-
Call Now
1800-102-2727
Plant Water Relations: Water Potential, Solute Potential, Pressure Potential, Osmosis, Osmotic Pressure and Osmotic Potential
We are often advised to drink sufficient water throughout the day for healthy functioning of the body. How is water transported to all the cells of our body? It is transported through blood, right? Have you wondered how water from the gut is transported against gravity to body parts that lie above the gut region? The answer is easy, we have a heart that pumps the blood to different parts of the body, even against gravity.
But what about the plants? Plants need water for photosynthesis and to maintain form and structure of the plant cells and they obtain this water by absorption from the soil. So how do you think water from the ground travels upwards and reaches the aerial parts of the shoot system?
There are multiple forces involved in this process and some of the primary factors that determine the direction of movement in the plant body are water potential, solute potential, pressure potential, osmotic potential and osmotic pressure of the plant cells. Let us try and understand more about these concepts.
Table of contents:
- Transport of water in plants
- Water potential
- Osmosis
- Diffusion pressure deficit
- Practice problems
- FAQs
Importance of water in plants
Before we get into the details of how water is transported in a plant, let us first understand the importance of water in the plant body. The plant cells require water for various metabolic functions, photosynthesis being one of them. Water also helps to maintain turgidity of the plant cells and is needed for the transport of minerals and translocation of prepared food to different plant parts. Seeds need water for germination and transpiration of water helps to cool down the plant body.
Transport of water in plants
Only 1% of the water absorbed is used for photosynthesis and most of the water absorbed by the plants is lost through transpiration. But the absorbed water has to reach every part of the plant, although the distribution of water in different parts of the plant does vary. Transport of water within the plant can be across short distances or across long distances.

Fig: Types of water transport
Long distance transport
Long distance transport is the transport of water from the roots to the different parts of the plant. It happens through the xylem tissue.

Fig: Long distance transport of water
Short distance transport
The transport of water at a cellular level is called short distance transport. This occurs when water enters the cell from the environment and from one cell to the other. There are two boundaries that the water has to cross before it enters the cells- the freely permeable cell wall and the selectively permeable cell membrane.
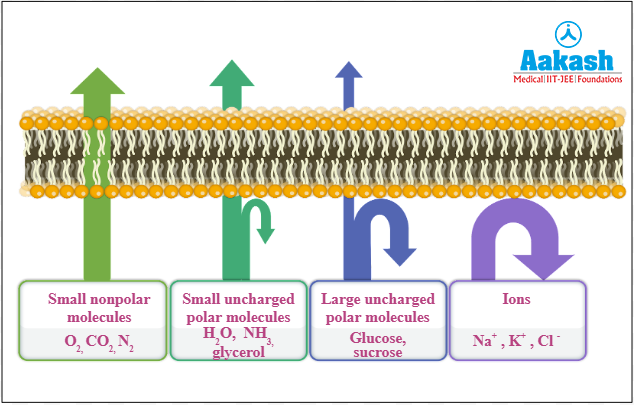
Fig: Transport of substances through cell membrane
But what drives the movement of water out of one cell and into another cell? The answer to this can be found through a U tube experiment.
U tube experiment
A U-shaped tube with a selectively permeable membrane in the middle is taken. Then equal amounts of water is added on both sides. At this stage, pure water (with just water molecules) is present in both arms of the U-tube apparatus. Kinetic energy of water molecules in pure water is the maximum.
Then glucose (solute) is added into the water on the left side of the tube and is dissolved well. The addition of glucose into pure water initiates an interaction between the glucose molecules and the water molecules which prevents the water molecules from moving freely. This lowers the kinetic energy of water molecules.
After a while, the water level in the left side is observed to have increased and that in the right side to have decreased. This shows that there is a movement of water from the right side to the left side (from region of higher kinetic energy to region of lower kinetic energy). This happens due to the diffusion of water molecules across the semipermeable membrane. This difference between the free energy of water molecules in a pure system and a solution can be measured in terms of water potential.
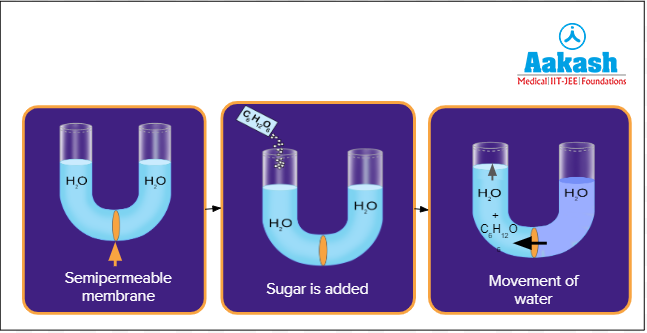
Fig: U Tube experiment
Water potential
From the U tube experiment we found that random movement of water molecules is due to kinetic energy of the water molecules and pure water has the highest kinetic energy. The kinetic energy of water molecules can also be represented in terms of water potential.

