditionally, aromatization reactions require altering the substrate's C:H ratio. The aromatic conjugate base cyclopentadienyl anion, isolable as sodium cyclopentadienide, is produced when proton removal is applied to cyclopentadiene.
Hydride elimination may be necessary during aromatization. The aromatization of cycloheptatriene with hydride acceptors produces tropylium,
Aromatization from Acyclic Precursors
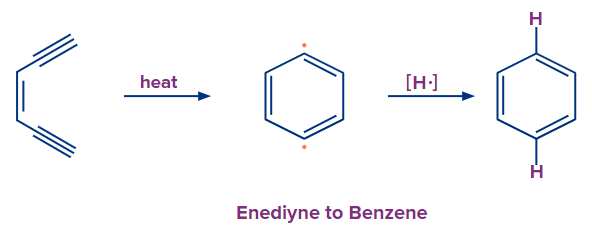
Although it happens less frequently in organic synthesis, the aromatization of acyclic precursors contributes significantly to the formation of benzene, toluene and xylene in oil refineries.
Alkynes are among the acyclic precursors that are most susceptible to aromatizations because they have undergone partial dehydrogenation. An enediyne is transformed by the Bergman cyclization into a dehydrobenzene intermediate diradical, which absorbs hydrogen to aromatize.
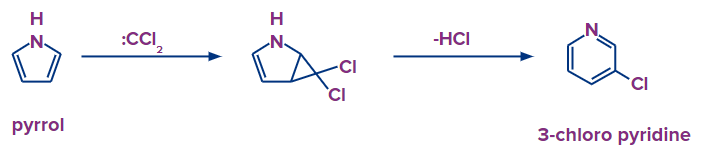
Dearomatization to Aromatization
The initial stage in this conversion of pyrrole to pyridine involves de-aromatization. The aromatization process comes next. Pyrroles can engage in a [2+1]-cycloaddition reaction with carbenes, such as dichlorocarbene (:CCl2). The Ciamician-Denstedt rearrangement is another name for it.
Uses of Aromatization Reactions
- Methylcyclohexane to toluene conversion is a typical aromatization process. The synthesis of gasoline from petroleum liquids and crude oil uses this platinum (Pt)-catalysed method on a large scale.
- An essential step in oil reforming in the petrochemical industry is aromatization. The conversion of aromatics from naphthenes through dehydrogenation is one of the main reforming processes. A good example of the platinum-catalysed process is the transformation of methylcyclohexane (naphthene) into toluene (aromatic).
- Paraffins (acyclic hydrocarbons) undergo dehydrocyclization to become aromatic compounds which is important in the chemical as well as the petrochemical industry.
- Normal aromatization is desirable and serves a major biological purpose; it aids in the body's maintenance of a temperature, hormonal, and mass balance that is in harmony with health. Additionally, it aids in the production of lipids and cholesterol.
- The enzymes known as aromatases aromatize the steroid ring structure. This particular conversion is important in regulating hormonal balances and also essential in manufacturing several steroids in medicinal and pharmaceutical chemistry.
Recommended Videos
Hydrocarbons Class 11 Chemistry One-Shot by Rakhi Ma'am | JEE Mains & Adv 2023 | One-Day One-Chapter
Aromaticity | General Organic Chemistry | Class 11 Chemistry | NEET 2022 & 2023 | Aakash NEET
Practice Problems
1. Aromatization can be performed for n-alkanes having (C≥6) using which of the following?
a. Fe3O4
b. Al2O3
c. Cr2O3/Al2O3, 600C
d. conc. HNO3
Answer: C
Solution: n-Alkanes (C≥6) on heating to 873 K at 10-20 atm in presence of oxides of molybdenum, vanadium or chromium supported over alumina get aromatized to benzene and its homologues. In this reaction, each terminal carbon atom of n-hexane receives one hydrogen atom in order to create cyclohexane. This is a cyclisation reaction and is called oil reforming.
