-
Call Now
1800-102-2727
Adsorption - Important Terms, Difference Between Adsorption and Absorption, Mechanism, Thermodynamics, Types of Adsorption, Factors Affecting Adsorption of Gases on Solids, Applications
Do you know why doctors recommend wearing N-95 masks during and after the Covid-19 pandemic?
(No, not because pharmaceutical companies can make a lot of money!)
N-95 masks eliminate viruses and other harmful microbes, allowing us to breathe only virus-free air.
But how do they accomplish this?
It all comes down to a phenomenon known as 'adsorption.'
N-95 masks are made of multiple layers of filters and membranes that adsorb viruses and other harmful microbes.
Most of us are familiar with the term absorption, but are we familiar with adsorption?
Are absorption and adsorption synonymous?
Let's get to know.

TABLE OF CONTENTS
- Adsorption
- Adsorbate
- Adsorbent
- Desorption
- Absorption
- Difference Between Adsorption and Absorption
- Competitive Adsorption
- Mechanism of Adsorption
- Thermodynamics of Adsorption
- Types of Adsorption
- Factors Affecting the Adsorption of Gases on Solids
- Practice Problems
- Frequently Asked Questions - FAQ
Adsorption
It is the accumulation of a molecular species at the surface rather than in the bulk of a solid or a liquid.
It is the buildup of a molecules at the surface of a solid or liquid rather than in its bulk.
Heinrich Kayser, a German physicist, was the first to coin the term adsorption in 1881. Adsorption is a surface phenomenon by the virtue of which particles are attached to the surface of a material. Generally, adsorption involves atoms, molecules or ions of a gas, liquid or solid in a dissolved state that is attached to the surface of a material.
Adsorbate
The molecular species or substance, which concentrates or accumulates at the surface of the adsorbent.
The substance or molecules that accumulates or concentrates at the adsorbent's surface.
Adsorbent
The material on the surface of which the adsorbate molecules get adsorbed.

Since adsorption is a surface phenomenon, as the surface area of the adsorbent increases, the amount of adsorbate getting attached to the surface of the adsorbent increases, i.e. adsorption increases.
Desorption
The process of removing an adsorbed substance (adsorbate) from the surface on which it is adsorbed (adsorbent).
Absorption
The phenomenon in which the particles of a gas or a liquid get uniformly distributed throughout the body of the solid. The process by which the particles of gas or liquid get distributed uniformly throughout the body is called adsorption.
Difference Between Adsorption and Absorption
|
Adsorption |
Absorption |
|
Adsorption occurs when the concentration of another material on the surface is higher than in the bulk. |
Absorption, on the other hand, is a phenomenon in which a substance's molecules are equally dispersed throughout the body. |
|
It is a surface phenomenon. |
It is a bulk phenomenon. |
|
Example: When silica gel is placed in a watery environment, it adsorbs the water. At the surface of silica gel, there is a high concentration of water vapour. |
Example: Calcium chloride, on the other hand, absorbs water when placed in a moist environment. The liquid is equally dispersed throughout the body, unlike silica gel which absorbs water only on its surface. |
Competitive Adsorption
When an adsorbent is in contact with more than one species, there will be competition amongst them to get adsorbed on the surface of the adsorbent. The one that is more strongly adsorbed gets deposited first in preference to the others. A strongly adsorbed substance may displace a weakly adsorbed substance.
Example: NH3 can displace O2 or N2 from the surface of the charcoal.

Mechanism of Adsorption
Inside the adsorbent, all the forces acting between the particles are mutually balanced. Inside the adsorbent, all the forces acting are mutually balanced between the particles. On the surface of the adsorbent, the particles are not surrounded by the same particles on all sides. On the adsorbent’s surface, the particles are not surrounded on all sides by the same particles. Hence, they possess unbalanced or residual forces of attraction.
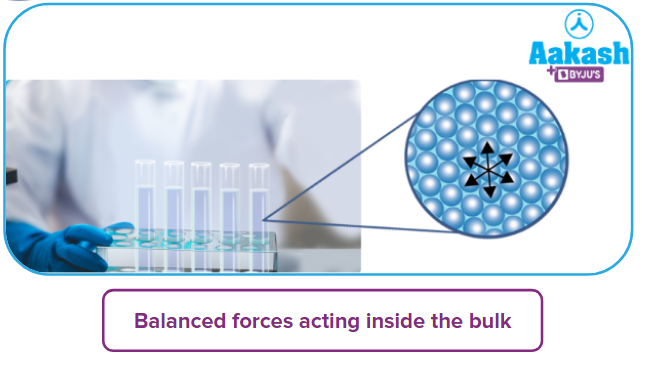