Fig: Conclusion of U tube experiment
Water potential is the measure of concentration and free energy of water in a system per unit volume. Water concentration is the amount of water molecules in a unit volume of a system. Here free energy is the energy available for the molecules to move freely.

GIF: Movement of water molecules in a pure system
Higher the concentration of water in a system, greater is its kinetic energy or ‘water potential’. Thus, pure water has the greatest water potential. Movement of water always occurs from a region of high water concentration (high kinetic energy) to a region of low water concentration (low kinetic energy). Thus water will move from a system containing water at higher water potential to the one having lower water potential.
Water potential is denoted by a Greek symbol psi or Ψw. It can be expressed in the unit of pressure known as pascal. The water potential of pure water, which is at standard temperature and not under any pressure, is assigned to be zero.
Ψw of pure water = 0
What happens when solute is added to water?
When a solute is added to water, some of the free water molecules bind to the solute which decreases the number of water molecules that can move freely. As a result the overall free energy as well as the concentration of water decreases.
Thus the water potential of a solution is always less than the water potential of pure water. Since the water potential of pure water is zero, the water potential of a solution is always negative.
Ψw of a solution = Negative

GIF: Reduced movement of water molecules in a solution
Factors affecting water potential
There are two major factors that affect the water potential. They are solute potential and pressure potential. Let’s discuss more about it.
Solute potential (Ψs)
The magnitude of the decrease of water potential from pure water due to the dissolution of a solute is known as solute potential or osmotic potential. Solute potential is represented by Ψs. Ψs is always negative as the water potential of pure water is zero and the addition of solutes decreases its potential.

Fig: Relationship between solute concentration and solute potential of a solution
Solute potential can be calculated by the formula,
Ψs = CRT
C is the concentration of solute particles
R is the gas constant
T is the temperature in Kelvin scale
Pressure potential (Ψp)
The magnitude of the change of water potential due to the mechanical pressure generated as a result of diffusion of water molecules into the cell and their inward movement into the cell sap is known as pressure potential. Pressure potential is represented by Ψp. Ψp can be positive or negative.
Hence the equation of water potential can be written as:

Fig: Formula for water potential
But at atmospheric pressure, the pressure potential is 0. Hence the water potential will be equal to solute potential.

Fig: Value of water potential at atmospheric pressure
At atmospheric pressure, the pressure potential is zero and the solute potential of the pure water is also zero. Hence the water potential of pure water will also be zero.

Fig: Water potential of pure water at atmospheric pressure
Positive and negative pressure potential
How does pressure potential apply to a plant cell? Let's do an experiment to understand this.
To conduct this experiment we need a balloon, water, beaker and straw. In the first scenario, we shall take the balloon, place it inside the beaker and then start filling it with water. As we add more and more water, the balloon expands and eventually starts applying pressure on the walls of the beaker.
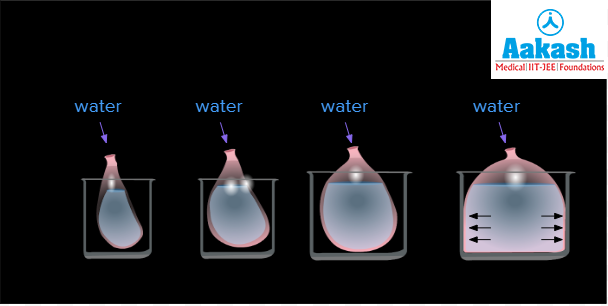
Fig: Scenario 1
Just like this when water enters a regular plant cell through diffusion, the cell contents expand and apply pressure on the cell walls of the plant. This is an example where the pressure potential is positive. As this occurs in cells which are turgid and swollen with their cell membrane pressing against their cell wall, the pressure potential is also referred to as turgor pressure.