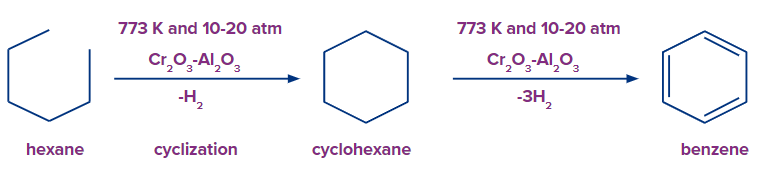
So, option C is the correct answer.
2. Give an example of catalytic aromatization forming a condensed aromatic compound.
Solution: With platinum or palladium catalysts that are either finely powdered or deposited on activated charcoal, the reactions are conducted at temperatures of about 300°C. The elimination of hydrogen from the equilibrium causes the process to move toward dehydrogenation. This is the principle applied in the preparation of naphthalene, which is a condensed aromatic compound.
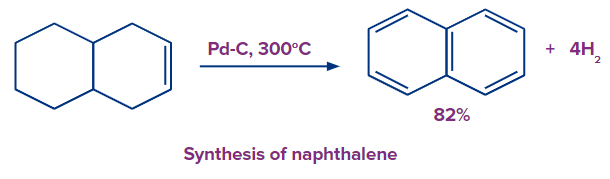
3. The enzyme essential for the aromatization of biomolecules such as steroids, is
a. Isomerase
b. Aromatase
c. Ligase
d. None of the above
Answer: B
Solution: The enzymes known as aromatases aromatize the steroid ring structure. Androstenedione to estrone and testosterone to estradiol are the two particular transformations that are assisted by this enzyme.
So, option B is the correct answer.
4. Catalyst used in the aromatization of methylcyclohexane to toluene is
a. Platinum
b. Nitrogen dioxide
c. Silica
d. V2O5
Answer: A
Solution: Methylcyclohexane to toluene conversion is a typical aromatization process. Petroleum is used in a large-scale process that uses platinum (Pt) to produce gasoline.
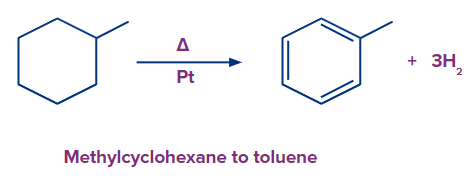
So, option A is the correct answer.
Frequently Asked Questions – FAQ
1. Is aromatization of saturated as well as unsaturated hydrocarbons possible?
Answer: Yes, aromatization of both saturated and unsaturated hydrocarbons via catalytic dehydrogenation is possible. Majorly for unsaturated hydrocarbons, the temperature conditions necessary may not be too vigorous due to slight ease of reaction. But for straight-chain hydrocarbons (saturated or unsaturated), the first step involved in cyclisation, followed by aromatization.
2. What are the two types of reforming processes used in industry that involve aromatization?
Answer: To improve the combustion properties of low-quality gasoline stocks, two types of reforming or aromatization procedures are utilised in the petroleum sector.
Thermal Reforming: Heat and pressure are used to change low-grade naphtha into molecules with a high octane number.
Catalytic Reforming: An alkane or cycloalkane with six or more carbon atoms can be transformed into the corresponding aromatic hydrocarbon by a process called catalytic reforming.
3. How does catalytic reforming involve aromatization?
Answer: Catalytic reforming converts low-grade naphthas (a mixture of cyclic and straight-chained hydrocarbons having between five and six carbon atoms) into high-octane reformate product by coming into contact with an acidic catalyst containing platinum at high temperatures and pressures (aromatic hydrocarbons). In order to produce high-octane, aromatic hydrocarbons, low-octane, linear hydrocarbons known as paraffin are converted into branched alkanes known as isoparaffins and cyclic naphthenes. Changing methylcyclohexane into toluene is one example.
4. What is the significance of aromatase in our body?
Answer: The enzyme known as aromatase is responsible for converting testosterone into oestrogen. Additionally, it aids in regulating the body's active levels of cortisol, which is used to manage the immunological system. More testosterone will be turned into oestrogen during a testosterone spike if the body has a higher level of aromatase. Because it converts androstenedione to estrone and testosterone to estradiol, aromatase is to blame for the emergence of chronic disorders including cancer and autoimmune disease.