These unbalanced forces of attraction of the adsorbent are responsible for attracting the adsorbate particles on its surface.
Thermodynamics of Adsorption
During adsorption, there is always a decrease in the residual forces of the surface, i.e., there is a decrease in the surface energy which appears as heat. Thus, heat is released during adsorption (exothermic process). So, the heat of adsorption is negative. Therefore, adsorption is always exothermic, i.e. ΔH <0.
With adsorption, the degree of randomness of the particles decreases as the adsorbate particles are bound to the surface, i.e. its freedom is restricted. Hence, entropy decreases, i.e. ΔS < 0.
For a reaction be to be spontaneous, Gibbs free energy, ΔG< 0.
According to the equation ΔG = ΔH - TΔS, ΔG can be negative only if ΔH is significantly negative, as - TΔS is positive.
We know that for adsorption, H <0, ΔS < 0
If initially, at constant temperature and pressure, ΔH is significantly negative, then the combination of these two factors make ΔG negative.
As adsorption proceeds, ΔH becomes less and less negative such that after some time ΔH becomes equal to TΔS and ΔG becomes zero. At this point, equilibrium is attained.
Enthalpy of Adsorption (∆HAdsorption)
The amount of the heat released when 1 mol of an adsorbate gets adsorbed on a particular adsorbent at adsorption equilibrium. It depends on the nature of both the adsorbent and the adsorbate.
Types of Adsorption
Physical Adsorption or Physisorption

In physisorption, the accumulation of gas on the surface of a solid occurs on account of weak van der Waals’ forces. In physisorption, due to the presence of weak van der Waals’ forces, the accumulation of gas on the surface of a solid occurs. Generally, it occurs at low temperatures and is multi-layer adsorption.

Characteristics of Physisorption
- Lack of Specificity: A given surface of an adsorbent does not show any preference for a particular gas as van der Waals’ forces are universal. As van der Waals’ forces are universal, a given surface does not have any particular preference of gas.
- Nature of the Adsorbate: The amount of gas adsorbed by a solid depends on the nature of the gas. The nature of the gas influences the amount of gas adsorbed by a solid. Easily liquefiable gases that have a higher critical temperature (TC) are readily adsorbed. For instance, 1 g of activated charcoal adsorbs more sulphur dioxide than methane as the critical temperature of sulphur dioxide (TC(SO2) = 630 K ) is greater than the critical temperature of methane (TC (CH4)= 190 K).
Solid + Gas ⇋ Gas/solid + Heat
3. Reversible Nature: Physical adsorption of a gas by a solid is generally reversible. By a solid, the physical adsorption of a gas is generally reversible.
Solid + Gas ⇋ Gas/solid + Heat
More gas is adsorbed when pressure increases, as the volume of gas decreases (Le–Chateliers’s principle). The gas can be removed by decreasing the pressure. According to Le Chatelier’s principle, since adsorption is exothermic, physical adsorption occurs readily at low temperatures and decreases with increasing temperatures. According to Le Chatelier’s principle, since adsorption is exothermic, at low temperatures, physical adsorption occurs readily and on increasing the temperature, it decreases.
4. Surface Area of the Adsorbent: The extent of physisorption increases with an increase in the surface area of the adsorbent. Thus, finely divided metals and porous substances having large surface areas are good adsorbents. Thus, porous substances and finely divided metals are good sdsorbents as they have large surface areas.
5. Enthalpy of Adsorption: Physical adsorption is an exothermic process but the enthalpy of adsorption is very low (20– 40 kJ mol-1). Though physical adsorption is an exothermic process, the enthalpy of adsorption is very low (20– 40 kJ mol-1). This is due to the fact that the attraction between gas molecules and the solid surface is due to weak van der Waals’ forces.
Chemical Adsorption or Chemisorption

In chemical adsorption or chemisorption, the gas molecules or atoms of the adsorbate are held to the solid surface of the adsorbent by the chemical bonds. Chemical bonds may be covalent or ionic in nature. It generally, occurs at high temperatures and is monolayer adsorption. As chemisorption involves high activation energy, it is often referred to as activated adsorption.
Characteristics of chemisorption
1. High Specificity: Chemisorption occurs only if there is a possibility of chemical bonding between adsorbent and adsorbate and is highly specific. Chemisorption is highly specific and occurs only if there is a possibility of chemical bonding between adsorbate and adsorbent.
Example: Hydrogen is adsorbed by transition metals, due to hydride formation. Due to hydride formation, transition metals adsorb hydrogen.