Fig: Positive Pressure Potential
In the 2nd scenario, we will insert a straw into the balloon filled with water and start sucking through the straw. As we start sucking, it creates a negative pressure in the straw that causes the water to go up and it starts coming out of the balloon. If we keep sucking the water out of the balloon through the straw, eventually the whole balloon will become empty. This is an example of negative pressure potential.
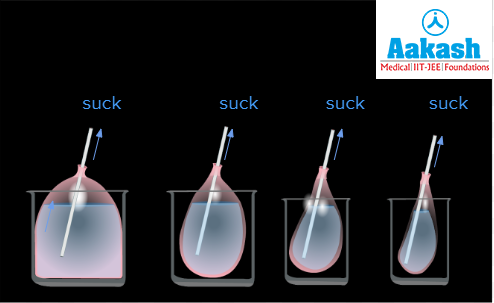
Fig: Scenario 2
Negative pressure potential is seen in the xylem tissue that transports water. When the water is lost from the leaf surface through the stomata, this creates a suction or negative pressure that causes the water in the xylem to be sucked in, and this negative pressure in the xylem is what causes the water from the soil to be sucked by the roots also.

Fig: Negative pressure potential
Osmosis
The diffusion of water across a selectively permeable or differentially permeable membrane from its region of higher concentration to its region of lower concentration is osmosis. Direction and rate of osmosis is dependent on two factors - pressure gradient and concentration gradient.
In fact, the movement of water molecules observed in the U-tube experiment, from the region of pure water to the region of sugar solution, across the semipermeable membrane is osmosis. So during osmosis, the water molecules move from the region of higher water potential to lower water potential until equilibrium is established. As more water flows into the left arm of the U-tube, the solute concentration on the left side decreases, meaning the solution becomes more dilute.

Fig: Osmosis
To understand more about osmosis we can conduct another experiment called potato osmometer experiment. Let’s see how it is done.
Potato osmometer experiment
In this experiment, when a potato with some sugar solution is placed in a beaker containing water, after a while, we can see that the water level outside the potato slightly decreases, while the water level inside the potato increases. This is because there is a higher solute concentration and a lower water potential inside the potato, while the outside has a lower solute concentration and a higher water potential. So, there is osmosis, i.e., movement of water from outside to inside.
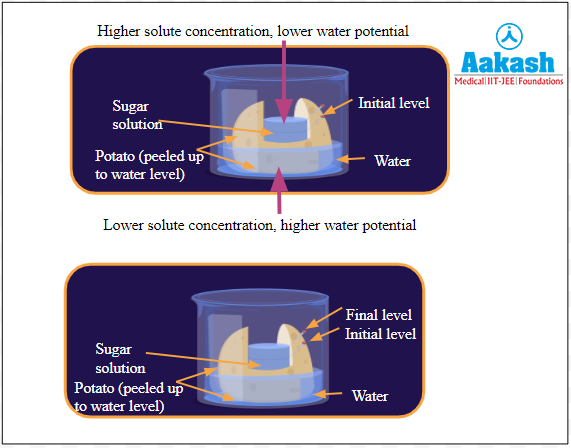
Fig: Potato osmometer experiment
So now we can conclude that during osmosis water moves from higher water potential to lower water potential or from low solute concentration to high solute concentration.
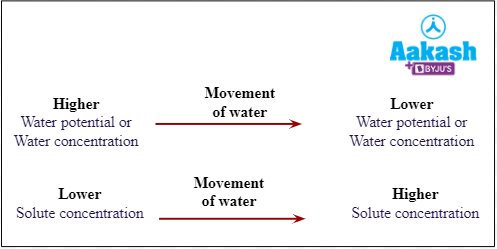
Fig: Direction of movement of water during osmosis
Now let us conduct an experiment to see how we can manipulate the movement of water during the process of osmosis.
Thistle funnel experiment
Requirements
Requirements for this experiment are a thistle funnel, rubber band, cellophane paper (semipermeable membrane), sugar and water.

