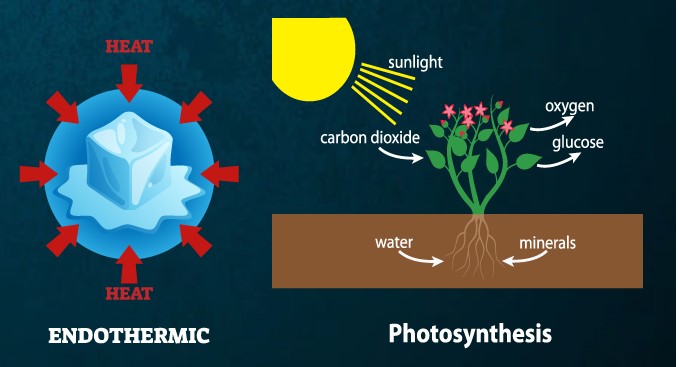
Two Greek terms are combined to form photosynthesis. ‘Phos’ is a Greek word that signifies “light”, and ‘sunthesis’ means “combining.” This is defined as “combining with the aid of light.”
Photosynthesis is the conversion of sunlight, carbon dioxide (CO2), and water into food (sugars) and oxygen by plants, algae, and some microorganisms. Light energy is collected and used by green plants during the process to convert water, carbon dioxide, and minerals into oxygen and energy-rich organic molecules.
The phenomenon of photosynthesis is extremely significant for the sustenance of life on Earth. If photosynthesis is stopped, there will be little or no food or other organic materials on Earth. Most life would perish, and the Earth’s atmosphere would eventually be almost empty of gaseous oxygen. The whole ecological balance would be disrupted.
Meaning of Photosynthesis
Photosynthesis produces glucose, which is subsequently utilised to power numerous cellular functions. This physio-chemical process produces oxygen as a byproduct.
Glucose molecules (or other sugars) are made from carbon dioxide and water in a light-driven process, with oxygen released. Glucose molecules give two essential resources to organisms: energy and fixed organic carbon.
- Energy: ATP or Adenosine Triphosphate is a tiny energy-carrying molecule that fulfils the immediate energy demands of the cell. Processes like cellular respiration and fermentation produce this ATP.
- Fixed Carbon: Carbon from carbon dioxide (inorganic carbon) may be integrated into organic molecules, a process referred to as carbon fixation. The carbon in organic molecules is referred to as fixed carbon. During photosynthesis, the carbon that is fixed and integrated into sugars may be utilised to produce different types of organic compounds that organisms require. Industrial chemicals and other particles cause pollution if they settle on the leaf surface. This may clog stomatal pores, making it harder to absorb carbon dioxide.
Equation
6CO2 + 6H2O + Sunlight ———> C6H12O6 + 6O2
(Carbon Dioxide + Water + Sunlight ———> Glucose (simple sugar) + Oxygen)
Using light energy, six carbon dioxide molecules (CO2) mix with 12 water molecules (H2O). The final output is a single carbohydrate molecule (C6H12O6, or glucose) and six oxygen and water molecules each.
The different anoxygenic photosynthesis processes can also be summarised into a single formula:
[CH2O] + 2A + H2O = CO2 + 2H2A + Light Energy
The symbol A stands for a variable in the equation, while H2A stands for a potential electron donor. According to the medical and life sciences news site News Medical Life Sciences, “A” might stand for sulphur in the electron donor hydrogen sulphide (H2S).
Also visit our important concept pages to know more about this process.
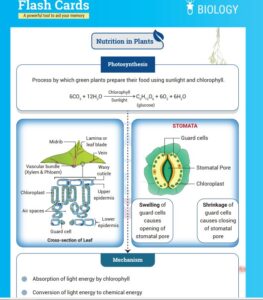
Process of Photosynthesis
The phenomenon of photosynthesis consists of two stages, light reaction and dark reaction.
- Light reaction: Light reaction occurs exclusively during the day in the presence of sunlight. The light-dependent reaction takes place in the thylakoid membranes of chloroplasts in plants. Inside the thylakoid are some structures surrounded by a membrane and are known as Photosystems. These photosystems play a key role in the whole process of photosynthesis. Photosystems are divided into two categories: photosystem I and photosystem II.
The light energy is converted to ATP and NADPH through light-dependent processes used in the second phase of photosynthesis. Two electron-transport chains, water, produce ATP and NADPH during light reactions.
Chemical Equation
2H2O + 2NADP+ + 3ADP + 3Pi → O2 + 2NADPH + 3ATP
- Dark reaction: Carbon-fixing reaction is another name for a dark reaction. In a light-independent process, sugar molecules are generated from water and carbon dioxide molecules. This reaction occurs in the stroma of the chloroplast, where the light reaction’s NADPH and ATP products are used. Plants take carbon dioxide from the environment through stomata and enter the Calvin photosynthesis cycle. ATP and NADPH generated during the previous stage fuel the Calvin cycle and convert six carbon dioxide molecules into one sugar molecule, or glucose.