2. Irreversibility: As chemisorption involves compound formation, it is usually irreversible in nature. Chemisorption is generally irreversible in nature as it involves the formation of a compound. Chemisorption, like physisorption, is also exothermic. But due to high activation energy, it is slow at low temperatures. Like most chemical reactions, chemisorption increases with the increase in temperature.
3. Surface Area of the Adsorbent: Like physisorption, chemisorption also increases with an increase in the surface area of the adsorbent.
4. Enthalpy of Adsorption: As chemical bonds are formed in chemisorption, the enthalpy of formation is high (80– 240 kJ mol-1).
Difference between Physisorption and Chemisorption
|
Property |
Physisorption |
Chemisorption |
|
Nature of adsorption |
Weak (van der Waals’ forces) |
Strong (Chemical bond formation) |
|
Specificity of adsorption |
Not specific |
Highly specific |
|
Reversibility of adsorption |
Reversible |
Irreversible |
|
Temperature at which adsorption is more pronounced |
Low temperatures |
High temperatures |
|
Effect of change in temperature |
Decreases with increase in temperature |
Increases with increase in temperature |
|
Enthalpy of adsorption |
Low (20-40 kJ mol-1) |
High (80-240 kJ mol-1) |
|
Nature of adsorbate layer |
Multi-layered (at high pressure) |
Mono-layered |
|
Energy of activation |
Low |
Significantly high |
|
Rate |
Rapid/Fast |
Slow |
|
Ease of desorption |
Easy, since van der Waals forces are involved |
Not easy, since chemical forces are involved |
|
Nature of gas |
Easily liquefiable gases are adsorbed readily |
Gases which can react with the adsorbent show chemisorption |
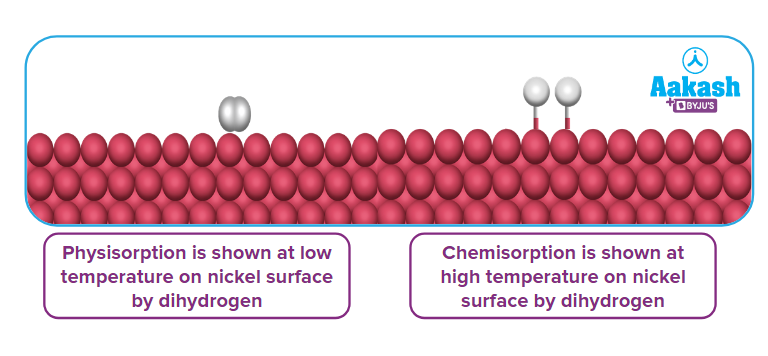
Physisorption, at low temperature, may pass into chemisorption, as the temperature is increased.
Example: Initially H2 is adsorbed on the surface of nickel by Van der Waals' forces of attraction. Later as temperature increases molecules of hydrogen dissociate to form hydrogen atoms (H) which are held on the surface of nickel by chemisorption.
Factors Affecting the Adsorption of Gases on Solids
- Nature of gases (adsorbate): Extent of adsorption is directly proportional to the ease of liquefaction, which is directly proportional to the critical temperature of the gas.
- Nature of the adsorbent: The extent of adsorption is directly proportional to the surface area of the solid.
So, finely divided adsorbents are more efficient than the normal ones.
Example: Activated charcoal, clay etc., can adsorb gases which are easily liquified. Gases such as H2, N2 and O2 are generally adsorbed on finely divided transition metals Ni and Co.

- Effect of temperature
Adsorbent (s) + Adsorbate (g) ⇌ [Adsorbent---Adsorbate] + ΔH
We know that adsorption is an exothermic process and desorption is an endothermic process. So, according to Le Chatelier’s principle, an increase in temperature favours desorption.
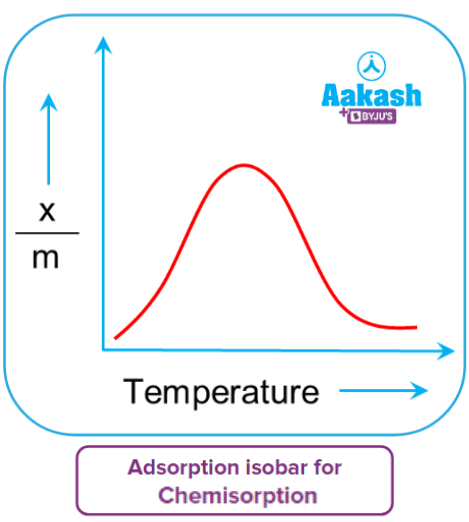
The variation in the amount of gas adsorbed by the adsorbent with temperature can be expressed by a curve termed as adsorption isobar. It is a plot of 

Adsorption isobars are plotted separately for physisorption and chemisorption. With the increase in temperature, physical adsorption decreases continuously or the gas is removed from the surface.