Fig: Thistle funnel
Procedure of the experiment:
First we will tie the cellophane paper around the mouth of the funnel with a rubber band. Then we will fill it with a sugar solution. And place it in a beaker containing pure water.
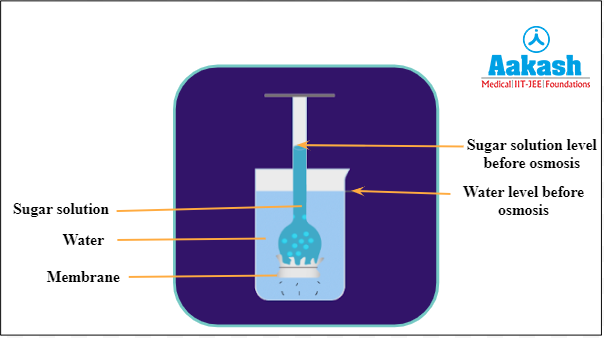
Fig: Initial stage of thistle funnel experiment
In this scenario due to osmosis, the solution level in the funnel starts increasing. As the water potential in the beaker is higher, the water moves from the beaker into the funnel, across the semipermeable membrane, until equilibrium is reached.
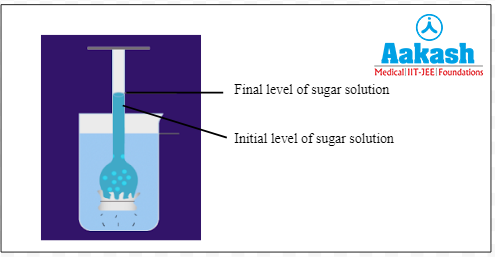
Fig: Final stage of thistle funnel experiment
Now we will check another scenario where we add a piston in the funnel to apply pressure on the sugar solution and place this setup into the beaker with water. Here there will be no movement of water even though there is a difference in the water potential of the sugar solution in the funnel and the water in the beaker. The pressure that is required to prevent the water from diffusing or undergoing osmosis is known as osmotic pressure. Hence we can say osmotic pressure is directly proportional to solute concentration, that is, osmotic pressure increases with increase in solute concentration.
Relation between osmotic pressure and water potential
Remember that OP stands for osmotic pressure and not osmotic potential. Osmotic potential is equal to solute potential. The addition of more solute means that more pressure is required to stop the flow of water. Hence, osmotic pressure or OP is the negative version of osmotic potential.

Fig: Relationship between water potential and osmotic pressure
Hence, Water potential = Pressure potential - Osmotic pressure
Reverse osmosis
Let's consider another scenario of thistle funnel experiment which explains the reverse osmosis. If a pressure greater than the osmotic pressure is applied, then not only will the water not move into the thistle funnel, but the opposite will occur. The water will start moving from the sugar solution into the beaker. Remember that this is a movement from lower water potential to higher water potential. So, it is not osmosis, but it is reverse osmosis.
Reverse osmosis is a process by which a solvent passes through a selectively permeable membrane in the direction opposite to that which is natural for osmosis. It happens when subjected to a pressure greater than the osmotic pressure. This is exactly what takes place in an RO water filtration system.