Chemical Equation
G3P + 2H+ + 6 NADP+ + 9 ADP + 8 PiT = 3CO2 + 6 NADPH + 5H2O + 9ATP
Factors affecting Photosynthesis
- Light Intensity: As the intensity of light rises, the rate of the process of photosynthesis increases as well. Low light intensity leads to a decreased rate of photosynthesis. The CO2 concentration is as follows: This rate is aided by a higher carbon dioxide concentration. Carbon dioxide in 300–400 PPM is usually sufficient for photosynthesis.
- Temperature: A temperature range of 25° to 35° C is required for effective photosynthesis. When the temperature is low, the rate of photosynthesis is limited by the number of molecular collisions between enzymes and substrates and when the temperature is high, enzymes are denatured.
- Water: Since water is such an indispensable component of photosynthesis, a lack of it might cause issues with carbon dioxide absorption. Due to a lack of water, stomatal openings do not keep the water stored inside.
- Pollution: Pollutants from industry and other particles may settle on the surface of the leaves. This blocks the pores of stomata, making it difficult to take in carbon dioxide.
Types of Photosynthesis
- Oxygenic photosynthesis is more prevalent than anoxygenic photosynthesis, occurring in algae, plants, and cyanobacteria. Light energy takes electrons from water (H2O) to CO2 to make carbohydrates during oxygenic photosynthesis. The CO2 is “reduced” or gains electrons, while the water is “oxidised” or loses electrons in this process. Carbohydrates and oxygen are both created. Oxygenic photosynthesis is similar to respiration but occurs oppositely. It absorbs CO2 generated by all breathing creatures and returns oxygen to the atmosphere.
- Anoxygenic Photosynthesis: Anoxygenic photosynthesis does not create oxygen and uses electron donors other than water. Some examples that go through this process are phototrophic purple bacteria and green sulphur bacteria.
CBSE Exam Class 10 Syllabus 2026, Sample Papers
- CBSE Class 10 Date Sheet 2026
- CBSE Class 10 Syllabus 2026
- CBSE Class 10 Previous Years Question Papers with Solutions | Aakash
- CBSE Class 10 Sample Papers 2026
- CBSE Class 10 Mock Test Paper Solutions
- CBSE Class 10 Science Sample Paper Solutions
Importance of Photosynthesis
Photosynthesis’s fundamental job is to transform solar energy into chemical energy and store it for future use. This mechanism is responsible for the majority of the planet’s life systems. It’s sobering to contemplate that our bodies’ energy traverses 93 million miles in less than eight minutes. It is also important to note that life has tapped into that energy stream. That energy is stored in biological systems for a brief period before continuing on its trip into the darkness of space.
Chloroplasts are the parts of a cell where photosynthesis takes place. Green plants absorb carbon, hydrogen, and oxygen from water and carbon dioxide molecules and recombine them into a new molecule called glucose. Of course, this occurs in the presence of sunshine. The bonds of the glucose molecule store energy. Glucose is a basic sugar that is easy to digest.
Trees and other green plants, like animals, undertake to breathe, but they also practise photosynthesis. This is why ecologists classify green plants as “producers”, whereas most other living things are classified as “consumers.”
Photosynthesis produces oxygen, while respiration produces carbon dioxide as a byproduct. Trees are frequently attributed as the planet’s primary source of oxygen, although this is incorrect. The major portion of the earth is covered with water, and humble algae’s collective photosynthesis is the genuine oxygen machine. Trees and forests are substantial oxygen generators. We could easily survive without trees and forests if the only advantage they provided was oxygen. Furthermore, some woods create more carbon dioxide than oxygen. Fortunately, the advantages of trees and forests are substantially more extensive than that.
Cellulose, a very complex sugar, makes up a large portion of the essential structural substance of plants and wood. Carbon, hydrogen, and oxygen molecules may be recombined to make a variety of useful compounds, including ethanol, fragrances, bioplastics, textile fibers, and a variety of industrial components. It is well acknowledged that sources derived from renewable living ecosystems have different benefits over fossil fuels derived from old materials.
Watch out our video on Photosynthesis.
Conclusion
Plants and photosynthesis are the foundation of fossil fuels, but they date back millions of years. Introducing large quantities of those molecules back into live ecosystems has several downsides that science has grown fairly adept at detecting and characterising. Trees, forests, forest soils, and forest products play a significant role in the environment in carbon cycling and the relative size of different carbon pools. Other elements circulate in the woods as well. Science, too, has a strong grip on these connections.
Photosynthetic organisms might be used to produce clean-burning fuels like hydrogen. A research team from the University of Turku in Finland used green algae’s potential to make hydrogen. If green algae are subjected to dark and anaerobic situations before being exposed to light, they can create hydrogen for a few seconds.
Continued study of natural processes helps scientists identify new methods to use varied sources of renewable energy, and leveraging the power of photosynthesis is a reasonable first step towards making clean-burning, carbon-neutral fuels.