Chemical adsorption increases initially reaches a maximum then decreases continuously. The initial increase in chemisorption is caused by the heat supplied acting as activation energy, and more and more adsorbate molecules gain energy and possess energy greater than activation energy. As a result, adsorption increases as temperature rises.
- Effect of pressure
The extent of adsorption of a gas (

The extent of adsorption (
Consider a beaker fitted with a piston as shown below. Charcoal is placed at the bottom of the beaker and NO2 gas is filled in the beaker.
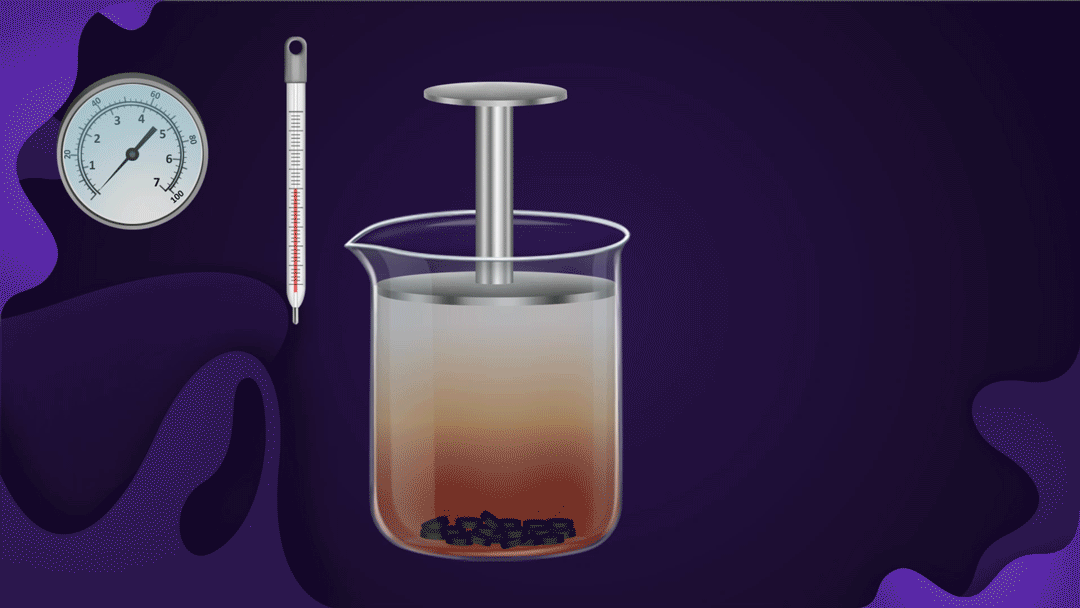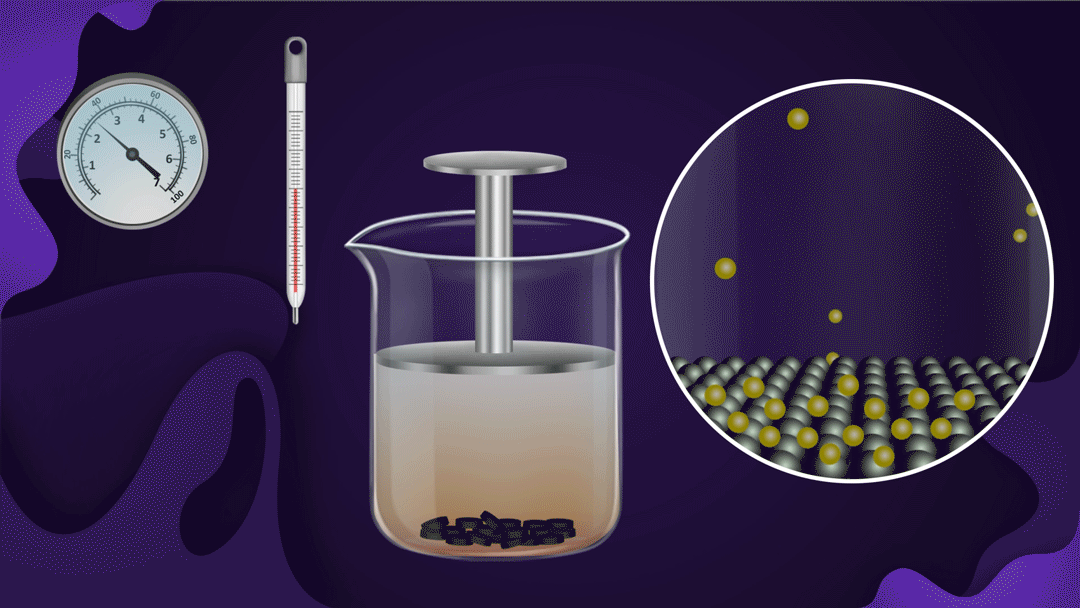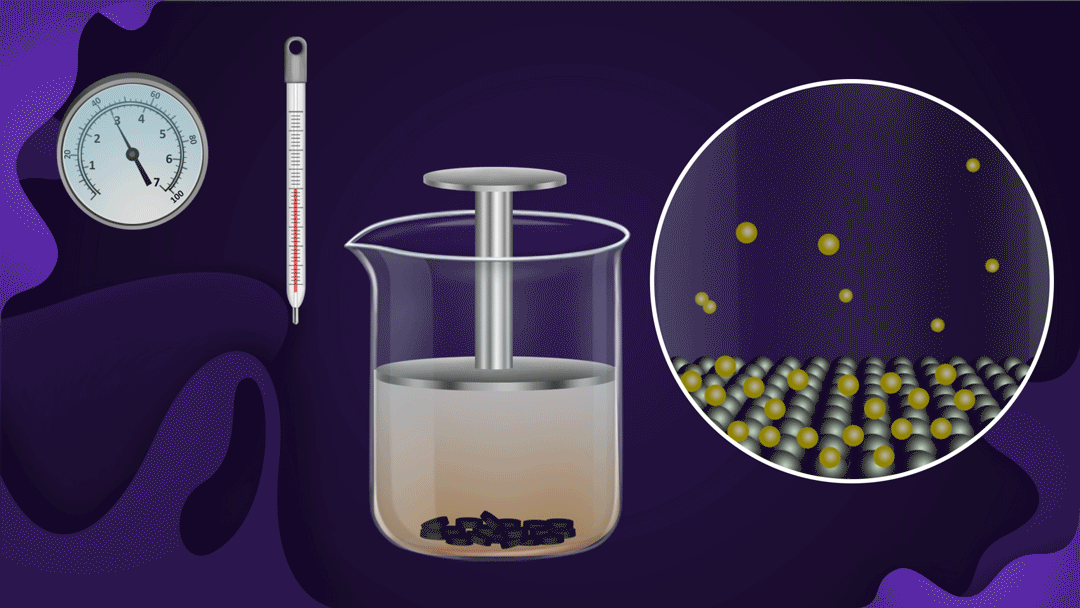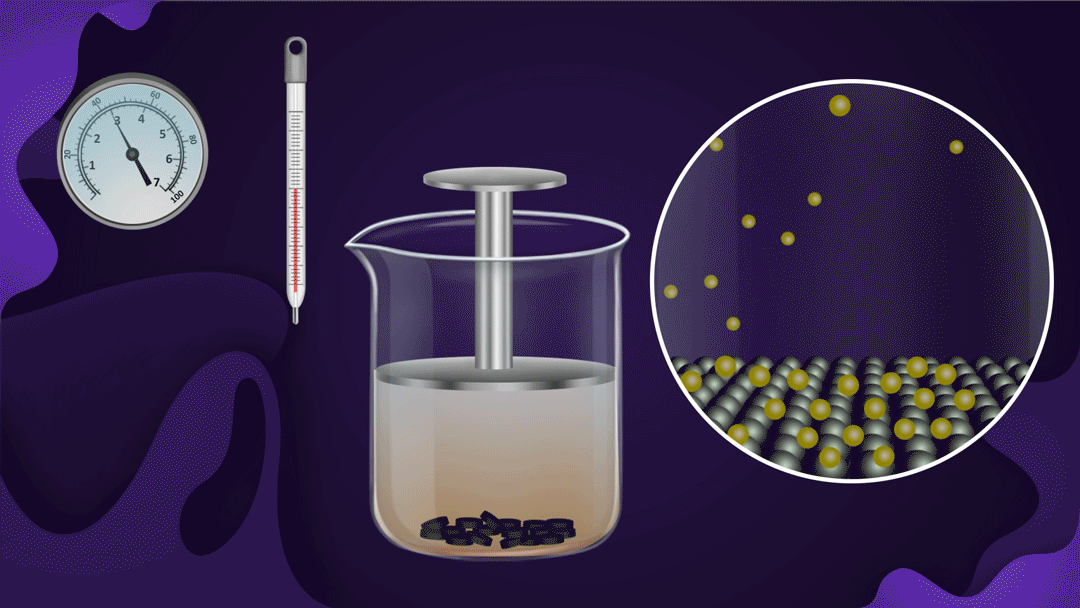
When the piston is pressed at constant temperature, the pressure of the gas increases, which increases the adsorption of NO2 gas on the charcoal. Increased adsorption can be seen by observing the decrease in the colour intensity of the NO2 gas as the piston moves inwards.
There are two isotherms that explain the effect of pressure on adsorption, empirically.
1. Freundlich Adsorption isotherm
2. Langmuir Adsorption isotherm
Note: These isotherms are studied for physical adsorption, only.
Practice Problems
Q1. How can we increase the surface area of an adsorbent?
A. Adsorbent can be subdivided into smaller pieces
B. Adsorbent are used in large pieces
C. Surface area of the absorbent doesn’t matter.
D. None of the above
Solution: Adsorption increases with the increase in surface area of the adsorbent. Therefore, to increase adsorption, the adsorbent can be divided into smaller pieces such that the surface area is increased. This improves the absorption power of the adsorbent. So, option A) is the correct answer.
Q2. The given plot is for which type of adsorption?