Fig: RO water filtration system
Diffusion pressure deficit (DPD)
Pure water has maximum diffusion pressure due to high kinetic energy. Addition of solute particles to water lowers its diffusion pressure. The lowering of the diffusion pressure of water in a solution over its pure state is known as diffusion pressure deficit (DPD). Basically, it is another term for water potential, but it is always positive in magnitude. Water potential and DPD have the same value but water potential is negative.
Water potential = - (Diffusion pressure deficit)
Hence, we can say that water moves from a lower DPD to a higher DPD as long as there is diffusion pressure deficit.
In a cell, diffusion pressure deficit is the difference between its osmotic pressure and the wall pressure (pressure exerted by the cell wall of a turgid cell on to the cell contents) or turgor pressure (pressure exerted by the cell contents of a turgid cell on the cell wall).
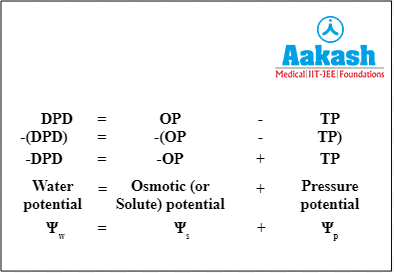
Fig: Relationship between DPD and OP
Practice Problems
Q1. If a plant cell’s 𝜳p = 2 bars and its 𝜳s = -3.5 bars, what is the resulting 𝜳w?
A. 0
B. -1.5 bars
C. 1.5 bars
D. 5.5 bars
Solution: 𝜳s (solute potential or osmotic potential) is the magnitude of decrease of water potential with respect to that of pure water due to the dissolution of a solute. 𝜳p (pressure potential also called as turgor pressure or turgor potential) is the pressure exerted by the swelling protoplast on its walls due to the osmotic entry of water into it. 𝜳w (water potential) is the difference in potential of water in a system over its pure state at the same temperature and pressure. The water potential of pure water is zero and it is negative for any solution.
𝜳w = 𝜳p + 𝜳s = 2 bars + (-3.5 bars) = -1.5 bars.
Hence the correct option is b.
Q2. Cell ‘A’ with OP = 10 atm and TP = 5 atm is in contact with cell ‘B’ having OP = 15 atm and TP = 12 atm. The flow of water will be
A. from A to B
B. equal flow
C. from B to A
D. no flow
Solution: We know that, DPD = OP - TP.
In cell A: DPD = 10 - 5 = 5 atm. In cell B: DPD = 15 - 12 = 3 atm. Diffusion pressure deficit (DPD) is the reduction in diffusion pressure (DP) of water in a system over its pure state. Diffusion pressure is the pressure exerted by the molecules that are undergoing diffusion. DP of pure solvent is maximum. DPD determines the direction of movement of water. Water always moves from an area of lower DPD to the area of higher DPD. In this case cell B has a lower DPD than cell A. Thus, the flow of water will be from cell B to A.
Hence the correct option is c.
Q3. Exchange of substances between a cell and its environment can occur through
A. osmosis
B. diffusion
C. active transport
D. all of the above
Solution: Diffusion is the movement of molecules (solid, liquid, gas) from a region of higher concentration to a region of lower concentration till a level of equilibrium is reached. Osmosis is the movement of solvent molecules (water) from lower solute concentration to a solution having higher solute concentration when the two solutions are separated by a semipermeable membrane.
Active transport is the movement of molecules against the concentration gradient by utilising energy. All these are the processes carried out by the cell in order to exchange substances with its environment.
Hence the correct option is d.
Q4. What happens if turgor pressure becomes equal to osmotic pressure?
A. water leaves the cell
B. water enters the cell
C. no exchange of water takes place
D. solute pass out of the cell
Solution: The pressure that is required to prevent water from diffusing or undergoing osmosis in spite of a difference in water potentials of two systems, separated by a semipermeable membrane, is called osmotic pressure (OP). Turgor Pressure (TP) is the pressure exerted by the swelling protoplast on its walls due to the osmotic entry of water into it. Diffusion pressure deficit (DPD) is the reduction in diffusion pressure (DP) of water in a system over its pure state. Water always moves from an area of lower DPD to the area of higher DPD.
DPD = OP - TP
When turgor pressure (TP) becomes equal to osmotic pressure (OP), there will be no DPD (DPD = 0). Thus, there will be no exchange of water.
Hence the correct option is c.
FAQs
Q1. What is soil water potential?
Answer: Soil water potential is formally defined as (Aslyng, 1963) "the amount of work that must be done per unit quantity of pure water in order to transport an infinitesimal quantity of water from a reference pool. It can be transported reversibly (independent of path taken) and isothermally to the soil water at a considered point."
Q2. What tool is used to determine the water potential of leaf tissue?
Answer: Only thermocouple psychrometers can measure the in situ water potential of intact leaves and may be used to monitor leaf water potential.
Q3. What are osmometers?
Answer: Osmometers are instruments that are used to measure the osmotic pressure and osmotic concentrations of samples. An osmometer measures osmotic concentration in terms of milliosmoles per unit of weight (mOsm/kg).
Q4. What role does osmosis play in the human body?
Answer: Osmosis is vital in the human body, particularly in the gastrointestinal and renal systems. Osmosis aids in the extraction of nutrients from meals. It also removes waste items from our bloodstream and helps in osmoregulation, that is, regulation of constant osmotic balance of fluids in the body.