A. Physisorption
B. Chemisorption
C. Both A and B
D. None of the above
Solution: The plot of extent of adsorption vs temperature decreases continuously for physisorption or physical adsorption. For chemisorption, the plot of extent of adsorption vs temperature increases initially, reaches a maximum, then decreases continuously.
Q3. With the increase in surface area, ____ also increases.
A. Physisorption
B. Chemisorption
C. Both A and B
D. None of the above
Solution: As the surface area of the adsorbent increases, more adsorbate molecules get adsorbed on the surface of the adsorbent. The extent of adsorption is directly proportional to the surface area of the solid for both physisorption and chemisorption. Therefore, with the increase in surface area, both physisorption and chemisorption increases. So, option C) is the correct answer.
Q4. Which of the following statements is correct regarding chemisorption?
A. It is irreversible in nature.
B. It is highly specific in nature.
C. Has higher enthalpy of adsorption.
D. All of the above
Solution: As chemisorption involves compound formation, it is usually irreversible in nature. Chemisorption occurs only if there is a possibility of chemical bonding between adsorbent and adsorbate. Therefore it is highly specific in nature. As chemical bond formation is involved, the enthalpy of adsorption of chemisorption is high. Hence, all the given statements are correct regarding chemisorption. So, option D) is the correct answer.
Frequently Asked Questions - FAQ
Question 1. Why no chemical bond is formed in physisorption?
Answer: Physisorption usually occurs at low temperatures. At these temperatures only weak van der Waals’ forces exist. At low temperatures, there is not enough activation energy to form chemical bonds. So, chemical bonds are not formed in physisorption,
Question 2. What is the critical temperature of gases?
Answer: The critical temperature of a substance is the temperature at and above which vapour of the substance cannot be liquefied, no matter how much pressure is applied.
Question 3. Why easily liquefiable gases readily adsorbed?
Answer: The van der Waals’ force of attraction are greater in easily liquefiable gases like NH3, SO2, etc than in permanent gases like H2, N2, etc. As a result, easily liquefiable gases are absorbed readily.
Question 4. Why is the enthalpy of adsorption low for physisorption?
Answer: As weak physical forces like van der Waals’ forces are involved in physisorption, the attraction between the adsorbate and the adsorbent is less. This is the reason why the enthalpy of adsorption is low for physisorption.
Related Topics
|
Preparation of Lyophobic Colloids - Condensation Methods, Dispersion Methods |
Osmosis and Osmotic Pressure |
|
Properties of Colloids |
Coagulation of Colloids |
|
Emulsion |
Colloidal Solutions |



